Clear Sky Science · en
TIPE2 serves as a favorable prognostic biomarker and suppresses cholangiocarcinoma progression by targeting RAC1-mediated integrin αvβ6 trafficking
Why this rare bile duct cancer matters
Cholangiocarcinoma, a cancer of the bile ducts inside and around the liver, is uncommon but devastating. Most patients are diagnosed late, surgery is rarely possible, and fewer than one in twenty survive five years. Doctors urgently need better ways to predict which patients will do poorly and new angles for treatment. This study uncovers a molecule called TIPE2 that both helps forecast outcomes and appears to slow the cancer’s growth and spread.
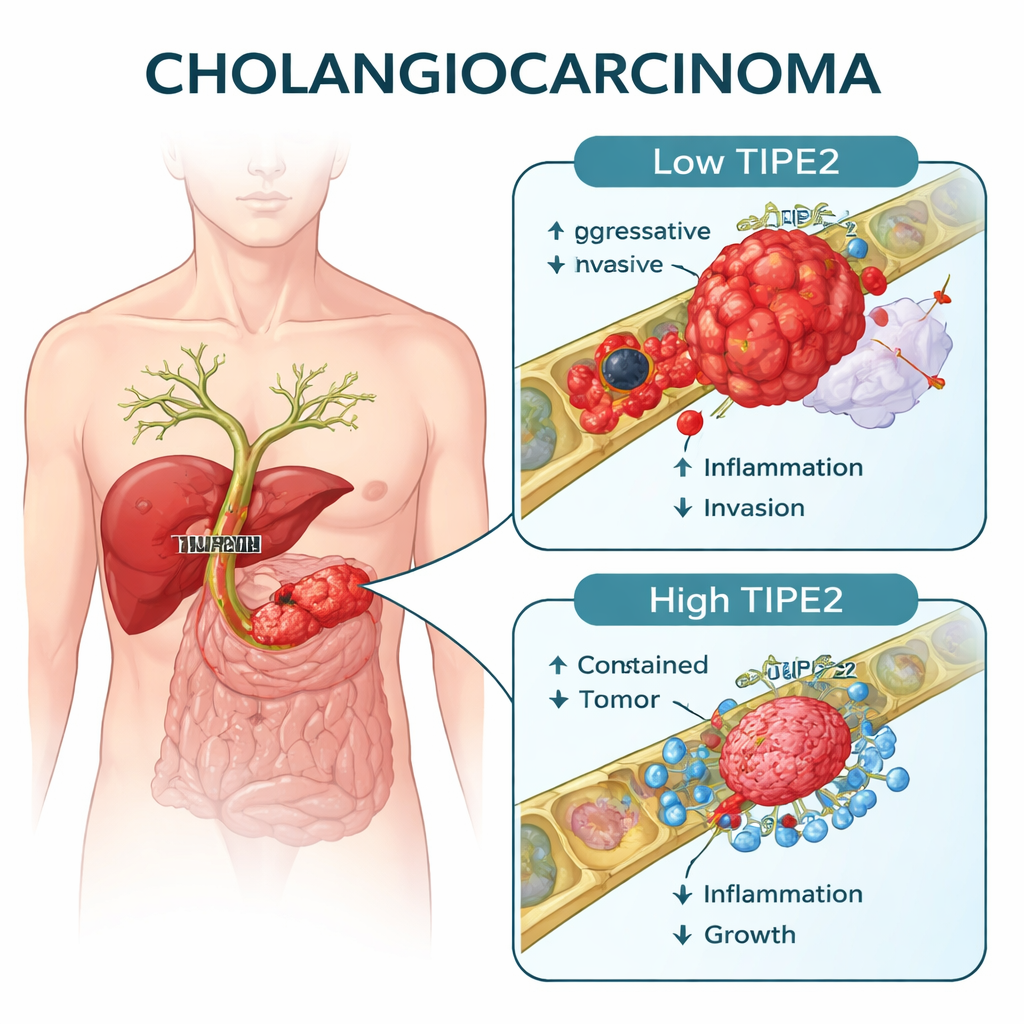
A built‑in brake that goes missing
TIPE2 is a protein best known for calming down the immune system, preventing runaway inflammation. The authors wondered whether it might also act as a natural brake in bile duct cells that turn cancerous. They examined tumor samples from a large group of 218 patients who had surgery for cholangiocarcinoma, and from a second, independent group of 95 patients. Using immunohistochemistry, a staining method that shows how much of a given protein is present in tissue, they measured TIPE2 levels in tumors and in nearby noncancerous bile duct tissue.
