Clear Sky Science · en
Engineering novel ceramic metal borates containing carbon for efficient sequestration of Toluidine Blue O from wastewater
Why dyed water is everyone’s problem
From blue jeans to printed paper, modern life depends on dyes—and so does much of our wastewater. One of these dyes, Toluidine Blue O, is a vivid blue stain used in labs and industry. It can irritate skin and eyes, may harm internal organs after repeated exposure, and it blocks light in rivers and lakes, stressing aquatic life. This study explores new, low‑cost ceramic materials that can sponge this stubborn dye out of water with remarkable efficiency, pointing to a practical tool for cleaner streams and safer taps.
Designing a smart sponge for blue dye
The researchers set out to build tiny, solid “sponges” that grab Toluidine Blue O from water and hold on tight. They used a relatively simple chemical recipe called the Pechini sol–gel method to make two related materials, heated to 500 °C and 700 °C and nicknamed AFB500 and AFB700. Each grain is a nanohybrid—a mix of several borate and iron‑oxide minerals knit together with a small amount of carbon. This blend was chosen so different parts of the surface could attract the dye in different ways, increasing both the speed and the total amount of dye removed.
What these tiny grains look like
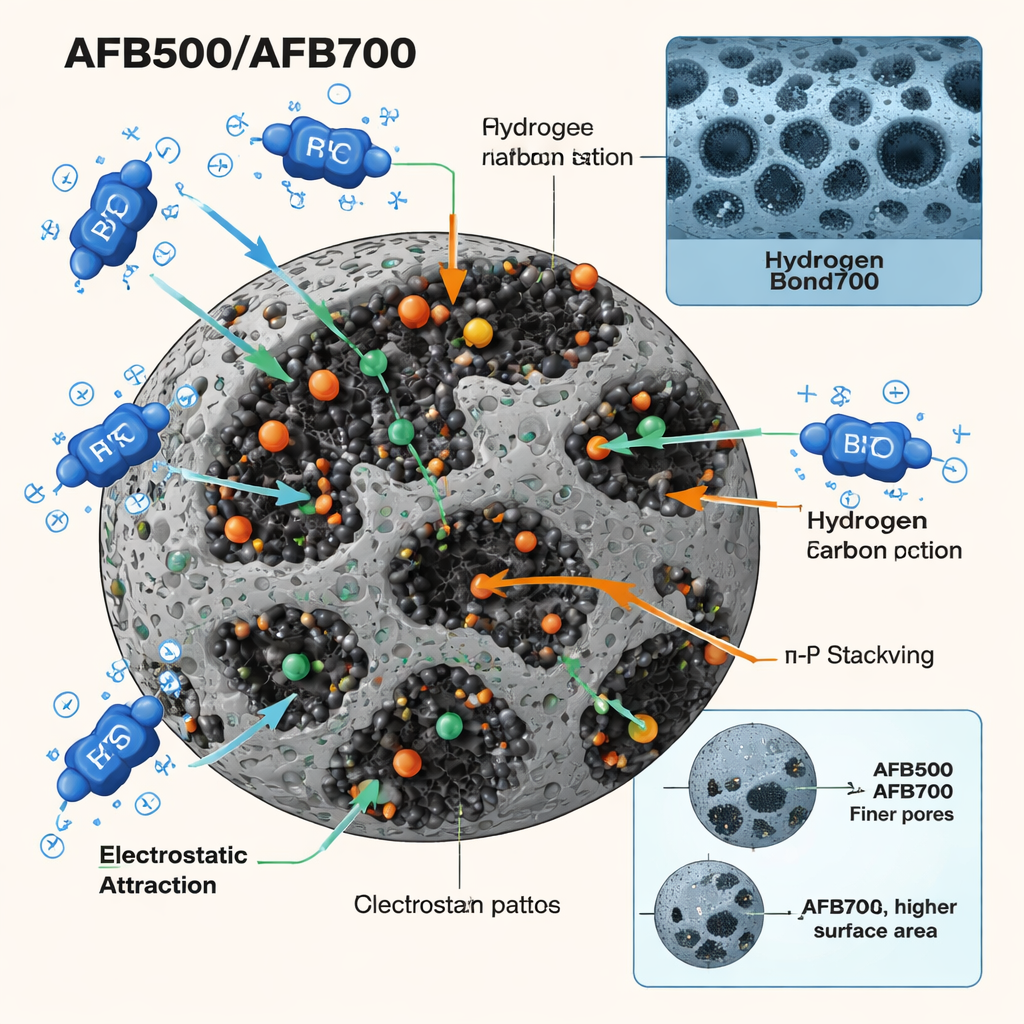
To understand how these nanohybrids might work, the team used X‑ray diffraction and electron microscopes to view their internal structure and shape. AFB500, the material fired at the lower temperature, formed thin plates and sheets made of many fine crystals, giving it a relatively high surface area riddled with small pores. AFB700, fired hotter, reorganized into more compact, rounded grains with larger but fewer pores and more highly ordered crystals. Both contained the intended mix of boron, iron, aluminum, oxygen, and carbon, but AFB500 held more carbon, while AFB700 was richer in inorganic mineral content—differences that turned out to matter for performance.
