Clear Sky Science · en
Clinical features and inflammatory signatures of patients with persistent gastrointestinal long COVID two years after severe SARS-CoV-2 infection
Why stomach troubles after COVID still matter
Many people think of COVID-19 as a lung infection, yet for a surprising number, the illness leaves a long-lasting mark on the digestive system. This study followed patients in Manaus, Brazil, who survived severe COVID-19 during the first wave of the pandemic and looked at them again two years later. The researchers wanted to know how common ongoing gut problems were and whether lingering changes in the immune system might help explain why some people still struggle with symptoms like heartburn, pain, and diarrhea long after the virus is gone.
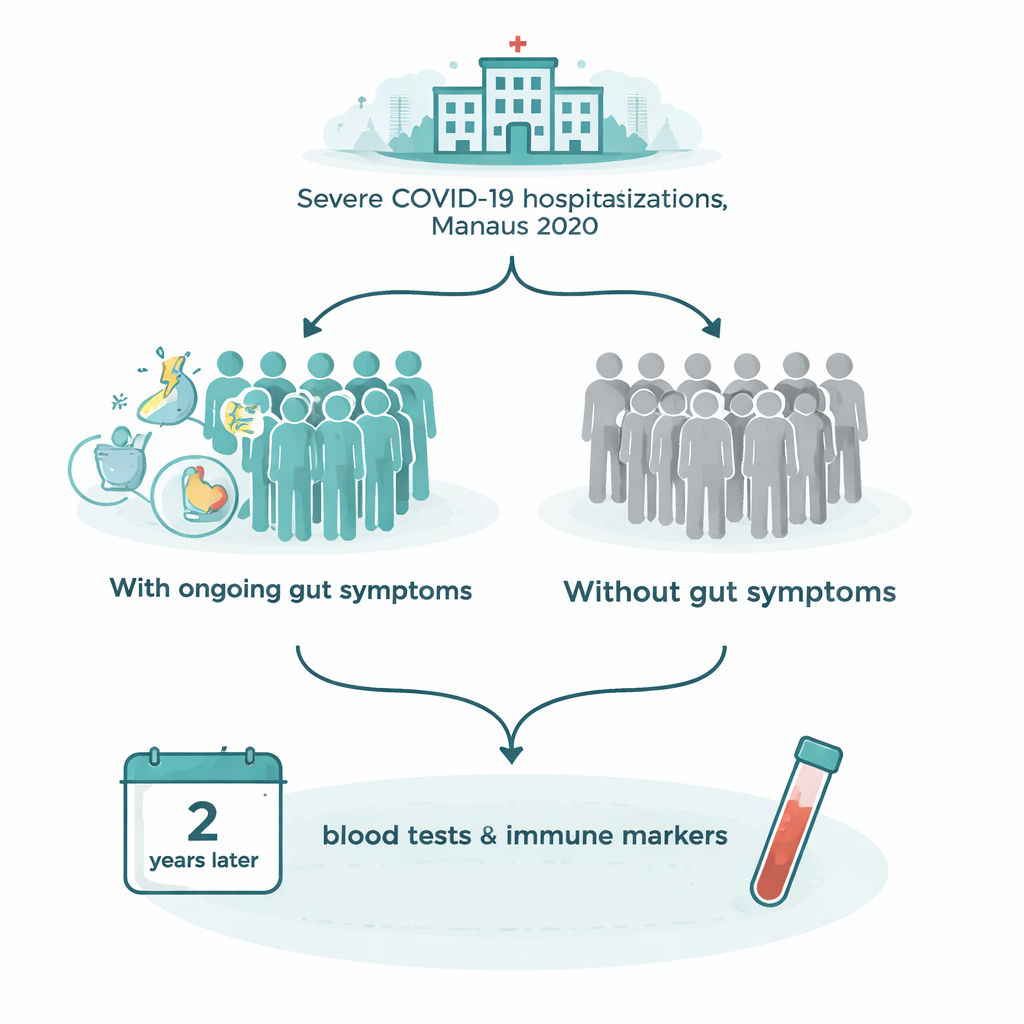
Who was followed and what happened to them
The team revisited 80 adults who had been hospitalized with severe COVID-19 in early 2020, before vaccines were available. Two years later, they interviewed each person about their health and checked blood tests that had been collected from the first days in the hospital through the long recovery period. About four in ten participants (30 people) said they still had at least one troublesome digestive symptom. The most frequent complaints were burning acid in the chest or throat (gastroesophageal reflux), stomach or belly pain, and ongoing bouts of diarrhea. Many of these people had more than one symptom, and women were affected more often than men.
Long COVID rarely stops at the gut
Those with persistent digestive issues were also more likely to report other long COVID problems, including pounding heartbeats, headaches, joint pain, hair loss, fatigue, and cough. This pattern suggests that lingering gut symptoms are often part of a broader, body-wide condition rather than an isolated stomach ailment. Importantly, the length of the original hospital stay and the need for intensive care were similar between those with and without lasting gut problems, hinting that factors beyond the obvious severity of the early illness help determine who develops long-term trouble.
Clues from the blood: an immune system out of balance
To look for deeper explanations, the researchers focused on small messenger proteins in the blood called cytokines, which help control inflammation. They tracked several of these signals, including IL-6, IL-1, IL-8, IL-10, IL-12, and TNF, from the first day in the hospital up to two years later. During the acute infection, people who later developed long-term gut symptoms actually had lower levels of IL-6 than others. But four months after infection, IL-6 levels in this group rose and stayed relatively high, suggesting a delayed and lingering inflammatory response. Other markers, like ferritin and creatinine, were also different at the two-year mark, and platelet counts were higher, all pointing toward an ongoing disturbance in the body’s equilibrium.