Less TIPE2, more aggressive tumors
Across both patient groups, TIPE2 was consistently lower in cancer tissue than in normal bile ducts. Moreover, the less TIPE2 a tumor had, the more advanced and aggressive it tended to be. Low TIPE2 was linked to larger tumors, spread to nearby lymph nodes, invasion of blood vessels, and higher overall cancer stage. When the team followed patients over time, those with low TIPE2 died sooner than those whose tumors still expressed higher amounts. Statistical analyses showed that TIPE2 was an independent predictor of survival, even after accounting for standard measures such as tumor size and lymph‑node status.
Turning a biomarker into a clinical tool
To make their findings useful at the bedside, the researchers built a “nomogram,” a visual scoring tool that combines several risk factors into a single prediction. In this chart, each patient’s tumor size, lymph‑node involvement, blood‑vessel invasion, and TIPE2 level are translated into points that estimate the chances of being alive one, three, or five years after surgery. The model performed better than the conventional TNM staging system alone, suggesting that adding TIPE2 staining to routine pathology reports could sharpen doctors’ ability to counsel patients and choose follow‑up treatments.
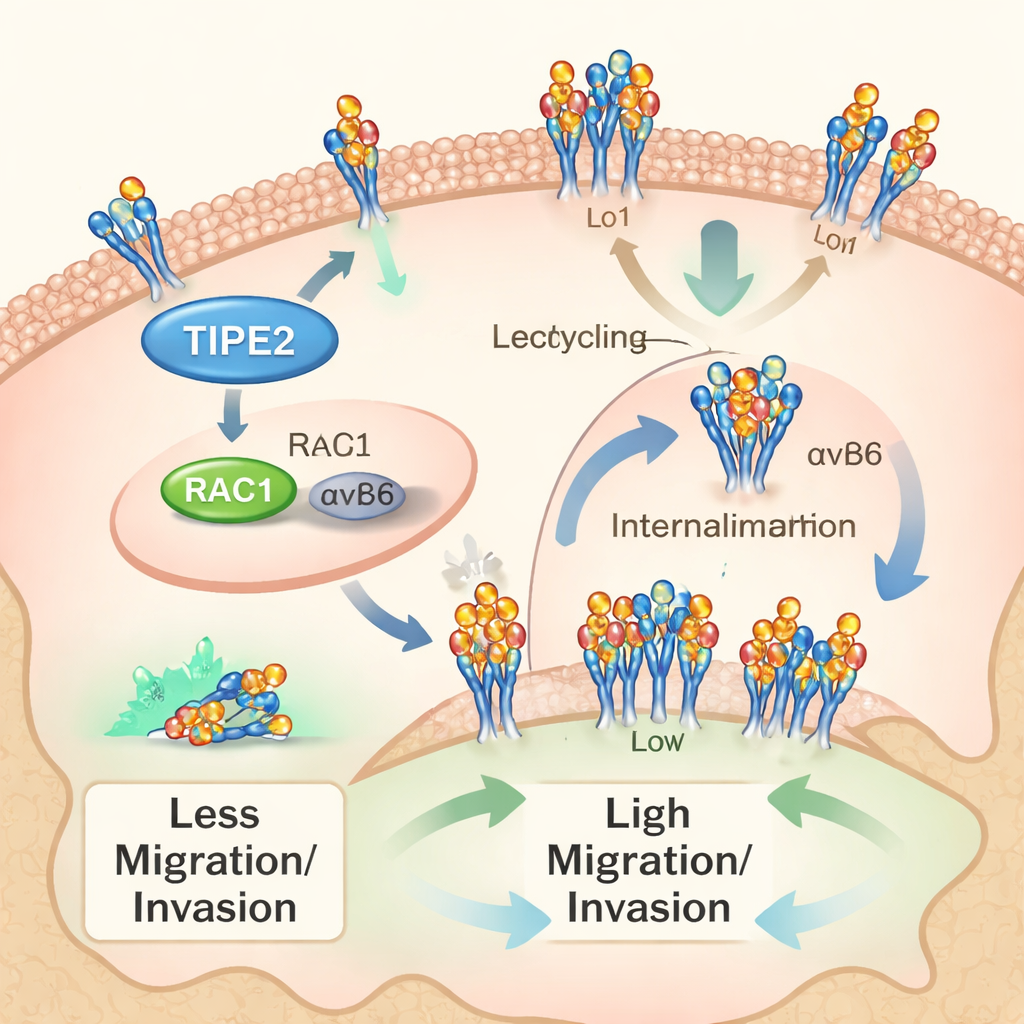
How TIPE2 restrains cancer cells
Beyond prognosis, the team explored how TIPE2 affects cancer behavior. In laboratory experiments with human cholangiocarcinoma cell lines, artificially lowering TIPE2 made cells grow faster and move and invade more readily through artificial membranes. Boosting TIPE2 had the opposite effect, slowing cell division and blunting their ability to migrate and penetrate barriers. In mice implanted with bile duct cancer cells, delivering extra TIPE2 directly into the tumors shrank their size and weight, showing that TIPE2 can curb tumor growth in living animals as well as in dishes.
A traffic problem on the cell surface