How the sponges catch and hold the dye
Tests in dyed water revealed that pH, or acidity, plays a big role. At low pH, the grain surfaces are positively charged, just like the Toluidine Blue O molecules, so they repel each other and very little dye is captured. At pH 10, the surfaces become negatively charged, setting up a strong electrostatic pull on the positively charged dye. On top of that, oxygen‑rich groups on the borate and iron‑oxide surfaces can form hydrogen bonds and weak complexes with the dye, while the carbon regions interact with the dye’s flat aromatic rings through stacking interactions. Together, these mechanisms allow AFB500 to remove about 92% of the dye under optimal lab conditions, and AFB700 about 64%, with the dye loading onto a single, well‑organized layer on the surface.
Putting performance and durability to the test
The team pushed the materials under a range of conditions that mimic real‑world use. They varied contact time, temperature, the amount of adsorbent, salt levels, and dye concentration. AFB500 consistently outperformed AFB700, thanks to its finer pores and larger surface area, reaching a maximum dye‑holding capacity of about 424 milligrams per gram of material—higher than many previous adsorbents such as zeolites, gypsum, or several magnetic composites. The uptake followed a simple time law (pseudo‑first‑order kinetics) and decreased slightly at warmer temperatures, showing that adsorption is a spontaneous but gently heat‑releasing process. Common ions like sodium and chloride had modest effects, while other positively charged dyes competed strongly for the same sites, as expected in realistic mixtures.
From lab flask to real wastewater
Crucially, these nanohybrids do not appear to be single‑use. The researchers stripped off the captured dye by rinsing the grains with hydrochloric acid, which flips the surface charge and pushes the dye back into solution. At an acid strength of 2 molar, nearly all of the dye was released, and both AFB500 and AFB700 retained most of their capturing power over at least five reuse cycles, with little structural change and no detectable leaching of metals. When challenged with actual lab wastewater—containing a cocktail of salts and trace metals, plus added Toluidine Blue O—the materials still held large amounts of dye, with AFB500 again leading the way. This combination of high capacity, reusability, and simple, scalable synthesis using inexpensive ingredients makes these ceramic–carbon grains promising candidates for dye‑laden effluents.
What this means for cleaner water
In plain terms, the study shows that carefully engineered ceramic particles, laced with a bit of carbon, can act as powerful, reusable filters for a hazardous blue dye. By tuning the firing temperature, the authors could shift the balance between surface area and crystallinity, with the lower‑temperature AFB500 offering the best trade‑off for mopping up dye quickly and in large amounts. Because the starting chemicals are common and the process resembles standard ceramic production, these materials could, in principle, be made at scale and packed into treatment units for textile plants, laboratories, or other facilities that discharge colored wastewater. In helping remove stubborn dyes like Toluidine Blue O, they support broader efforts, such as the United Nations’ goal of clean water and sanitation, by turning a complex chemistry problem into a practical filtration step.
Citation: Basha, M.T., Alhamzani, A.G. & Abdelrahman, E.A. Engineering novel ceramic metal borates containing carbon for efficient sequestration of Toluidine Blue O from wastewater. Sci Rep 16, 4526 (2026). https://doi.org/10.1038/s41598-026-37604-w
Keywords: wastewater treatment, dye removal, nanomaterials, adsorption, Toluidine Blue O