Clear Sky Science · en
H19 enhances pancreatic cancer proliferation and invasion by reducing miR-29c-5p’s inhibitory effects on ATF2/ECM1
Why This Hidden Cell Message Matters
Pancreatic cancer is one of the deadliest cancers, in part because it is often found late and resists current treatments. This study looks beyond genes that make proteins and instead focuses on “silent” genetic messages—long and small RNA molecules—that help pancreatic tumors grow, spread, and evade therapy. By uncovering how one long RNA called H19 and a tiny partner called miR-29c-5p work together to reshape the tumor’s surroundings, the authors point to a new molecular wiring diagram that could guide earlier diagnosis and more precise treatments.
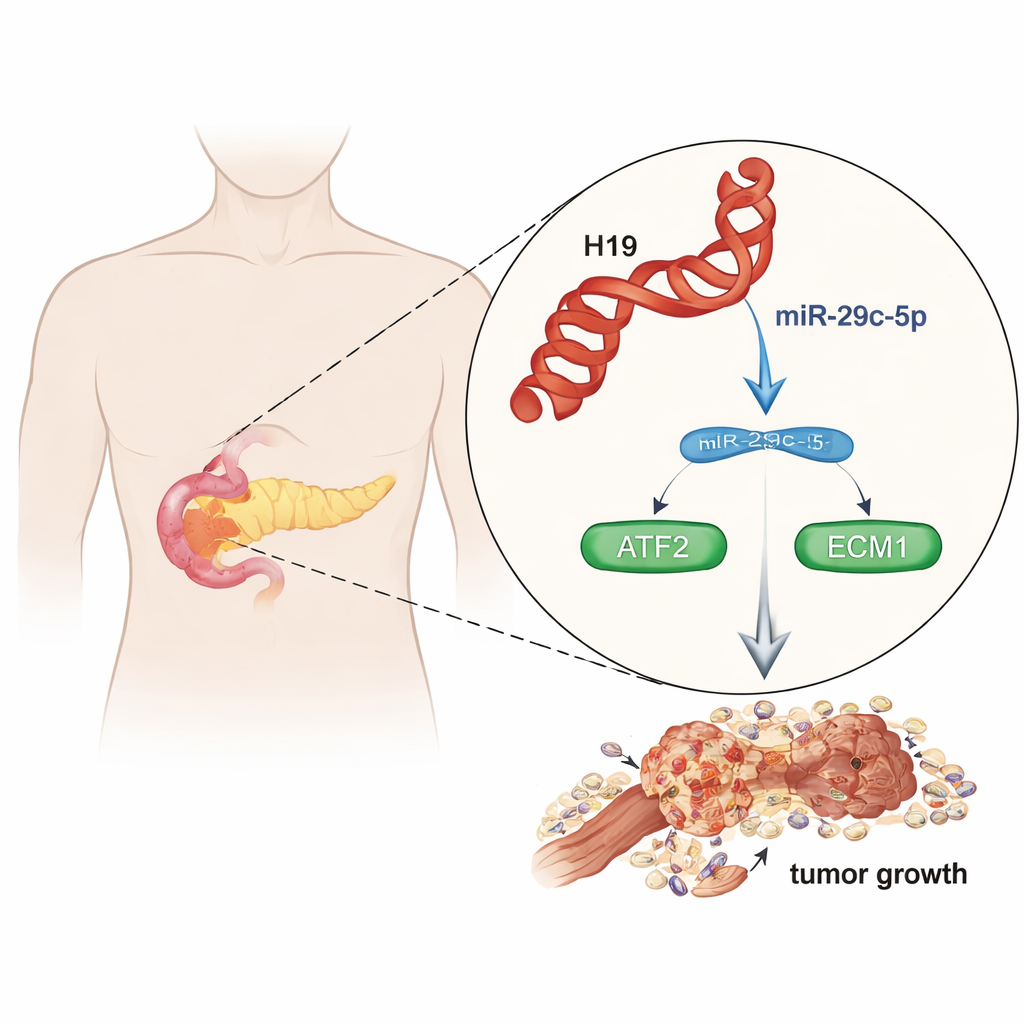
A Dangerous Cancer with Few Good Options
Pancreatic ductal adenocarcinoma (PDAC) has a five-year survival rate below 10 percent, mainly because it spreads early and responds poorly to chemotherapy. Researchers increasingly realize that non-coding RNAs—genetic messages that do not make proteins—are powerful switches that control cancer behavior. In this work, the team analyzed large public datasets and patient samples and found that one such long non-coding RNA, called H19, is consistently higher in pancreatic tumors than in normal pancreas tissue. Patients whose tumors had more H19 died sooner, suggesting that H19 is not just a bystander but a driver of aggressive disease.
How H19 Tips the Balance Inside Tumor Cells
Inside cells, H19 mainly sits in the watery cytoplasm, where it can interact with other RNAs rather than directly altering DNA. The authors discovered that H19 acts like a molecular sponge: it grabs and holds onto a small RNA called miR-29c-5p, which would otherwise help keep tumor growth in check. When H19 levels are high, less free miR-29c-5p is available to perform its watchdog role. Laboratory experiments showed that boosting H19 made pancreatic cancer cells multiply faster and move more easily through artificial membranes, while silencing H19 slowed their growth and invasion. Conversely, restoring miR-29c-5p reversed many of these harmful effects, pointing to a tug-of-war between the two molecules.
