Clear Sky Science · en
Computational analysis of X2CaZnH6 (X = K, Rb and Cs) hydrides for hydrogen storage
Hydrogen in a Box
Hydrogen is often hailed as a clean fuel of the future, but storing it safely and compactly remains a major obstacle. This study explores a new family of crystal materials that can hold hydrogen inside their atomic framework, much like water in a sponge. By designing these compounds on a computer before anyone makes them in a lab, the authors show how they might serve as solid fuels for future hydrogen-powered technologies.
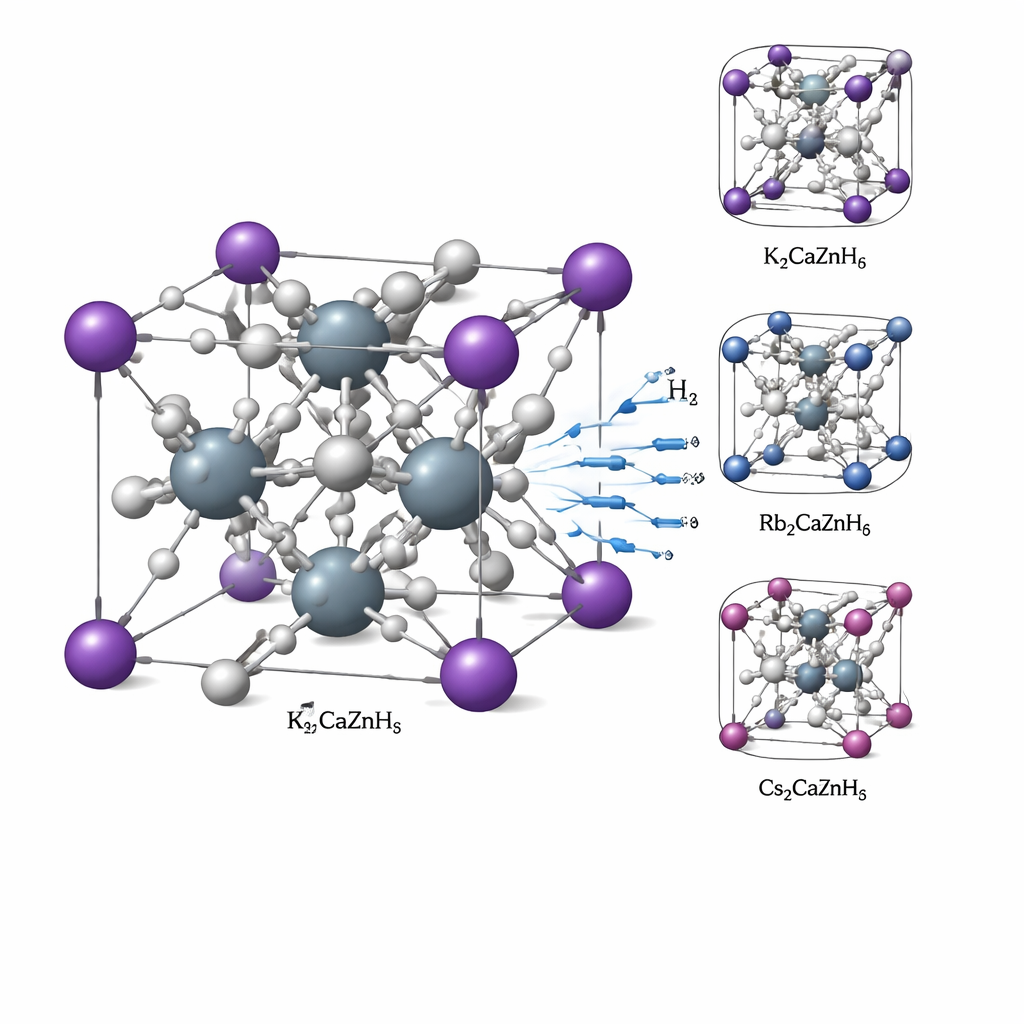
A Crystal Built Like a 3D Lattice
The materials examined are called double perovskite hydrides, with the formula X2CaZnH6, where X can be potassium (K), rubidium (Rb), or cesium (Cs). All three form a simple cube-like structure in which heavy atoms sit at well-defined positions and hydrogen atoms occupy spaces around them. Using quantum-mechanical calculations, the authors confirm that these crystals are structurally sound: the atoms fit together with the right sizes, the overall energy of the material is low enough that it should form naturally, and the vibrations of the atoms do not hint at any hidden instabilities. Mechanical tests carried out in silico show that the crystals are stiff yet not overly fragile, a balance that helps them keep their shape while still allowing small rearrangements when hydrogen is taken up or released.
How Much Hydrogen Can They Hold?
