Clear Sky Science · en
Copy-number amplification drives IFI30 overexpression and coordinated immune activation, identifying a novel diagnostic and therapeutic target in gastric adenocarcinoma
Why a stomach protein matters
Gastric cancer is one of the world’s deadliest cancers, largely because it is often found late and can be hard to treat. This study focuses on a little-known protein called IFI30, found in cells that help the immune system recognize threats. By tracing how IFI30 is altered in stomach tumors and how it shapes the body’s defenses, the researchers suggest it could become a powerful tool for earlier diagnosis and for choosing more effective, personalized treatments.
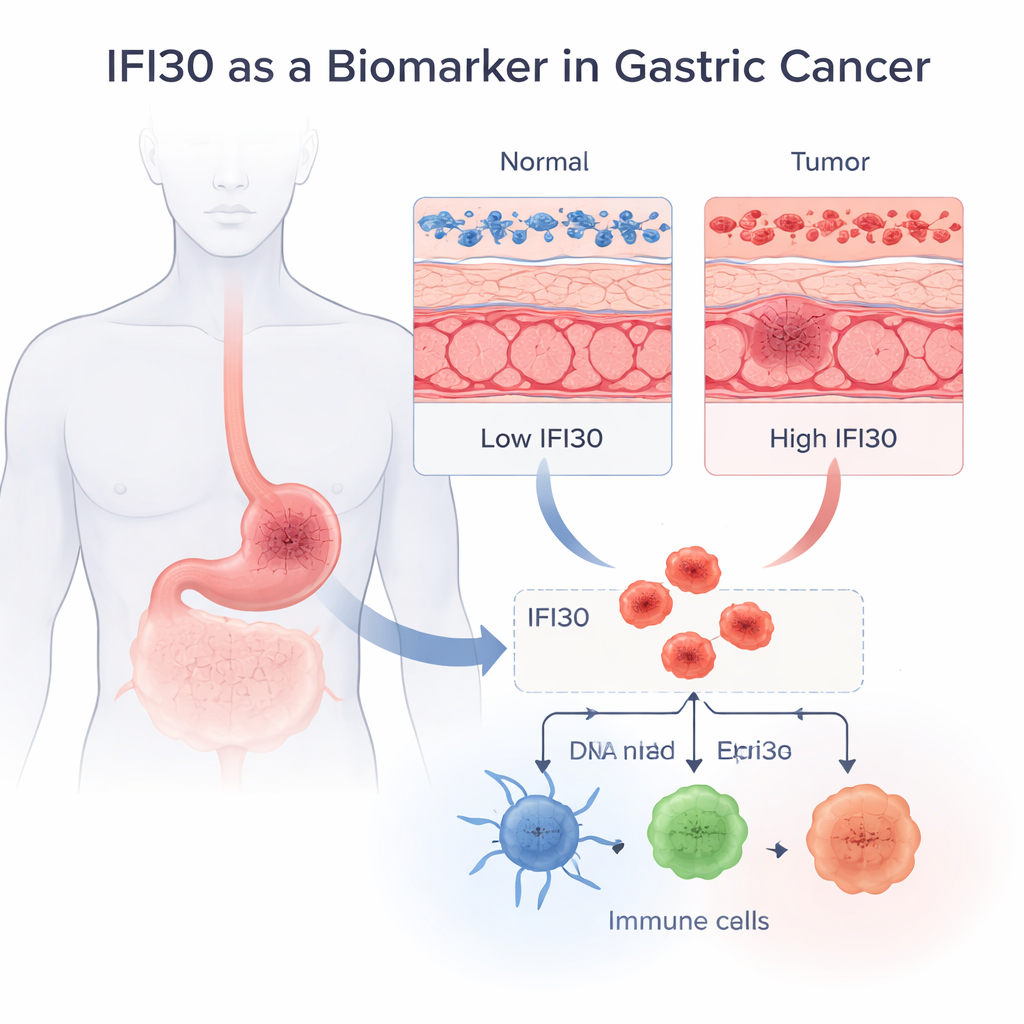
A hidden signal in stomach tumors
The team began by asking a basic question: is IFI30 present at different levels in healthy versus cancerous stomach tissue? Using large public datasets that combine gene activity from thousands of samples, they found that IFI30 is consistently much higher in gastric adenocarcinoma—the most common type of stomach cancer—than in normal stomach lining. This held true across multiple independent patient groups and at both the RNA and protein levels. When the authors tested how well IFI30 alone could distinguish tumor tissue from healthy tissue, its performance was striking: a diagnostic accuracy (AUC) of 0.92, which is considered excellent for a single marker. Microscopy images also placed IFI30 within the cell’s internal recycling system, where antigens are processed for immune recognition.
DNA changes that crank up IFI30
Next, the researchers looked at why IFI30 is so elevated in these tumors. Scanning cancer genomes from The Cancer Genome Atlas, they found that extra copies of the IFI30 gene—known as copy-number amplifications—were the main driver of its overexpression. Tumors with amplified IFI30 tended to show greater overall genome instability and often carried well-known cancer mutations such as TP53 and PIK3CA. As IFI30 copy number climbed, its RNA levels rose too, implying a direct link between the structural DNA changes and the heightened production of this protein. This paints IFI30 not as a passive bystander, but as part of a broader pattern of aggressive tumor biology.
