Clear Sky Science · en
Insights into the structural differences between homo- and heterodimers enriched from a cocktail of monoclonal antibodies against SARS-CoV-2
Why mixing antibodies can create surprises
During the COVID-19 pandemic, antibody drugs such as the REGEN-COV cocktail were used to block the virus that causes the disease, SARS-CoV-2. These medicines are carefully designed, but once mixed together in a vial they can form rare pairings called heterodimers—two different antibodies joined as a pair. This study asks a practical question with big safety and efficacy implications: when these unusual pairs form, do they behave differently from the more familiar pairs of identical antibodies, and could that change how well the drug works?
Unusual pairs in a powerful COVID-19 drug
REGEN-COV combines two monoclonal antibodies, casirivimab and imdevimab, each designed to latch onto the spike protein of SARS-CoV-2 in a different way. When both are formulated together, a tiny fraction of the antibodies hook up in pairs. Some pairs are homodimers (two copies of the same antibody); others are heterodimers (one casirivimab plus one imdevimab). Heterodimers are considered an impurity because they are not part of the original drug design, and their behavior in the body has been largely unknown. The authors first used a multi-step purification process to isolate several distinct homodimer species and the heterodimer from the same REGEN-COV batch, giving them a rare opportunity to compare all of these assemblies side by side.
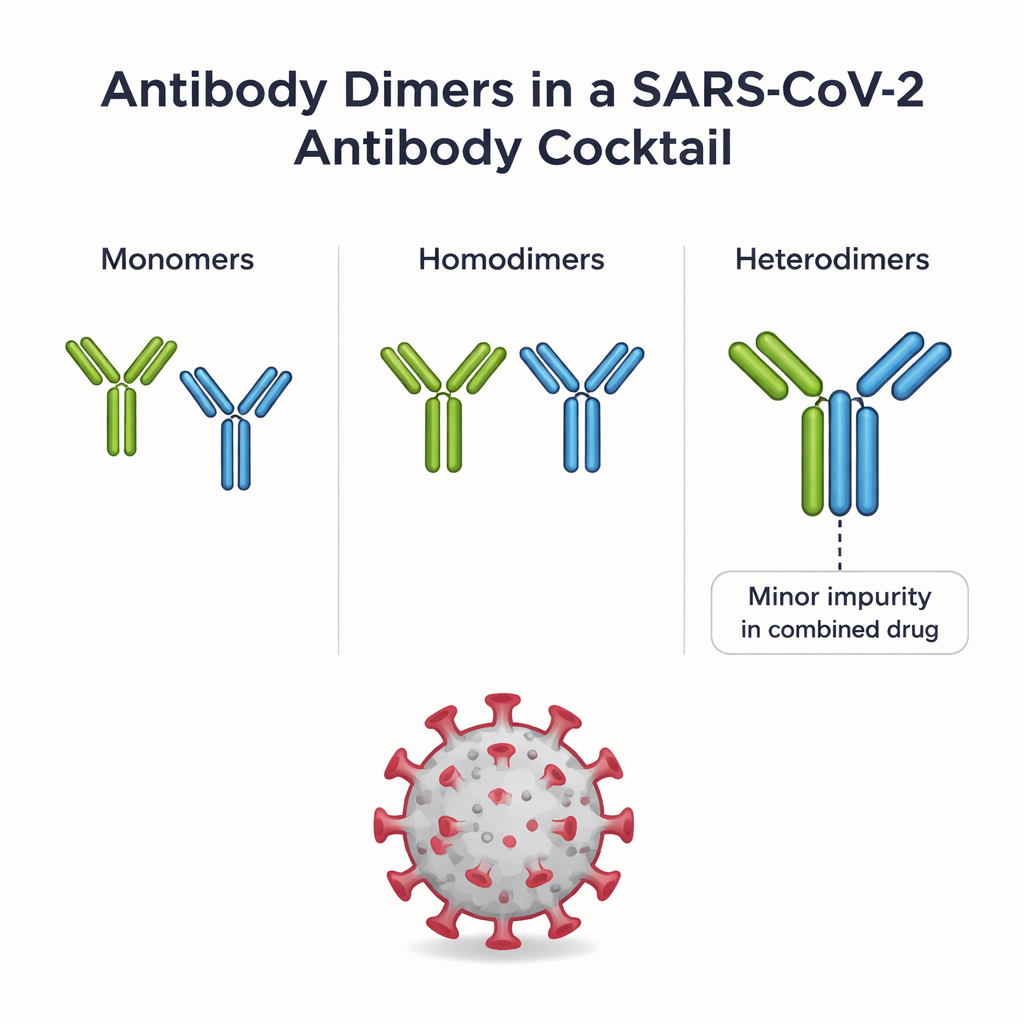
Probing shape, stickiness, and chemical bonds
To understand what makes each dimer type unique, the team combined several high-end analytical tools. Chromatography experiments revealed how the dimers differ in surface "stickiness" (hydrophobicity), while capillary electrophoresis and a specialized form of mass spectrometry showed whether the two antibodies in a dimer are held together by weak attractions or by stronger covalent bonds. Electron microscopy and analytical ultracentrifugation then provided insight into overall shape—whether a dimer is elongated and flexible or compact and tightly packed. Together, these methods showed that casirivimab and imdevimab form several distinct homodimers with different shapes and bonding patterns, and that the heterodimer is especially compact and enriched in covalent links involving different parts of the antibody arms and stems.
