Clear Sky Science · en
Model for the description of interactions between ellagitannins and Fe(II) ions
Why Plant Compounds in Animal Feed Matter
Antibiotic-resistant bacteria are a growing worry for both farmers and public health officials. Many of these hard-to-treat microbes arise in livestock, where antibiotics are still widely used in feed. This study looks at tannins—natural compounds from plants—as a promising alternative. In particular, it explains how a special group of tannins from chestnut wood can grab onto iron in ways that may help starve bacteria of a nutrient they desperately need to grow.
Natural Defenders Hidden in Trees
Tannins are bitter, plant-made molecules that give red wine its astringency and have been used for centuries in leather tanning and inks. They are abundant in many feeds and foods and are generally safe for animals and humans. Some types, called ellagitannins, are especially interesting because they have many small chemical “hands” that can latch onto metal ions such as iron. Chestnut wood extract, already used in animal feed, contains several ellagitannins, including two large ones called roburin A and roburin D. Earlier work showed that simpler relatives of these compounds can bind iron and may block bacteria from accessing this essential element.
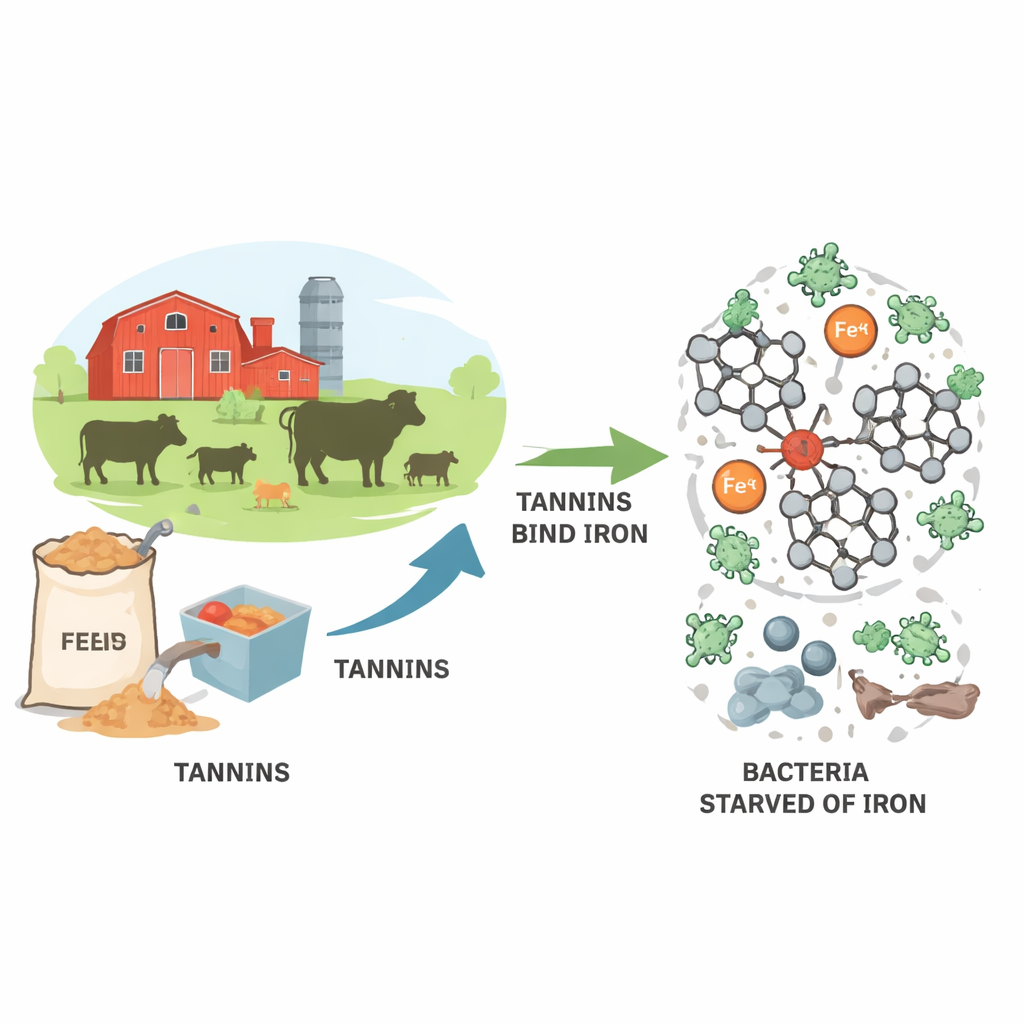
Starving Bacteria of Their Favorite Metal
Bacteria cannot thrive without iron. They use it to power respiration, build DNA, and run many enzyme reactions. In the animal gut or in growth media, they normally obtain iron using their own small iron-grabbing molecules. Ellagitannins interfere with this by forming tight complexes with iron ions, effectively “locking” the metal away from bacteria. The authors focused on iron in its Fe(II) form, which is the state quickly captured by tannins in water before it slowly oxidizes to Fe(III). By isolating roburin A and D from chestnut extract and studying them in carefully controlled solutions, the team could follow how well these large tannins remove iron from the liquid phase.