Immune crosstalk around the tumor
Because IFI30 is involved in processing material that immune cells present as “wanted posters” to T cells, the team explored where, exactly, it sits within the tumor microenvironment. Single-cell sequencing, which reads out genes in individual cells, showed that IFI30 is especially enriched in dendritic cells, macrophages, and CD8 T cells—key players in launching and sustaining anti-tumor immunity. Network analyses revealed dense communication between IFI30-rich immune cells and surrounding tumor and support cells. Tumors with high IFI30 showed strong activation of pathways linked to antigen presentation, inflammatory signals, and communication molecules that attract and organize immune cells. At the same time, these tumors displayed features of invasion and stress, such as cell-cycle acceleration, tissue remodeling, and low-oxygen adaptation, highlighting IFI30’s role at the intersection of attack and defense within the cancer.
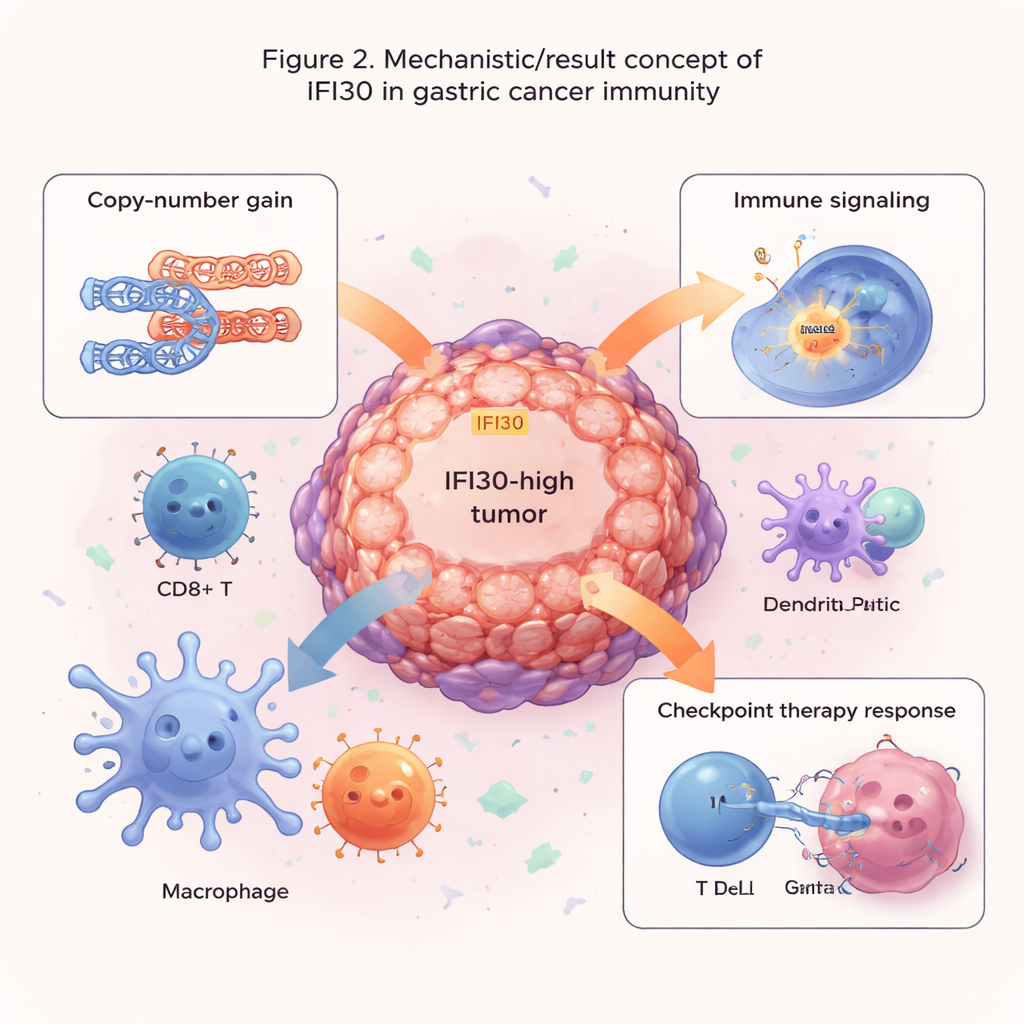
Clues for drug choice and immunotherapy
The authors then asked whether IFI30 could help predict how tumors respond to treatment. By pairing IFI30 levels with large drug-response datasets, they found that IFI30-high tumors tend to resist some medicines that target cell division or a common growth pathway (MAPK), but appear more sensitive to drugs that block EGFR and PI3K/AKT signaling. Notably, IFI30 levels tracked closely with PD-L1, a major brake on T cells that is targeted by today’s checkpoint inhibitors. Across several immunotherapy studies, patients whose tumors had higher IFI30 expression were more likely to respond to checkpoint blockade than those with low levels. An IFI30-based score outperformed established measures such as tumor mutation burden and PD-L1 alone, and was especially enriched in tumors with high microsatellite instability, a subgroup already known to respond better to immunotherapy.
From lab bench to possible new target
To test whether IFI30 is just a marker or also helps drive cancer growth, the team reduced its levels in gastric cancer cell lines using genetic tools. When IFI30 was knocked down, cancer cells grew more slowly, pointing to a direct role in sustaining tumor proliferation. Taken together, the data suggest that IFI30 is both a readout of immune activity and a contributor to malignant behavior. For patients, this means that a simple measure of IFI30 could one day help doctors detect gastric cancer more reliably, predict who might benefit from certain targeted drugs or immunotherapies, and perhaps guide new treatments that directly or indirectly block IFI30’s tumor-promoting actions.
Citation: Liu, Q., Yuan, W., Zhaowang, R. et al. Copy-number amplification drives IFI30 overexpression and coordinated immune activation, identifying a novel diagnostic and therapeutic target in gastric adenocarcinoma. Sci Rep 16, 6945 (2026). https://doi.org/10.1038/s41598-026-37574-z
Keywords: gastric cancer, IFI30, biomarker, tumor immunity, immunotherapy