Clear Sky Science · en
SMAD2 ubiquitination through PY motif regulates skeletal muscle mass and fibrotic degeneration
Why this muscle study matters
As we age, many of us notice our muscles getting weaker and smaller, and in chronic disease or injury muscle can gradually be replaced by stiff scar tissue. This study tackles a basic question behind those changes: how do muscle cells keep a powerful growth‑controlling signal, called TGF‑beta, from going too far and causing muscle loss and fibrosis (scarring)? By uncovering a tiny built‑in "off switch" in a key TGF‑beta protein, the authors reveal a new mechanism that helps preserve muscle mass and healthy tissue structure.
A built‑in brake on a powerful signal
TGF‑beta is a signaling molecule that tells cells when to slow down growth, change identity, or produce connective tissue. In skeletal muscle, too much TGF‑beta shrinks fibers and promotes fibrosis; too little can disrupt normal control of growth. Inside cells, TGF‑beta works mainly through a relay protein called SMAD2. After TGF‑beta binds its receptors at the cell surface, SMAD2 is activated and moves into the nucleus to alter gene activity. To keep this from becoming permanent, cells chemically tag SMAD2 with small molecules of ubiquitin, marking it for removal. A short sequence in SMAD2, known as the PY motif, is the docking site for enzymes that attach these ubiquitin tags. The researchers asked: if this docking site is removed in a living animal, does the SMAD2 brake fail, and what happens to muscles over time?
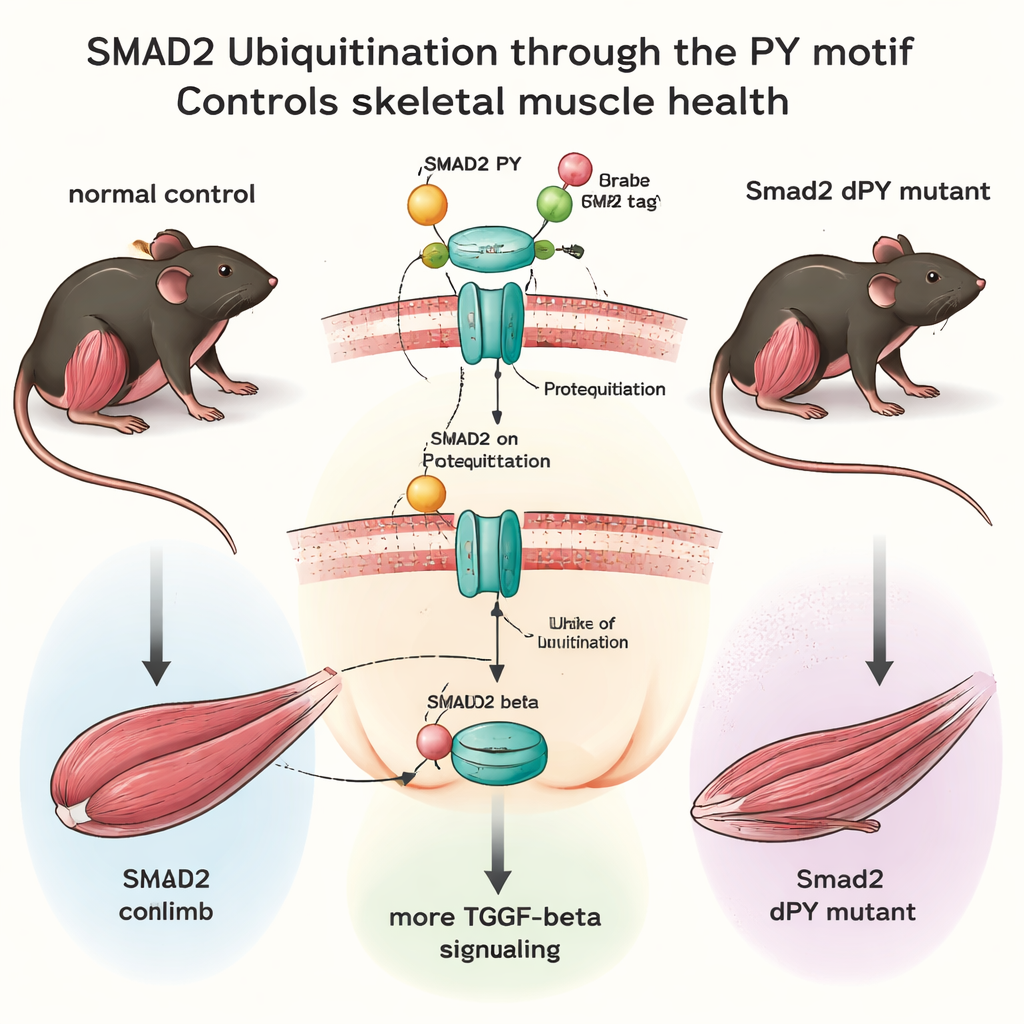
Engineering mice with a missing safety tag