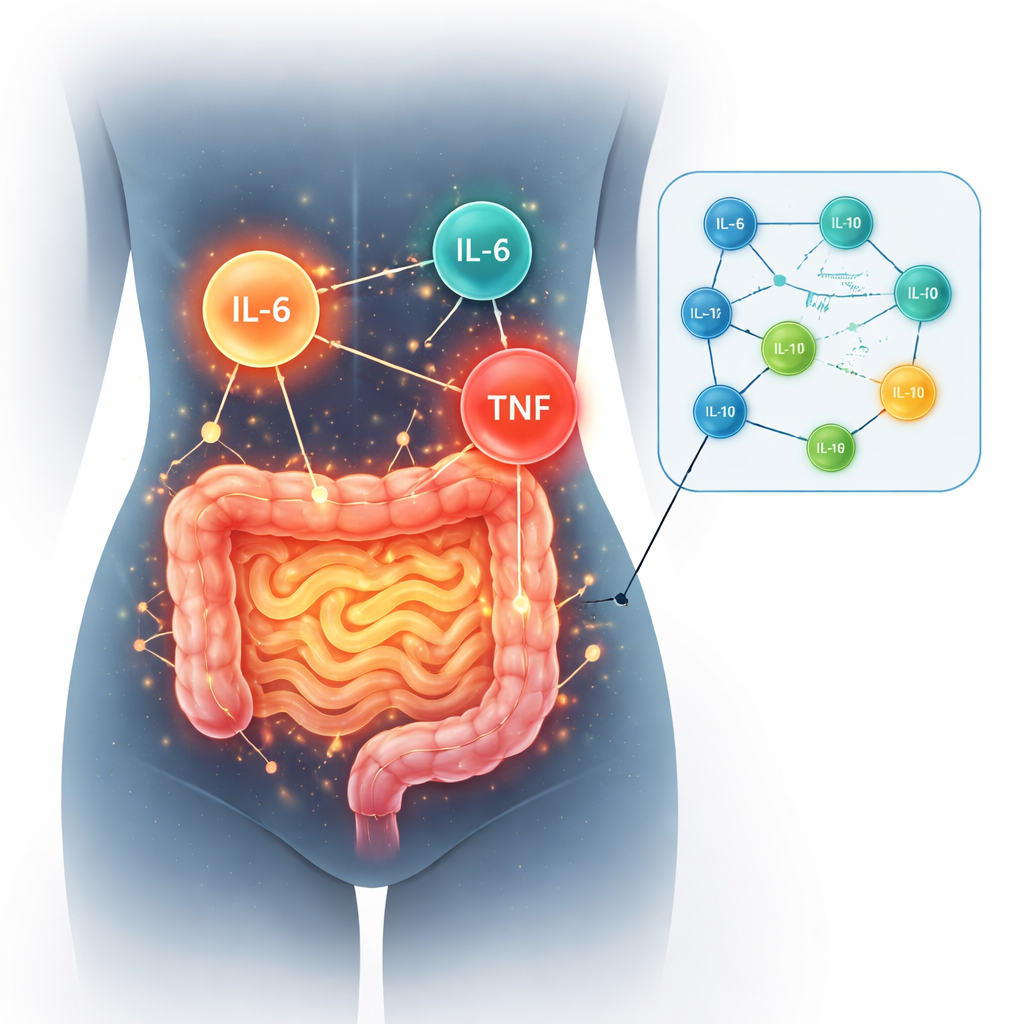
A tangled and weakened immune network
When the scientists mapped how these cytokines moved together over time, they saw striking differences between groups. In participants who recovered without gut problems, the immune signals formed a stable, tightly connected network, as if the body’s alarm system had settled back into a coordinated rhythm. In contrast, those with persistent digestive symptoms showed a progressive breakdown of these connections, especially by the two-year point. IL-6 stood out as one of the few molecules still strongly linked to others, hinting that it may be driving a kind of chronic low-grade inflammation. Early in the illness, another molecule called TNF appeared unusually high only in the group that later developed gut problems, raising the possibility that it could serve as an early warning sign for who is at risk.
What this means for patients and care
Put simply, this study shows that serious COVID-19 can leave a lasting footprint in the digestive system, with symptoms persisting for at least two years in a substantial minority of patients. These gut problems go hand in hand with a subtly disturbed immune system that never quite returns to normal balance, particularly around signals like IL-6 and TNF. For patients, this means that ongoing heartburn, pain, or diarrhea after COVID-19 are not “just in their head” but may reflect real, long-term immune changes. For doctors and health systems, the findings argue for routine screening and follow-up of digestive symptoms in COVID-19 survivors, and for more research into treatments that can gently calm this lingering inflammation and prevent lasting damage.
Citation: dos Santos Pinto, A., Mwangi, V.I., Neves, J.C.F. et al. Clinical features and inflammatory signatures of patients with persistent gastrointestinal long COVID two years after severe SARS-CoV-2 infection. Sci Rep 16, 6620 (2026). https://doi.org/10.1038/s41598-026-37595-8
Keywords: long COVID, gastrointestinal symptoms, inflammation, cytokines, SARS-CoV-2