How structure connects to virus blocking and cell killing
The researchers next asked how these structural differences affect what really matters: stopping the virus and engaging the immune system. They tested each dimer in a pseudovirus neutralization assay, which measures how well the antibodies block entry of a virus-like particle, and in an antibody-dependent cellular cytotoxicity (ADCC) assay, which gauges how effectively antibodies recruit immune cells to destroy infected targets. Surprisingly, most dimer forms were at least as good as, and sometimes better than, their individual monomer counterparts at neutralizing virus. In particular, imdevimab homodimers showed "hyperpotent" neutralization, likely because their geometry allows them to bridge multiple spike proteins on the viral surface. Some casirivimab homodimers produced especially strong ADCC signals, apparently because their orientation leaves the tail regions (the Fc portions) well exposed to immune cell receptors.
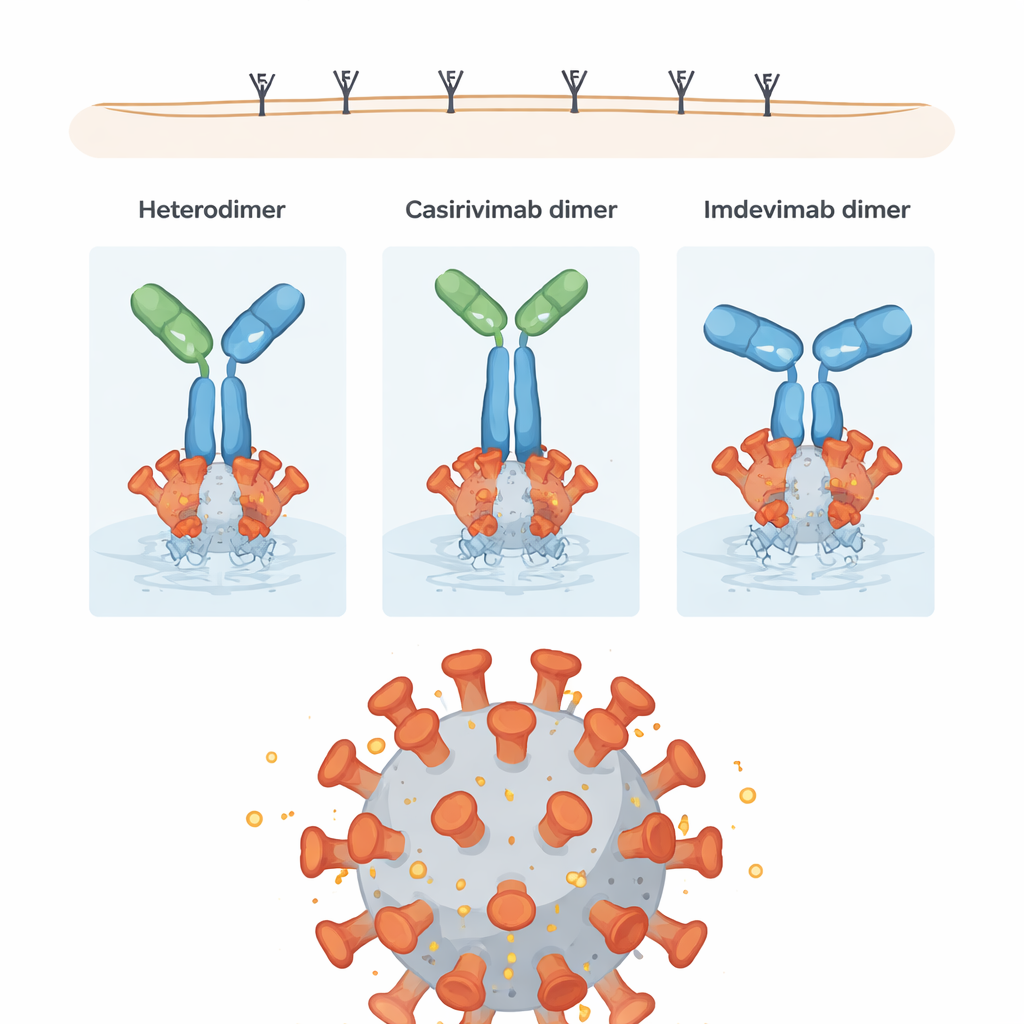
A compact impurity with mixed performance
The heterodimer, despite being rich in strong, compact connections between its two different antibodies, showed somewhat reduced performance: about 70% of the neutralizing and ADCC activity of the intended co-formulated mixture. The extensive crosslinking between its arms and stems appears to restrict how freely the molecule can rotate and reorient itself when bound to the viral spike, which in turn may limit both blocking of the virus and signaling to immune cells. Still, many of its bonds are of a type that can slowly break under physiological conditions, making the heterodimer relatively unstable over time and prone to fall back into individual antibodies.
What this means for antibody medicines
For patients and drug developers, the take-home message is reassuring but nuanced. This deep dive into REGEN-COV dimers suggests that most dimeric forms, including the unusual heterodimer impurity, retain substantial antiviral activity, and there is no clear signal of added safety risk from higher-order aggregates in this system. At the same time, the work shows that fine details—where antibodies touch, how tightly they are crosslinked, and how compact or elongated they are—can meaningfully shift potency. The authors argue that similar structural and functional profiling of dimers in other antibody drugs could become a powerful non-clinical way to understand and manage impurities, and might even inspire new, deliberately engineered multimeric antibodies that take advantage of geometry to better match and neutralize viruses.
Citation: Nguyen, J.B., Liu, S., Yan, Y. et al. Insights into the structural differences between homo- and heterodimers enriched from a cocktail of monoclonal antibodies against SARS-CoV-2. Sci Rep 16, 7024 (2026). https://doi.org/10.1038/s41598-026-37545-4
Keywords: antibody dimers, REGEN-COV, SARS-CoV-2 spike, monoclonal antibody cocktails, protein aggregates