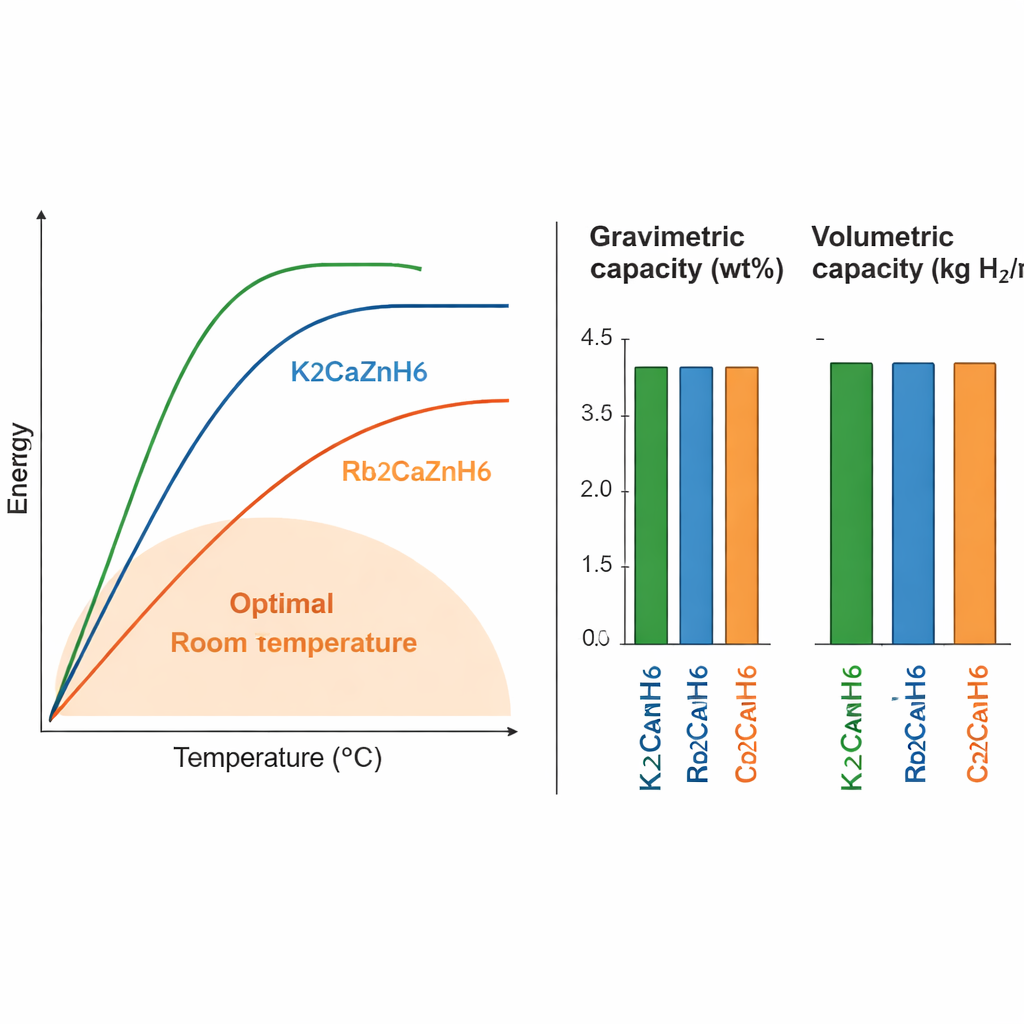
For any storage material, two key numbers are how much hydrogen it can hold by weight (gravimetric capacity) and by volume (volumetric capacity), as well as the temperature at which hydrogen can be released. The three compounds studied here store between about 1.6 and 3.2% of their weight as hydrogen, and about 15 to 18 kilograms of hydrogen per cubic meter of material. The potassium-based version, K2CaZnH6, holds the most hydrogen but needs higher temperatures (around 658 K, or 385 °C) to release it. The cesium version stores slightly less hydrogen and still requires fairly high temperatures. The rubidium compound, Rb2CaZnH6, stands out: it releases hydrogen at about 385 K (roughly 110 °C), a range much closer to what is considered practical for real devices, while keeping respectable storage density.
Why the Atoms and Electrons Matter
To understand why these materials behave as they do, the authors examine how electrons are shared between atoms and how this affects bonding. In all three compounds, potassium, rubidium, or cesium act as electron donors, while zinc, calcium, and hydrogen attract electrons. The hydrogen atoms pick up a partial negative charge, and their bonds to calcium and zinc are largely ionic with a touch of covalent character. This kind of bonding tends to hold hydrogen strongly enough that it does not leak out easily, yet weakly enough that heating can release it when needed. Importantly, hydrogen atoms do not form strong H–H bonds inside the crystal, meaning hydrogen is stored as separate atoms rather than pre-formed molecules, which is favorable for controlled release.
Light, Electricity, and Strength
These crystals are also semiconductors, with a modest energy gap between filled and empty electronic states. That means they can interact with light over a wide range of colors, especially in the visible and ultraviolet parts of the spectrum. The calculations show strong light absorption and significant optical conductivity, raising the possibility that light could help trigger hydrogen release, a kind of solar-assisted storage. At the same time, the materials meet standard criteria for mechanical stability: they resist compression, shear, and fracture within reasonable limits, and their atomic vibrations at room temperature remain well behaved in computer-simulated heating tests. Together, these traits point to a robust framework that can endure repeated hydrogen loading and unloading.
What This Means for Future Energy Systems
In everyday terms, the study identifies three new "hydrogen sponge" materials that are stable, reasonably strong, and able to pack hydrogen into a compact solid. Although their hydrogen content by weight does not yet meet the most ambitious targets, their volumetric storage is promising, and the rubidium-based compound in particular operates at temperatures compatible with many practical systems. Because their behavior can be tuned simply by swapping one alkali element for another, these double perovskite hydrides offer a flexible platform for designing better solid hydrogen fuels, and potentially for combining storage with light-driven control in future clean-energy technologies.
Citation: Al-Zoubi, N., Almahmoud, A., Almahmoud, A. et al. Computational analysis of X2CaZnH6 (X = K, Rb and Cs) hydrides for hydrogen storage. Sci Rep 16, 6889 (2026). https://doi.org/10.1038/s41598-026-37564-1
Keywords: hydrogen storage, metal hydrides, double perovskite, solid-state energy, clean fuels