Probing How Tannins Grab Iron
To understand the details, the researchers first examined how roburins gain and lose protons (a process called acid–base equilibrium) as pH changes. Using ultraviolet–visible light spectroscopy, they tracked how the roburins’ light absorption shifted across different pH levels. These shifts revealed that roburin A and D behave much like their smaller cousins vescalagin and castalagin, but with roughly twice as many sites that can lose a proton and then participate in iron binding. Next, they mixed tannins and iron in varying ratios and again used light absorption to create so-called Job plots, which show what mixing ratio produces the most iron–tannin complex. From these data, they concluded that each roburin molecule can bind six Fe(II) ions—double the capacity of the smaller ellagitannins.
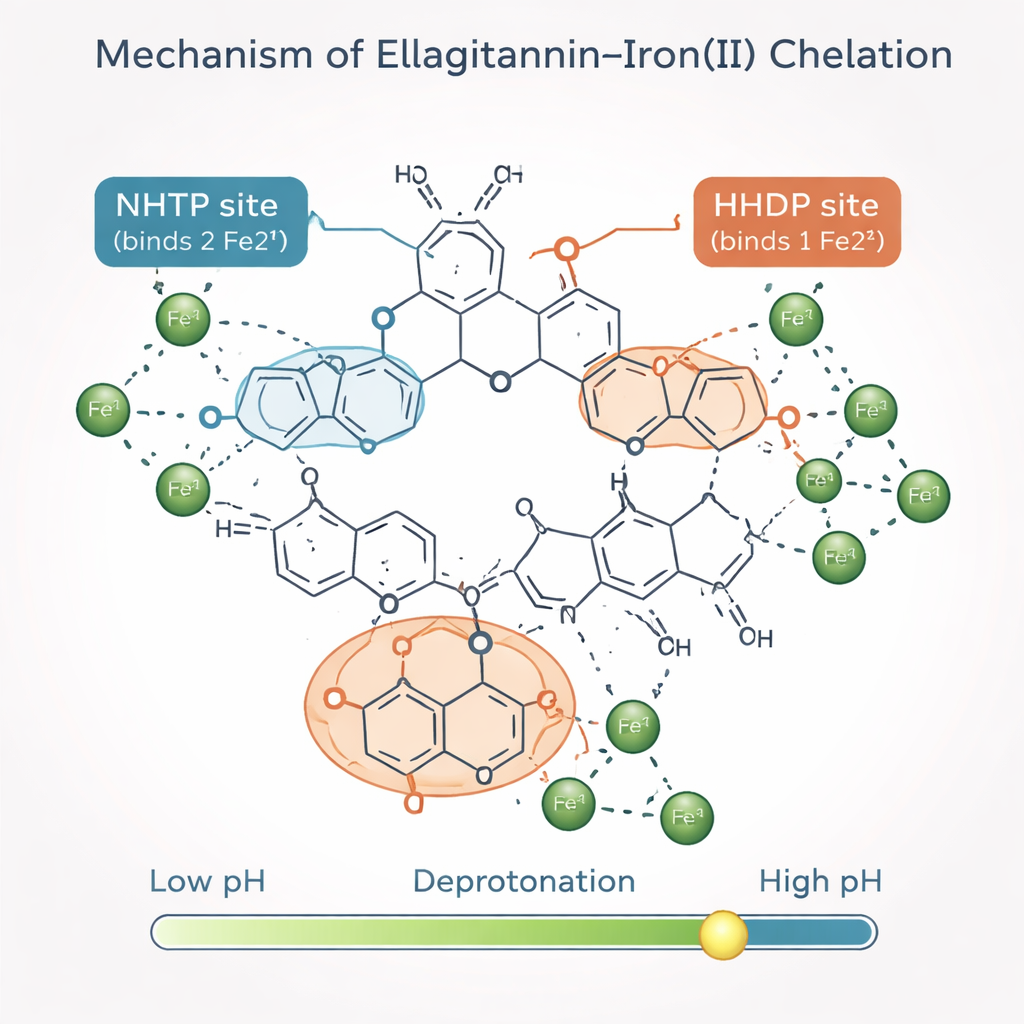
A Predictive Map of Iron-Binding Hotspots
Beyond counting how many iron ions can be captured, the authors wanted to know which structural “modules” of the ellagitannins do the work. They built a mathematical model that treats each ellagitannin molecule as a collection of repeating building blocks. Two key modules, called NHTP and HHDP groups, each offer specific binding spots for iron once they have lost their most acidic protons. By combining their new measurements with earlier nuclear magnetic resonance and spectroscopy data, the team showed that each NHTP group typically binds two iron ions, while each HHDP group binds one. With just a few adjustable parameters, their model accurately reproduced the experimental Job plots not only for the simpler ellagitannins but also for the larger roburins that were not used to train the model.
Implications for Greener Farming
In plain terms, this work turns the complex chemistry of tannins and iron into a usable rulebook. It shows that by counting NHTP and HHDP units in an ellagitannin, scientists can predict how many iron ions a given molecule will capture across a range of mildly acidic conditions. Because roburin-rich chestnut extracts seize more iron than smaller tannins or simple plant acids, they are strong candidates for limiting the iron supply available to harmful gut bacteria in farm animals. While more research is needed—especially for other tannin structures and for iron in different forms—this model helps guide the design and selection of plant-based feed additives that could reduce reliance on conventional antibiotics and support more sustainable, “greener” livestock production.
Citation: Frešer, F., Hostnik, G. & Bren, U. Model for the description of interactions between ellagitannins and Fe(II) ions. Sci Rep 16, 6631 (2026). https://doi.org/10.1038/s41598-026-37616-6
Keywords: ellagitannins, iron chelation, tannins in animal feed, antibiotic alternatives, chestnut wood extract