Clear Sky Science · en
Changes in EGFR activity following CRISPR/Cas9-editing of the EGF binding domain
Why Rewiring a Cancer Signal Matters
Cervical cancer often depends on a powerful growth switch called the epidermal growth factor receptor (EGFR). Many tumors have huge numbers of these receptors, yet drugs aimed at shutting them down have helped only a fraction of patients. This study asked a fundamental question with major treatment implications: what happens if you remove EGFR’s ability to grab its favorite growth signal, epidermal growth factor (EGF), without removing the receptor itself?
Turning Off a Molecular “On Switch”
The researchers focused on a cervical cancer cell line that carries large amounts of otherwise normal EGFR. Instead of destroying the gene, they used CRISPR/Cas9 genome editing as a molecular scalpel to change just a few amino acids in EGFR’s EGF‑binding pocket. Two particular positions, called L14 and Y45, help EGF dock to the receptor. By substituting these building blocks with different ones, the team aimed to keep EGFR present but blind to EGF, letting them isolate the role of this single interaction in cancer cell behavior.
Building Designer Cancer Cell Lines
In a first step, the team tested these mutations in cells using plasmids that transiently produce either normal or altered EGFR. Fluorescently tagged EGF revealed that all mutant forms bound far less ligand than the normal receptor, especially when both L14 and Y45 were changed together. The researchers then introduced these changes directly into the cancer cell genome with CRISPR/Cas9, generating several new cell clones. Some carried a single mutation, others carried the double mutation on one EGFR copy and inactivating changes on the other. Computer modeling of the resulting protein shapes showed that these alterations distorted the binding site enough to weaken or abolish EGF docking, consistent with the binding tests.
When the Receptor Moves but the Cells Survive
Detailed imaging revealed that the edited receptors behaved very differently inside the cell. In unedited cancer cells, EGFR sits on the cell surface, captures EGF, and then moves inward while becoming chemically activated through phosphorylation. In clones bearing the double mutation, EGF binding essentially vanished and EGFR was no longer seen on the membrane; instead, the small amount of remaining receptor accumulated in the cell’s interior. Even a single Y45 change substantially reduced binding and lowered overall EGFR levels. Yet, despite this disruption of the textbook EGF–EGFR signaling route, the cancer cells remained viable and continued to grow in culture with only modest changes to their cell cycle patterns.
