Clear Sky Science · en
Exploring the impact of substituents and π-conjugation on structural, optical and nonlinear optical studies in ferrocene-appended D-π-A and D-D’-π-A chromophores
Light Tricks in Tiny Metal-Based Dyes
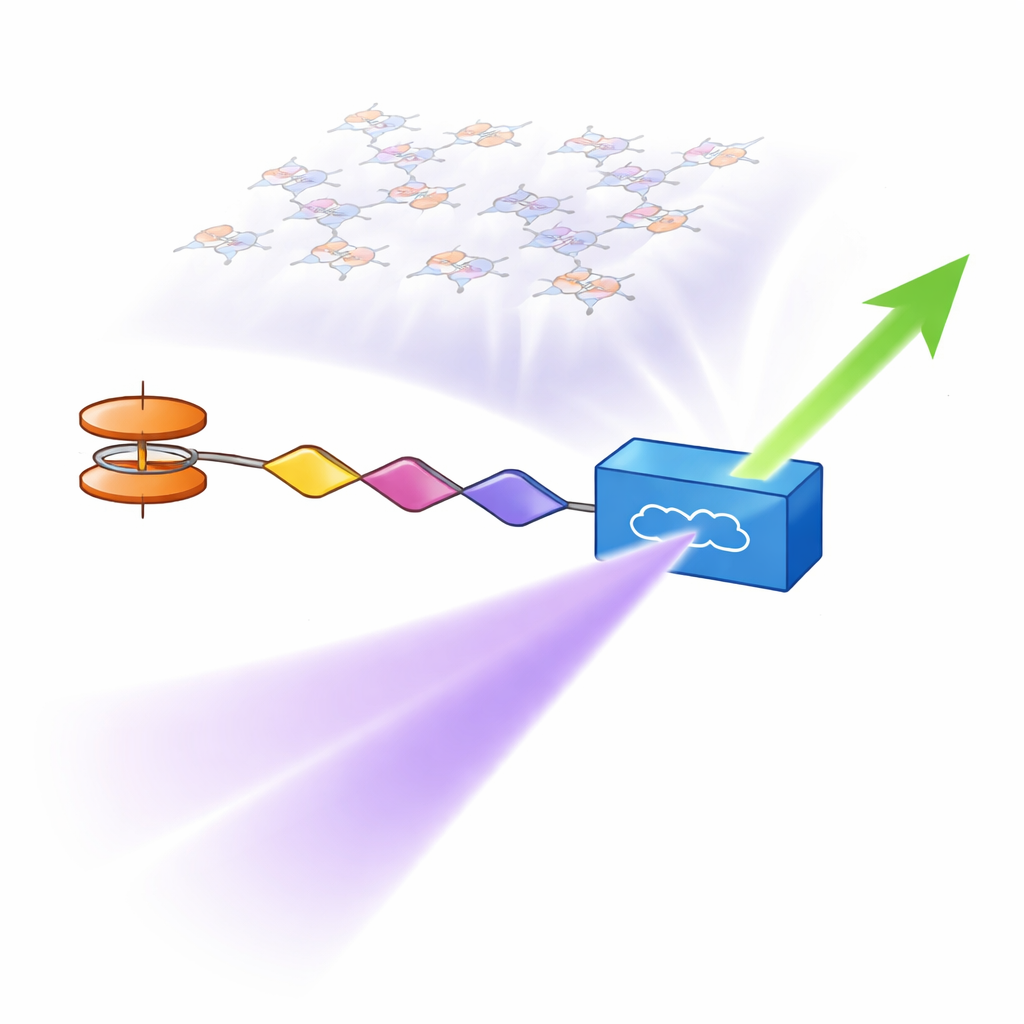
Modern technologies that move and process information with light—such as high-speed communications, sensing, and advanced imaging—depend on materials that can bend and transform laser beams in unusual ways. This paper explores a new family of metal-containing dye molecules that are specially built to turn one color of laser light into another more efficiently, a key trick in devices that double light frequency and generate new colors on demand.
Building Designer Light-Responsive Molecules
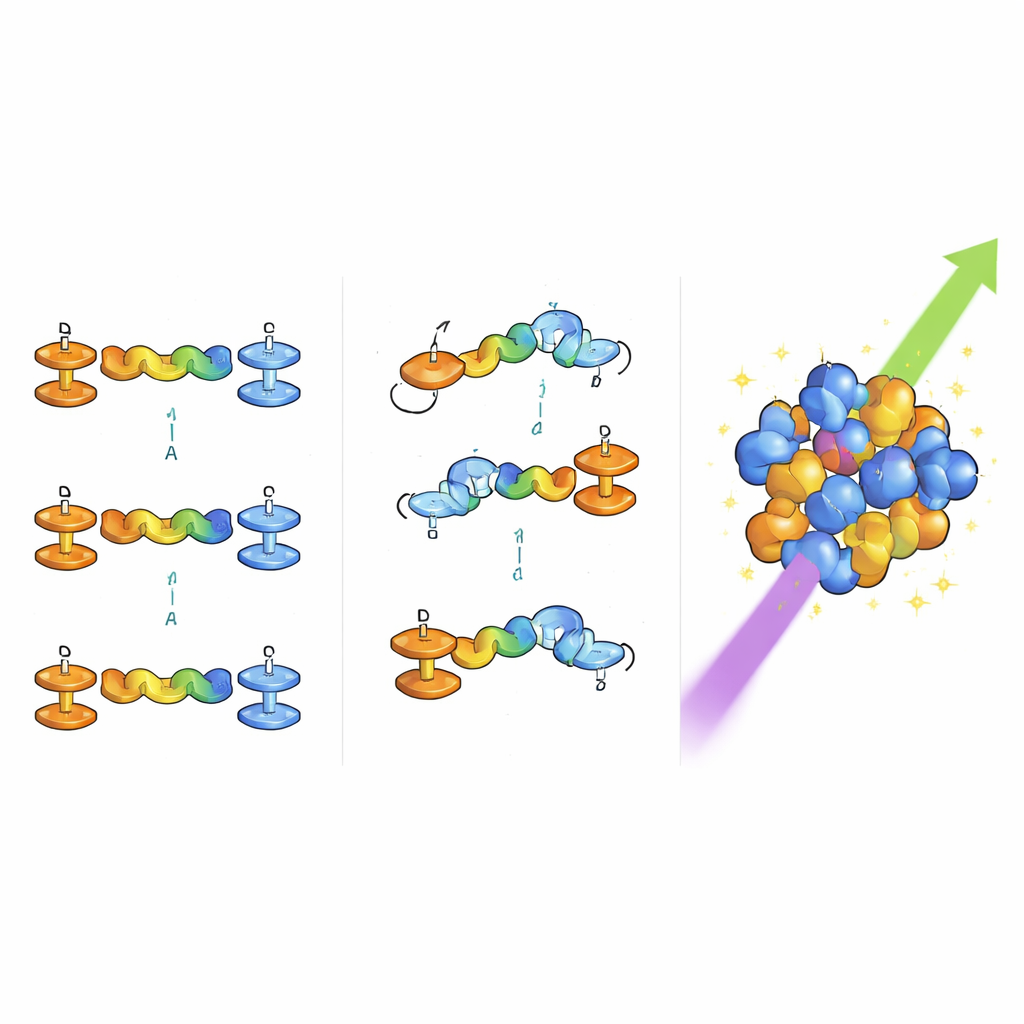
The researchers designed six closely related molecules, known as “push–pull chromophores,” that act a bit like microscopic batteries. One end strongly donates electrons, the other end strongly pulls them in, and the middle is a bridge that allows charge to shift when the molecule is hit by light. Here the donor side is based on ferrocene, a robust iron-containing unit, often paired with a methoxy-bearing ring, while the accepting side carries cyano and sometimes trifluoromethyl groups, which attract electrons. By adjusting these end groups and the length of the bridge, the team could fine-tune how charge moves through the molecule—an essential factor for amplifying nonlinear optical effects, where the light that comes out has a different color or frequency than the light that went in.
From Solution to Crystal: How Shape and Packing Matter
After synthesizing the six chromophores in a two-step process, the authors carefully checked their structures using a suite of spectroscopic tools and then examined selected examples as single crystals. X-ray diffraction revealed not only the exact positions of atoms but also how the molecules arrange themselves in the solid state. Although some crystals were formally “centrosymmetric” (with mirror-like symmetry that usually cancels out the desired optical effects), the molecules did not stack in simple head-to-tail pairs. Instead, subtle twists in the connected rings and a network of weak attractions—such as hydrogen bonds and interactions between hydrogen atoms and ring-shaped electron clouds—discouraged perfect back-to-back alignment. This imperfect packing turns out to be helpful, because it allows the molecules’ individual light-bending abilities to add up rather than cancel.
Watching Electrons Move and Light Get Absorbed
