Clear Sky Science · en
Optimized gradient of lyophilized platelet-rich plasma in biomimetic 3D-printed triphasic scaffold based on alginate and gelatin for osteochondral tissue engineering
Why rebuilding joint surfaces matters
When the smooth surfaces in our knees or other joints are damaged, everyday movements like walking, climbing stairs, or even standing up can become painful. These surfaces are made of a complex "osteochondral" unit: slippery cartilage on top, a thin hardened zone in the middle, and supporting bone underneath. Today’s surgical repairs often fail because they do not fully rebuild this three-part structure. This paper explores a new 3D-printed, multi-layered scaffold designed to better mimic natural joint tissue and to guide the body’s own stem cells to regrow healthy cartilage.
Building a layered support for damaged joints
To imitate real joint anatomy, the researchers designed a “triphasic” scaffold with three stacked layers: an upper cartilage-like layer, a middle calcified layer, and a lower bone-like layer. They used a blend of two natural polymers, alginate and gelatin, as the basic ink for 3D printing. To strengthen the bone side of the scaffold, they added tiny sheets of graphene oxide, a carbon-based nanomaterial known for its mechanical strength and good interaction with cells. To make the cartilage side more biologically active, they mixed in freeze-dried platelet-rich plasma (PRP)—a concentrated source of the growth factors that our own blood platelets release during healing. By gradually changing the amount of PRP from the bottom to the top, they created a gentle biological gradient that better reflects how signals vary across real joint tissue. 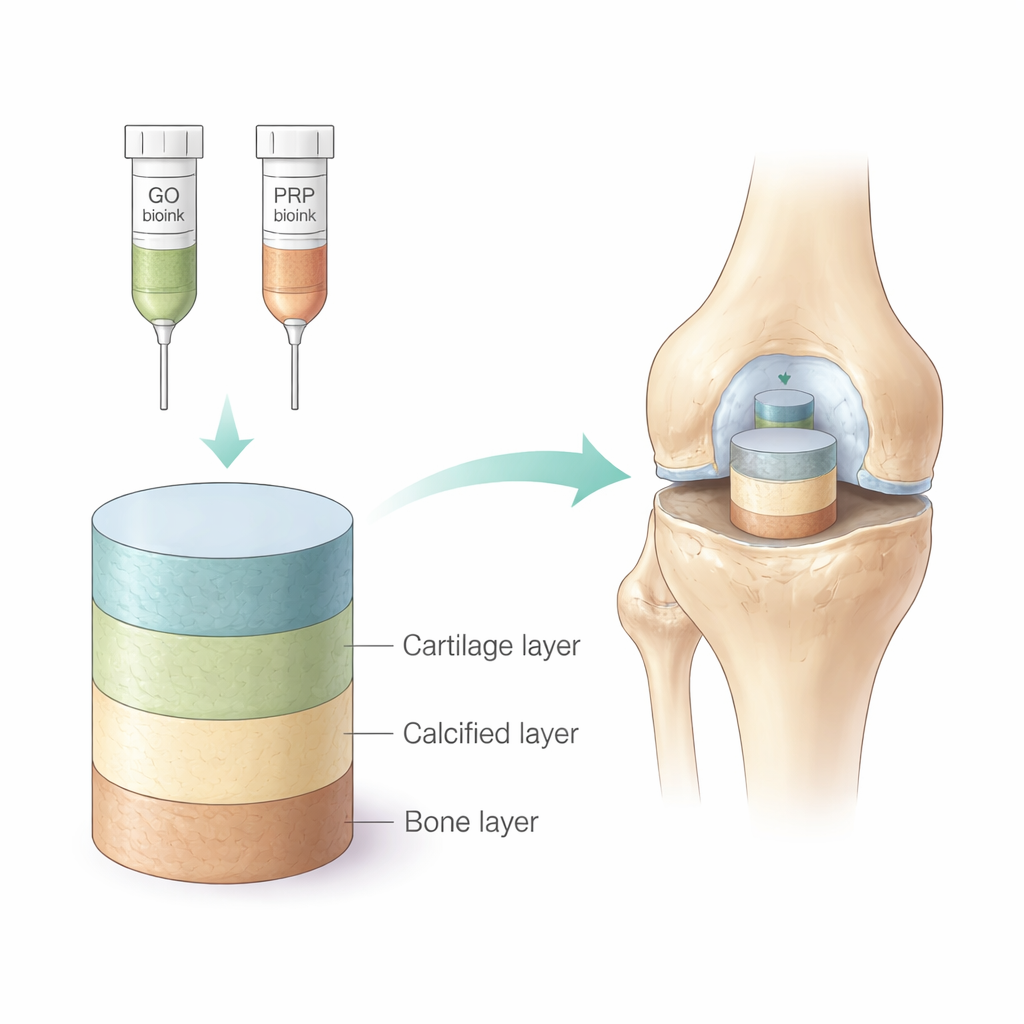
Finding the right balance of strength and stability
A major challenge in printing living tissue supports is making them strong enough to handle forces inside the body while still remaining soft and wet like natural cartilage. The team first optimized the graphene oxide content in the bone layer. They showed that adding a small amount (1% by weight) significantly increased compressive strength and helped the printed strands keep their shape, while still allowing the material to swell with water and remain permeable to nutrients. Higher graphene levels did not help further and even began to reduce stability. They then tested different PRP amounts in the cartilage region. Scaffolds with 1% or 2% PRP were easier to print cleanly and degraded at a controlled rate over about a month—long enough to support new tissue formation but not so long that the material would linger after its job was done.