Using modern genome‑editing tools, the team created mice in which just 15 DNA "letters" were deleted from the Smad2 gene, precisely removing the PY motif without disturbing the rest of the protein. These Smad2dPY mice were born normally, grew to adulthood, and were fertile, showing that this fine‑tuning system is not essential for basic development under standard lab conditions. However, when the scientists examined their muscles more closely, a different picture emerged. Young adult mice showed only subtle changes, but by 12 months—roughly middle age for a mouse—the major hind‑leg muscles weighed less and individual fibers were smaller compared with normal littermates. Inside these muscles, SMAD2 protein levels were higher, while its ubiquitination was reduced, confirming that the missing PY motif made SMAD2 more stable and less degradable.
Over‑sensitive muscle cells and stalled repair
To understand what this meant at the cellular level, the researchers isolated muscle precursor cells (myoblasts) from the mutant mice. When exposed to TGF‑beta in a dish, these myoblasts showed stronger activation of SMAD2 and greater induction of TGF‑beta‑responsive genes than cells from normal mice. At the same time, their ability to fuse into long, multi‑nucleated muscle fibers—a key step in muscle growth and regeneration—was impaired. Similar changes were seen in fibroblasts, the connective‑tissue cells that sit between muscle fibers: mutant fibroblasts responded more strongly to TGF‑beta and more readily expressed genes linked to scar‑forming myofibroblasts. Together, these findings suggest that without the PY motif, both muscle‑forming cells and support cells become hypersensitive to TGF‑beta, tipping the balance toward smaller fibers and more fibrous tissue.
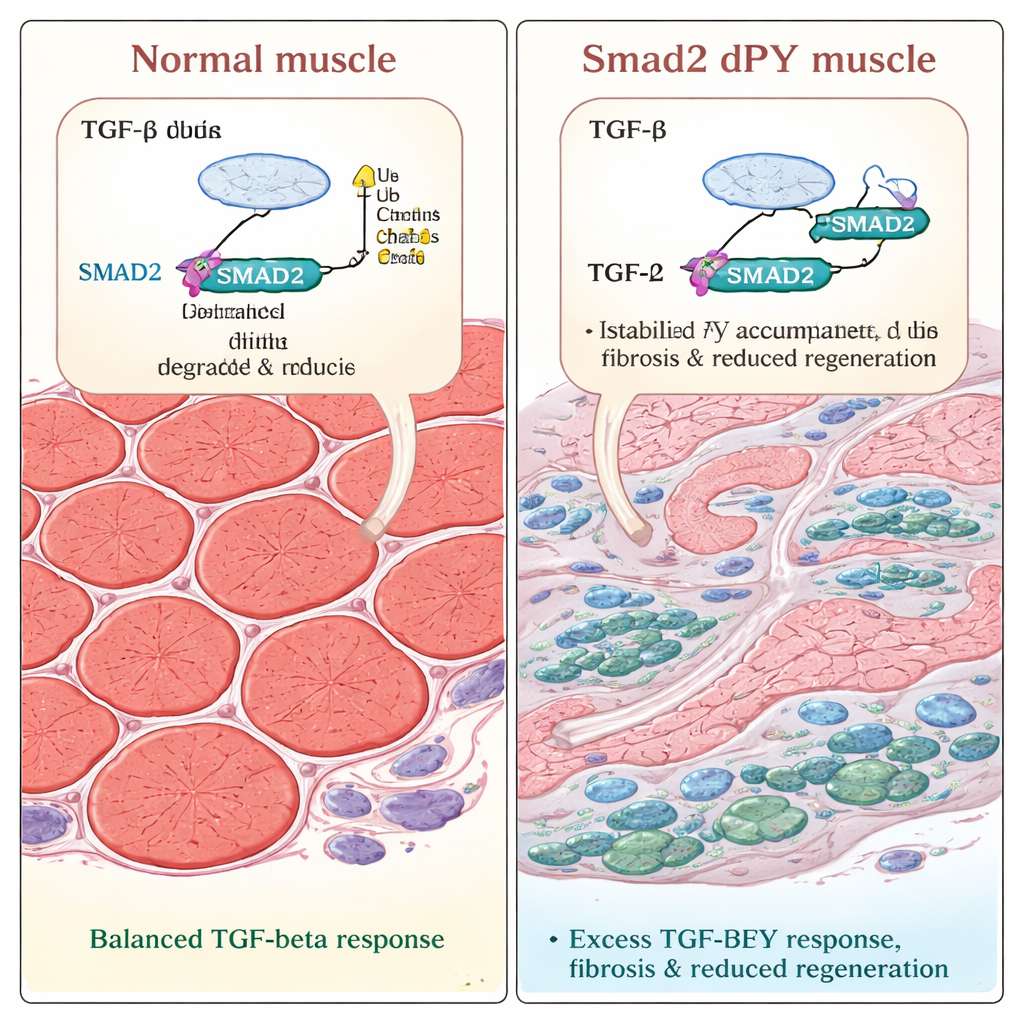
When injury strikes, scarring wins
