Clear Sky Science · en
Hybrid deep learning novel framework for classification of parkinson’s disease
Why this matters for patients and families
Parkinson’s disease often begins with subtle changes—slower movements, a softer voice, or a faint tremor—that can easily be mistaken for normal aging. By the time the diagnosis is certain, much of the brain damage is already done. This study presents a computer-based approach that reads brain scans to flag Parkinson’s disease with very high accuracy, even when signs are still mild. If such tools can be trusted and widely adopted, they could help doctors diagnose earlier, treat sooner, and give patients and families more time to plan.
Looking inside the brain with smart scans
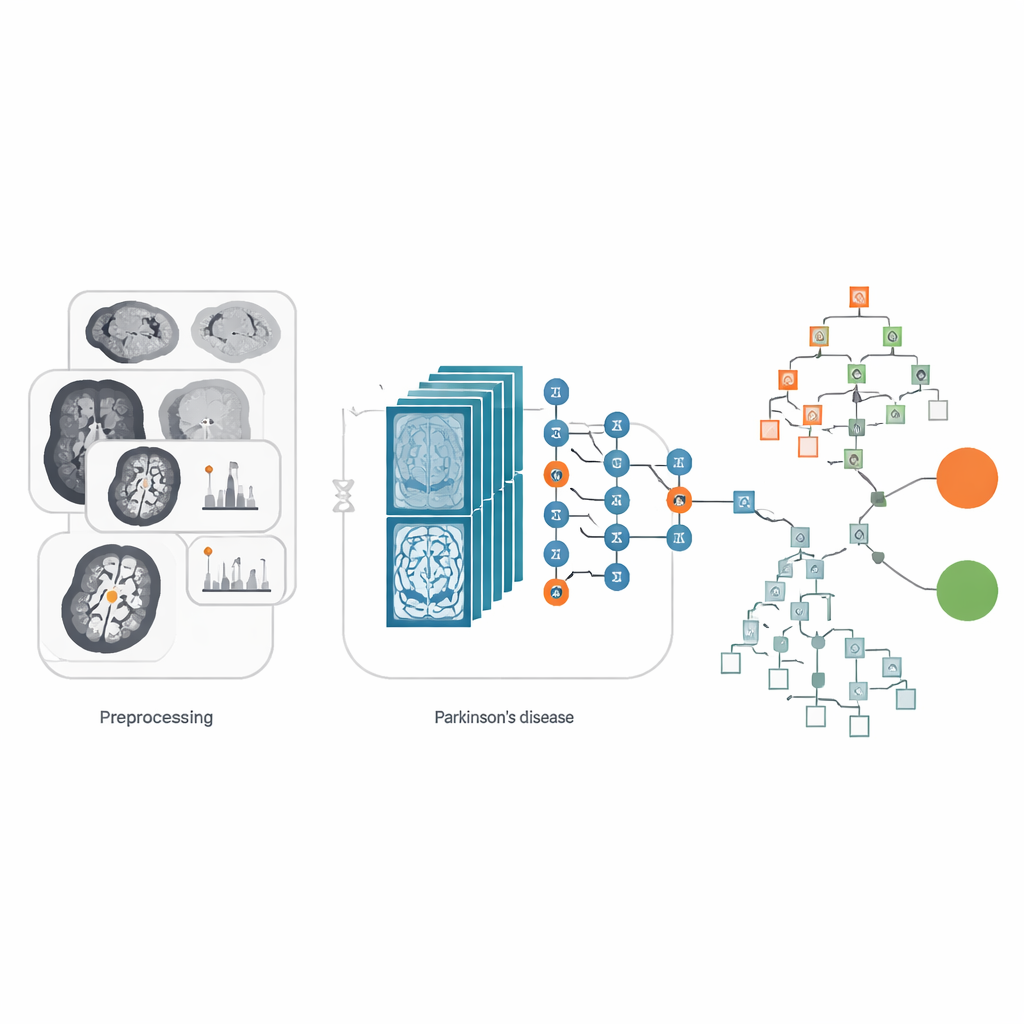
The researchers focused on magnetic resonance imaging (MRI), a scan that is already common in hospitals and does not involve radiation. They used a large public collection of brain images from the Parkinson’s Progression Markers Initiative, which includes scans from people with Parkinson’s disease and from healthy volunteers. Rather than analyzing the entire 3D scan at once, the team worked with slice-by-slice views through the brain, especially the midbrain region where cells that produce dopamine are found. These cells are crucial for smooth movement, and their loss is a hallmark of Parkinson’s disease. To make faint structures easier for a computer to “see,” the images first went through careful adjustments that standardize brightness and enhance contrast in key areas.
Teaching a hybrid model to spot patterns
At the heart of the study is a hybrid system that combines two powerful ideas from modern computing. The first is a deep learning network called EfficientNetB0, originally designed for general image recognition tasks. Here, it acts like a highly trained eye, scanning each brain slice to extract subtle patterns of shape and texture that may separate healthy brains from those affected by Parkinson’s disease. The second is a decision-making method known as XGBoost, which excels at drawing sharp boundaries between categories once it is given informative features. In simple terms, EfficientNetB0 distills each MRI slice into a compact signature, and XGBoost uses these signatures to decide whether the scan belongs to a healthy person or someone with Parkinson’s disease, and whether the images are of a particular MRI type.
Balancing limited and uneven data
One of the biggest practical problems in medical AI is that data are often both scarce and unbalanced: there may be many more scans from patients than from healthy volunteers, or vice versa. Here, the team started with scans from 77 healthy and 223 Parkinson’s subjects, a modest number for deep learning. To avoid teaching the system lopsided lessons, they expanded the image set in a controlled way. Each original brain slice was rotated or flipped to mimic the small differences in head position that occur during real scans. They also applied a contrast enhancement technique called CLAHE, which brightens fine details without exaggerating noise. This careful “stretching” of the dataset produced more than 26,000 images, giving the model enough variety to learn robust patterns while reducing the risk of overfitting to quirks in the original scans.
How well does the system perform?
To gauge reliability, the researchers compared several versions of their approach. They tested three popular image networks—VGG16, ResNet50, and EfficientNetB0—in three modes: used as-is, fine-tuned, and combined with the XGBoost decision stage. Across these options, the hybrid setups consistently did best. The winning configuration, EfficientNetB0 plus XGBoost, correctly classified scans 99.02 percent of the time in the test set. It handled all four categories well: healthy and Parkinson’s, each with and without a particular MRI setting known as FLAIR. Measures that focus on missed cases and false alarms, such as recall and F1-score, were also very high, suggesting the tool is not simply trading one type of error for another. Importantly, the model remained efficient in terms of computing time, making it more realistic for use in busy clinical environments.
What this could mean in the clinic
While no computer system can replace a skilled neurologist, this work shows that a carefully designed hybrid model can act as a powerful assistant. By rapidly scanning routine MRI images for subtle signs of Parkinson’s disease, such tools could flag at-risk patients for closer follow-up long before symptoms become obvious. The authors caution that their work still needs to be tested on larger and more diverse patient groups, and eventually combined with other information such as movement tests or blood markers. Even so, their results suggest that smart software built on existing hospital scans could become a fast, low-cost, and widely deployable ally in the early detection of Parkinson’s disease.
Citation: Desai, S., Vora, M., Shah, S. et al. Hybrid deep learning novel framework for classification of parkinson’s disease. Sci Rep 16, 9143 (2026). https://doi.org/10.1038/s41598-026-37553-4
Keywords: Parkinson’s disease, brain MRI, deep learning, medical imaging, early diagnosis