How stem cells respond inside the scaffold
To see whether this layered design truly encourages cartilage repair, the researchers seeded rat bone marrow stem cells into the printed scaffolds and cultured them in conditions that favor cartilage formation. They measured how well the cells survived, spread, and switched on cartilage-related genes. All scaffolds supported healthy cells, but those containing PRP clearly boosted cell growth compared with PRP-free versions. In particular, the scaffold with 2% PRP showed the strongest signals of cartilage-building activity: stem cells produced more of the hallmark cartilage genes SOX9 and collagen type II, while reducing collagen type I, which is associated with less desirable, fibrous repair tissue. Staining tests also revealed greater amounts of glycosaminoglycans, the sugar-rich molecules that give cartilage its shock-absorbing properties, in the 2% PRP group.
Slow, steady release of the body’s own healing signals
The freeze-dried PRP powder in the scaffold acted like a built-in reservoir of healing cues. Tests of the material alone and within the 3D structure showed that key growth factors such as PDGF and TGF-β were released in a controlled way over about three weeks. This slow release is important: rather than a brief burst that quickly fades, a long-lasting signal can keep stem cells on a cartilage-forming path and help them build up a richer, more durable matrix. At the same time, the printed architecture—an open, interconnected grid of pores—allowed nutrients to diffuse through the scaffold and gave cells room to attach, spread, and interact with each other, much as they would in native tissue. 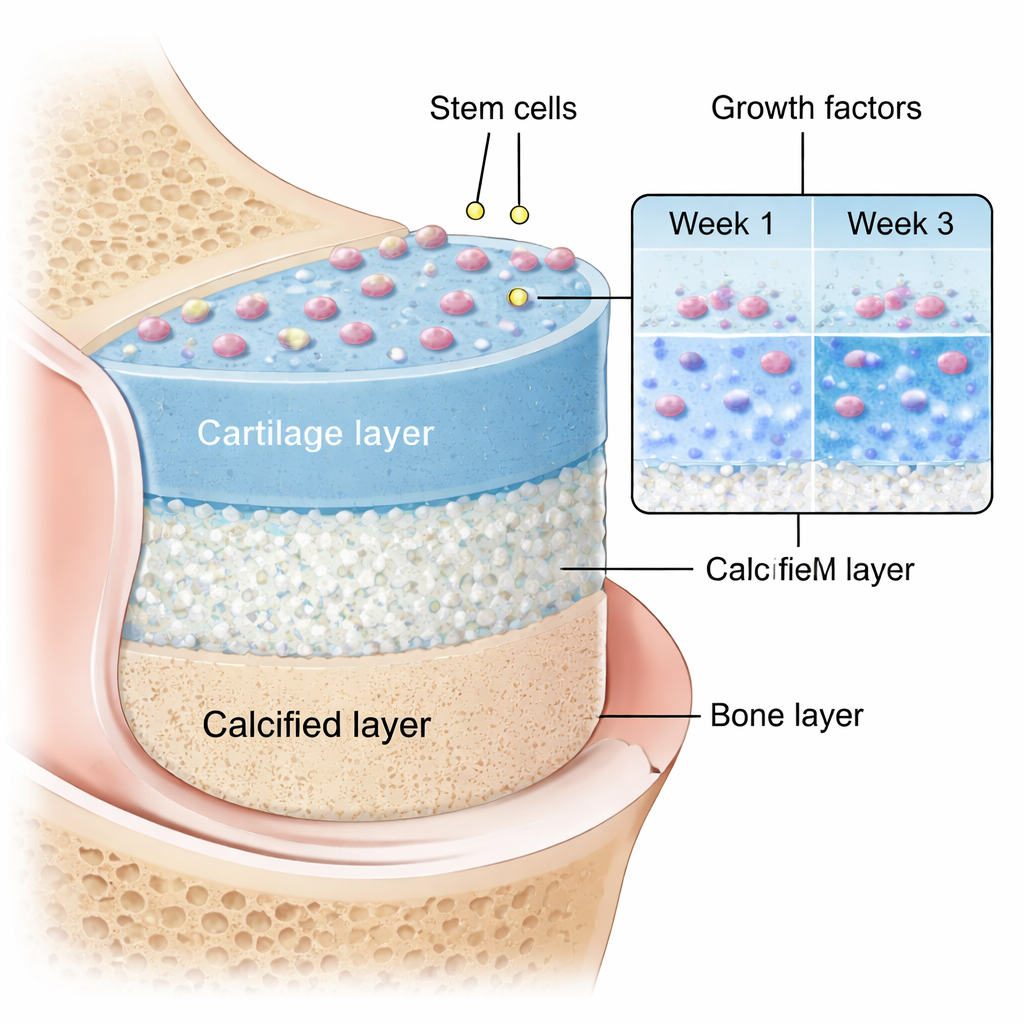
What this could mean for future joint repair
In simple terms, this study shows that a carefully tuned, 3D-printed, three-layer scaffold can both mechanically support a damaged joint surface and biologically coach stem cells to rebuild cartilage rather than scar-like tissue. A blend containing alginate, gelatin, 1% graphene oxide in the bone side, and 2% PRP in the cartilage side emerged as the most promising recipe. While these results come from lab studies with rat cells and not yet from tests in living animals or humans, they suggest a path toward more natural, long-lasting repairs for worn or injured joints by combining smart materials, blood-derived growth factors, and precise 3D printing.
Citation: Ghobadi, F., Mohammadi, M., Kalantarzadeh, R. et al. Optimized gradient of lyophilized platelet-rich plasma in biomimetic 3D-printed triphasic scaffold based on alginate and gelatin for osteochondral tissue engineering. Sci Rep 16, 6332 (2026). https://doi.org/10.1038/s41598-026-37615-7
Keywords: 3D-printed scaffold, osteochondral repair, platelet-rich plasma, graphene oxide, cartilage regeneration