Clear Sky Science · en
LIMK2 inactivation suppresses mechanical stimulation-induced dermal fibroblast differentiation and resistance to apoptosis
Why some scars grow out of control
Most cuts and surgical wounds leave behind a thin, flat line. But in some people, the healing process goes into overdrive, building thick, raised scars called hypertrophic scars or keloids that can itch, hurt, and limit movement. This study explores why ordinary skin cells sometimes become overzealous scar-makers, and points to a single molecular switch, called LIMK2, that could be turned down to keep scars from growing out of control.
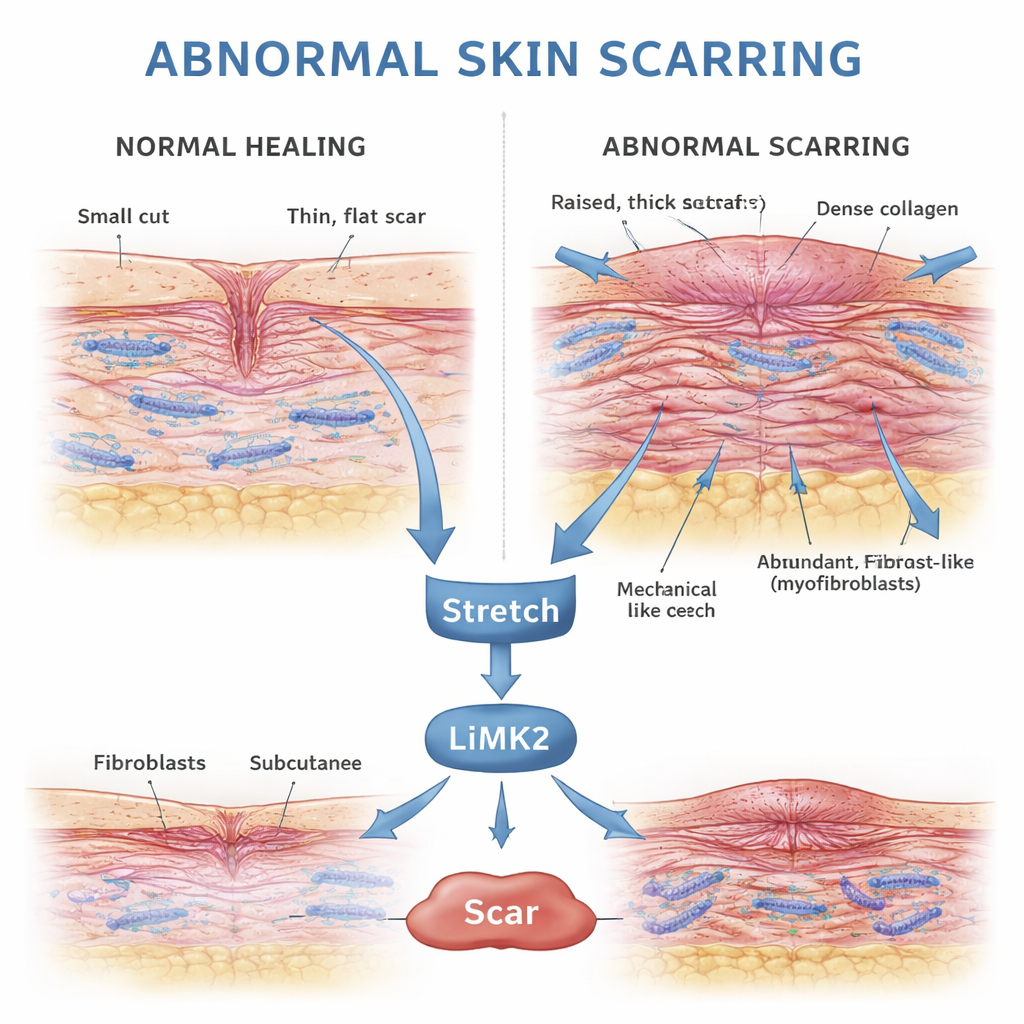
How skin normally heals after injury
When skin is injured, the body launches a carefully timed repair program. Fibroblasts—construction-worker cells in the deeper layer of skin—move into the wound, multiply, and lay down collagen, the protein scaffold of new tissue. Many of these fibroblasts temporarily shift into a stronger, more contractile form called myofibroblasts, which help pull the wound edges together. Once the surface has closed and the new tissue has stabilized, these myofibroblasts are supposed to die off through a controlled process known as apoptosis, leaving behind a modest, flexible scar.
When everyday forces make scars worse
Abnormal scars often appear where the skin is constantly being stretched—over the chest, shoulders, or lower belly. That observation has led researchers to suspect that mechanical forces, such as pulling and tension across a healing wound, may drive fibroblasts to stay active for too long. Earlier work showed that stretching these cells in the lab boosts a protein called α-SMA, a hallmark of myofibroblasts, and turns on genes that help cells resist apoptosis. The new study focuses on LIMK2, a key part of a mechanical signaling chain inside cells that links physical stretch to changes in the internal scaffolding made of actin filaments.
A key switch for scar-forming cells
To test LIMK2’s role, the researchers used human dermal fibroblasts from normal skin and from keloid tissue. They introduced a disabled version of LIMK2 into some cells, leaving others unaltered or giving them an always-on version as controls. When cells were mechanically stretched, normal fibroblasts responded as expected: a rapid rise in phosphorylated cofilin and stronger actin fibers, increased α-SMA, more collagen production, and higher levels of Bcl-2, a protein that protects against cell death, along with lower levels of the pro-death protein BAX. In striking contrast, fibroblasts with inactive LIMK2 barely changed under stretch. They did not boost α-SMA, did not increase protective Bcl-2 or reduce BAX, and showed less collagen. These cells also contracted collagen gels less strongly and crawled more slowly in a wound-like assay, suggesting weaker pulling power and reduced ability to invade surrounding tissue.