The team then tested how these altered muscles cope with damage. They injected a toxin into leg muscles to trigger injury and repair, a well‑established model of regeneration. In both normal and mutant mice, damaged fibers initially broke down and then began to regrow. But three weeks after injury, regenerated fibers in the Smad2dPY mice were noticeably thinner. Microscopy revealed larger areas filled with collagen, the main component of scar tissue, and increased numbers of alpha‑smooth‑muscle‑actin‑positive myofibroblasts, cells known to drive fibrosis. These results show that when SMAD2 cannot be properly tagged and turned over, the repair process is skewed: instead of fully rebuilding strong muscle, the tissue heals with excess scarring and reduced contractile mass.
What this means for muscle health
For non‑specialists, the main message is that a very small molecular feature—the PY motif on SMAD2—acts as a quiet but important safeguard in skeletal muscle. It lets cells dial back TGF‑beta signaling by promoting SMAD2 removal, helping maintain normal muscle size and limiting fibrosis, especially as TGF‑beta levels naturally rise with age or chronic injury. When this safeguard is disabled in mice, muscles gradually become smaller and more scar‑prone, and they fail to recover fully after damage. While this work is still in animals, it highlights SMAD2 ubiquitination as a potential target pathway for future strategies aimed at preserving muscle mass and preventing fibrotic degeneration in aging or disease.
Citation: Yamasaki, Y., Sakamoto, K., Yashiro, S. et al. SMAD2 ubiquitination through PY motif regulates skeletal muscle mass and fibrotic degeneration. Sci Rep 16, 6666 (2026). https://doi.org/10.1038/s41598-026-37582-z
Keywords: skeletal muscle, TGF-beta signaling, SMAD2, ubiquitination, fibrosis