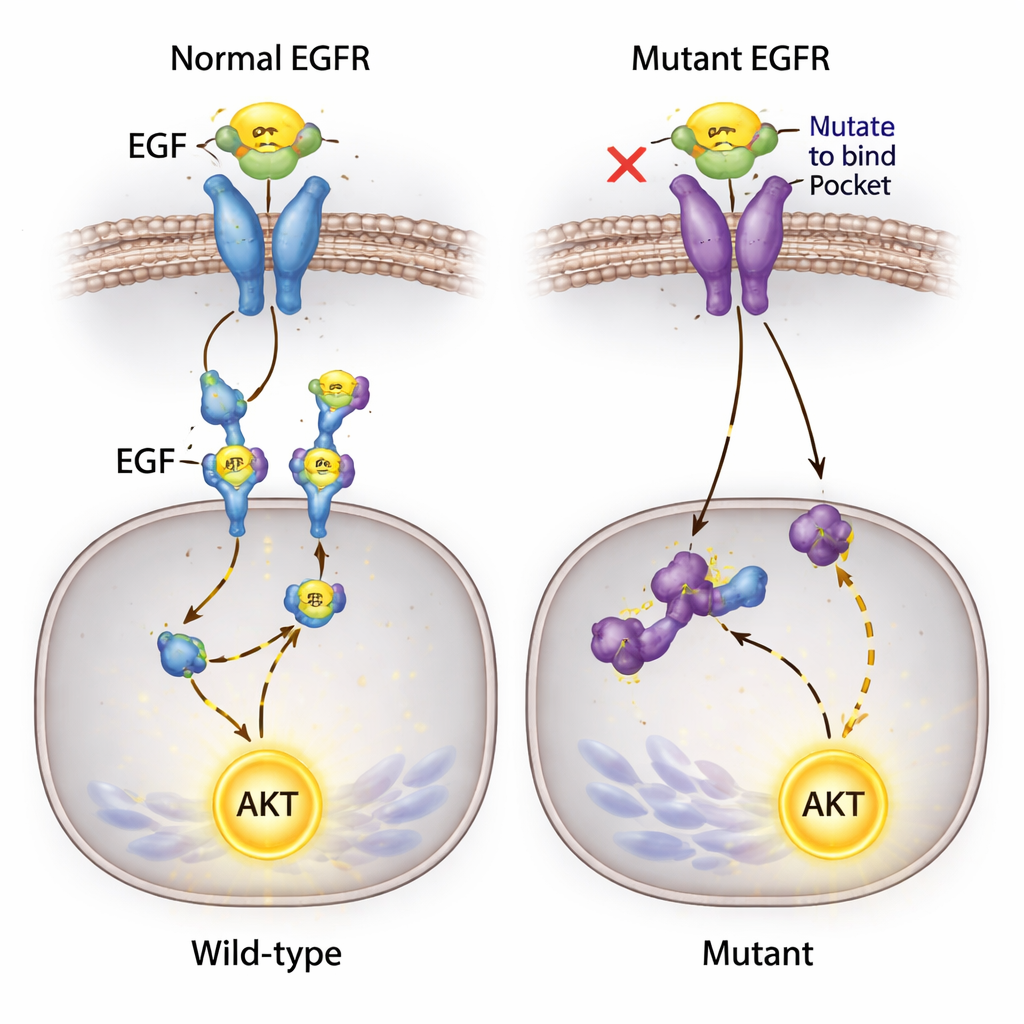
An Unexpected Signal That Won’t Quit
One of the most surprising findings involved AKT, a key protein that promotes cell survival and is usually activated downstream of EGFR. The researchers expected that blocking EGF–EGFR binding would silence this pathway. Instead, when they added EGF to the medium, AKT became strongly activated in all cell lines—including those where EGF could no longer bind EGFR and where receptor phosphorylation was undetectable. Because these cells carry a known mutation in another signaling gene, PIK3CA, the team suspects that alternative routes may allow EGF to feed into AKT activation, possibly through other surface proteins or background mutations, bypassing the edited EGFR.
Hidden Genetic Changes and Future Therapies
To make sure CRISPR was not causing widespread unintended edits, the scientists sequenced the entire genomes of the new cell lines. They confirmed that the desired EGFR mutations were present and found no evidence of cuts at predicted off‑target sites for the CRISPR guides. However, they did detect many spontaneous mutations scattered around the genome, some of which might influence how the cells behave. This underscores that even when CRISPR hits its intended address, cancer cells’ naturally unstable DNA can complicate how we interpret experimental results and how we design precise gene‑based therapies.
What This Means for Patients
For a layperson, the take‑home message is that simply blocking the handshake between EGF and EGFR—even very precisely at the atomic level—does not automatically stop cancer cells from receiving “grow and survive” commands. The study shows that cancer cells can reroute key signals like AKT activation around a blocked receptor, and that CRISPR editing itself must be evaluated carefully in such unstable genomes. These engineered cell lines now provide a powerful laboratory model to search for backup pathways that sustain EGFR‑dependent tumors, information that could ultimately lead to combination treatments targeting both EGFR and its hidden accomplices.
Citation: Popović, J., Hahut, A., Torres, G.E. et al. Changes in EGFR activity following CRISPR/Cas9-editing of the EGF binding domain. Sci Rep 16, 6797 (2026). https://doi.org/10.1038/s41598-026-37579-8
Keywords: EGFR, cervical cancer, CRISPR, EGF signaling, AKT pathway