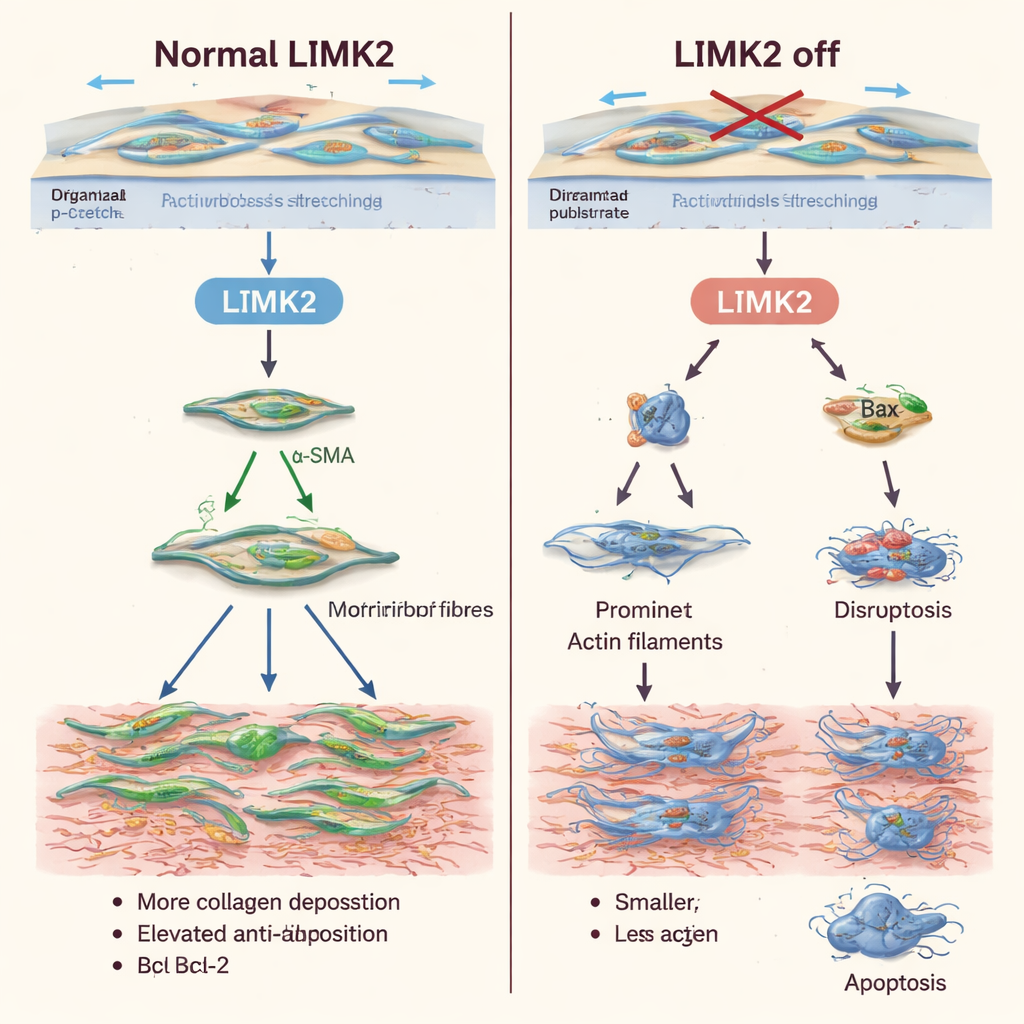
Making stubborn scar cells easier to remove
Beyond changing how fibroblasts behave under stretch, shutting down LIMK2 made them more likely to die and less likely to multiply. Cells with inactive LIMK2 showed higher baseline levels of apoptosis, and mechanical stretching—normally a survival signal—no longer protected them. Flow cytometry and fluorescent staining confirmed that more cells were in early or late stages of programmed cell death. At the same time, cell counts and DNA-labeling tests revealed that these cells divided far less than normal. Similar patterns appeared when the team used latrunculin A, a drug that disrupts actin filaments, supporting the idea that LIMK2 acts mainly by controlling the cell’s internal framework.
What this could mean for future scar treatments
The findings suggest that LIMK2 sits at a crossroads where mechanical forces, cell survival, and scar-building activity intersect. When LIMK2 is active, stretching pushes fibroblasts toward a tough, persistent myofibroblast state: they contract strongly, resist death, and churn out extra collagen, all of which favor thick, raised scars. Turning LIMK2 off reverses much of this—fibroblasts become less contractile, less mobile, less prolific, and more willing to undergo apoptosis. For patients, this points to a future in which creams, injections, or dressings that gently block LIMK2 or its downstream actin changes could help high-risk wounds heal with flatter, more comfortable scars.
Citation: Ishii, M., Kuroda, K., Otani, N. et al. LIMK2 inactivation suppresses mechanical stimulation-induced dermal fibroblast differentiation and resistance to apoptosis. Sci Rep 16, 7453 (2026). https://doi.org/10.1038/s41598-026-37610-y
Keywords: keloid scars, wound healing, fibroblasts, mechanical tension, apoptosis