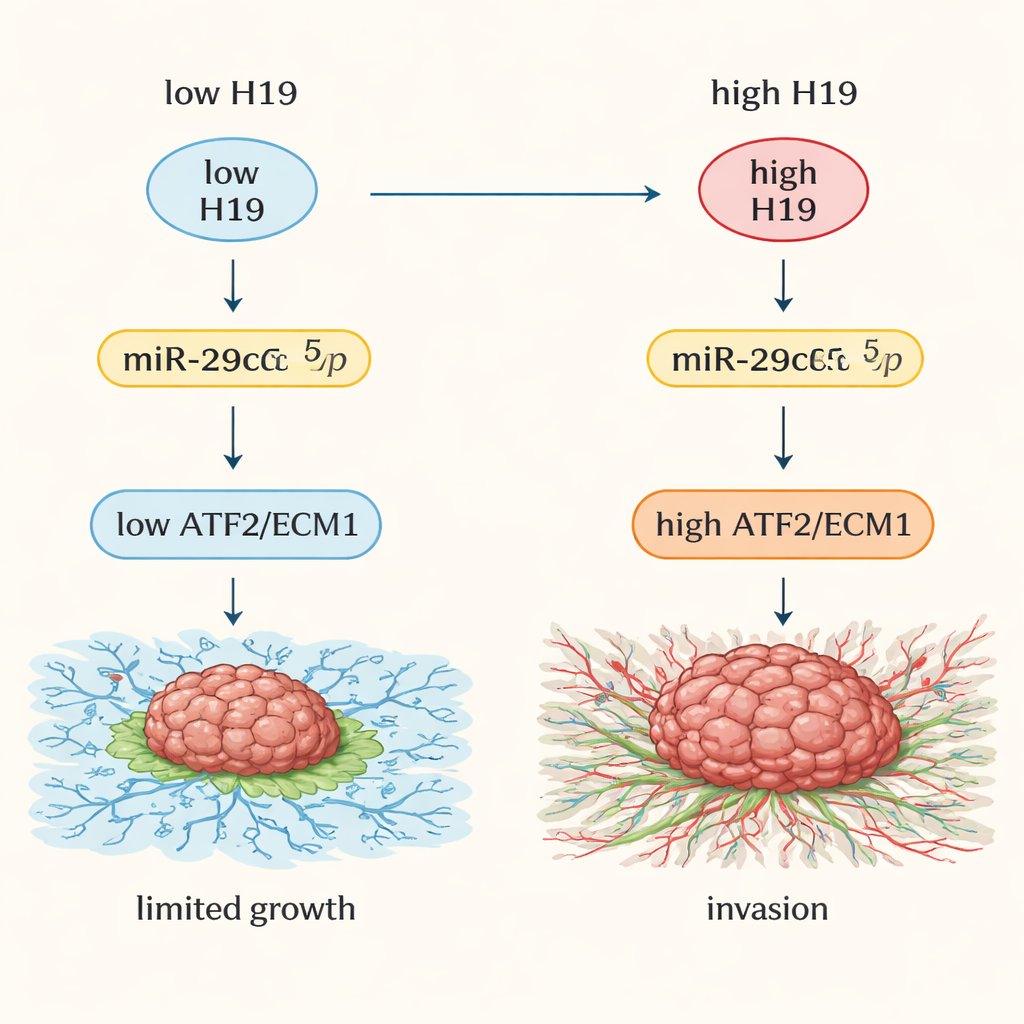
Downstream Targets: Releasing the Brakes on Tumor Growth
The small RNA miR-29c-5p normally helps restrain two important proteins: ATF2, a gene regulator inside the nucleus, and ECM1, a protein that shapes the material surrounding cells. When miR-29c-5p is soaked up by H19, ATF2 and ECM1 levels rise. In tumor samples and public cancer databases, both ATF2 and ECM1 were much higher in pancreatic cancers than in normal tissue. High levels of these proteins correlated with more advanced disease and poorer survival. In cell and mouse experiments, lowering H19 or increasing miR-29c-5p reduced ATF2 and ECM1, shrank tumors, and weakened their invasive behavior. The evidence indicates a chain reaction: H19 binds miR-29c-5p, freeing ATF2, which then boosts ECM1, promoting a stiff, fibrotic environment that favors tumor spread.
From Mechanism to Potential Markers and Therapies
Beyond mapping this signaling chain, the researchers tested whether these molecules might help doctors diagnose or classify pancreatic cancer. Using patient data, they found that ECM1 and ATF2 levels distinguished cancer tissue from normal pancreas with high accuracy, outperforming the commonly used blood marker CA19-9 in some analyses. Patients whose tumors had lower ECM1 tended to live longer, suggesting it may serve as a warning signal for aggressive disease. Although the clinical sample set from this hospital was small, the consistency across independent databases and animal experiments strengthens the case that this axis is central to PDAC biology.
What This Means for Patients and Future Research
In everyday terms, this study shows that a long “silent” RNA, H19, helps pancreatic tumors grow and invade by neutralizing a protective small RNA, miR-29c-5p, and unleashing two growth-promoting proteins, ATF2 and ECM1. This four-part chain—H19/miR-29c-5p/ATF2/ECM1—helps the tumor remodel its neighborhood into a supportive nest and is closely tied to worse outcomes. While more and larger clinical studies are needed, the work suggests that measuring components of this pathway could help identify high-risk patients, and that drugs designed to block H19 or ECM1, or restore miR-29c-5p activity, may one day offer new options against this notoriously hard-to-treat cancer.
Citation: Rui, M., Xiuping, L., Yu, C. et al. H19 enhances pancreatic cancer proliferation and invasion by reducing miR-29c-5p’s inhibitory effects on ATF2/ECM1. Sci Rep 16, 7623 (2026). https://doi.org/10.1038/s41598-026-37632-6
Keywords: pancreatic cancer, non-coding RNA, H19, tumor microenvironment, biomarkers