The study zeroed in on another player called integrin αvβ6, a cell‑surface adhesion molecule that helps cancer cells latch onto and remodel their surroundings, aiding invasion and spread. The researchers had previously shown that αvβ6 is abundant and harmful in cholangiocarcinoma. Here, they focused on the “trafficking” of αvβ6—its constant internalization into the cell and recycling back to the surface. Using biochemical tracking assays, they found that this shuttling process is active in bile duct cancer cells and that blocking internalization dampens cell growth and invasiveness. TIPE2, they discovered, interferes with both internalization and recycling of αvβ6, reducing the number and mobility of these molecules at the cell surface.
A key molecular switch: RAC1
Linking these pieces is RAC1, a small molecular switch that controls cell movement and the movement of membrane proteins. The authors showed that inhibiting RAC1 slows αvβ6 trafficking, much like TIPE2 does. Earlier work had revealed that TIPE2 can bind RAC1 and keep it in check. Computational modeling in this study suggested physical contacts among TIPE2, RAC1, and the tail of the αvβ6 integrin. When the team used a mutated form of TIPE2 that cannot properly interact with RAC1, its ability to slow cancer cell growth and invasion largely disappeared. Similarly, when cells were pre‑treated with a RAC1‑blocking drug, adding more TIPE2 produced much less additional benefit. Together, these findings support a pathway in which TIPE2 tames RAC1, which in turn reduces αvβ6 trafficking and the aggressive behavior of cholangiocarcinoma cells.
What this means for patients
For non‑specialists, the message is twofold. First, TIPE2 staining in tumor samples could give doctors a clearer picture of how dangerous a given bile duct cancer is, improving risk prediction beyond current staging systems. Second, TIPE2 itself, and the RAC1–αvβ6 path it controls, point toward fresh treatment ideas: boosting TIPE2 activity or directly targeting RAC1 or αvβ6 might help slow this otherwise lethal cancer. While such therapies will require more research and clinical testing, this work maps out a promising route toward more precise and effective care for patients with cholangiocarcinoma.
Citation: Wang, S., Jia, W., Sun, Y. et al. TIPE2 serves as a favorable prognostic biomarker and suppresses cholangiocarcinoma progression by targeting RAC1-mediated integrin αvβ6 trafficking. Sci Rep 16, 6638 (2026). https://doi.org/10.1038/s41598-026-37540-9
Keywords: cholangiocarcinoma, TIPE2, biomarker, integrin αvβ6, RAC1