The team then explored how easily the molecules give up or take on electrons using electrochemical tests, and how they absorb light using ultraviolet–visible spectroscopy in solution and in thin plastic films. They identified several distinct bands corresponding to electron motion within the organic rings, between the donor and acceptor parts of the molecule, and between the metal center and its surroundings. Small changes in the chemical substituents shifted these bands to longer or shorter wavelengths, signaling stronger or weaker internal charge transfer. In particular, molecules with both ferrocene and methoxy donors, plus the strongly pulling trifluoromethyl and cyano groups, showed narrower energy gaps between their highest filled and lowest empty electron levels—a combination that favors stronger responses when light is applied.
Doubling Light and Checking Theory Against Experiment
To test their practical performance, the chromophores were ground into powders and evaluated with a standard laser-based method that measures second-harmonic generation—the conversion of infrared laser light into visible light at twice the frequency. All six materials outperformed a widely used reference crystal, potassium dihydrogen phosphate (KDP). One standout molecule, containing both methoxy and trifluoromethyl groups, produced a signal about 2.9 times stronger than KDP. Computer simulations using density functional theory backed up these findings, linking the observed efficiencies to features such as molecular twisting, charge distribution, and a measure called hyperpolarizability, which captures how strongly the electron cloud distorts under an electric field. Theoretical analysis of how bond lengths alternate along the molecular backbone further supported the idea that a carefully balanced, extended network of bonds enhances the nonlinear response.
Why These Molecules Matter for Future Photonics
Overall, the study shows that combining a metal-based donor like ferrocene with powerful accepting groups, joined by an extended but slightly twisted bridge, is a successful strategy for creating solid materials that double light efficiently. The best-performing molecule in this series not only rivals but surpasses common benchmark crystals, while maintaining good stability and processability in polymer films. For a general reader, the key message is that by fine-tuning both the chemistry and the three-dimensional packing of these “molecular springs,” scientists can coax materials to reshape light in highly controlled ways—a step toward more compact, efficient components for optical communication, sensing, and next-generation photonic devices.
Citation: Chithra, V.S., Prabu, S., Archana, P.P.S. et al. Exploring the impact of substituents and π-conjugation on structural, optical and nonlinear optical studies in ferrocene-appended D-π-A and D-D’-π-A chromophores. Sci Rep 16, 9524 (2026). https://doi.org/10.1038/s41598-026-37577-w
Keywords: nonlinear optics, ferrocene chromophores, second harmonic generation, push-pull molecules, optoelectronic materials