Clear Sky Science · en
Machine learning-driven performance prediction of Z-scheme g-C3N4/SnS2 heterostructure photocatalyst for complete mineralization of indigo carmine and elucidation of degradation pathways
Turning Sunlight into a Water-Cleaning Tool
Many everyday products—from jeans and paper to medicines and food coloring—use strong synthetic dyes that can linger in rivers and lakes for years. One such dye, indigo carmine, gives rich blue color but is also toxic and hard to remove once it reaches wastewater. This study explores a sunlight-powered material that can not only strip the color from this dye but also break it all the way down into simple, harmless substances, with a little help from machine learning to predict how well the process will work in real-world conditions.
A Stubborn Blue Dye in Our Water
Indigo carmine is widely used because it is cheap, vivid, and stable—exactly the traits that make it difficult to clean up. Common treatment methods such as filtration, adsorption onto solids, or chemical coagulation often just move the dye from water to another place rather than truly destroying it. Worse, the leftover sludge can pose its own disposal problems. What is urgently needed are methods that actually “mineralize” the dye, meaning they break it down into basic molecules like carbon dioxide and water, leaving no persistent organic pollution behind.
Designing a Light-Driven Cleaning Material
The researchers created a new photocatalyst—a material that uses light to drive chemical reactions—by combining two known semiconductors into a so‑called Z‑scheme pair. One component, graphitic carbon nitride (g‑C3N4), is a metal-free, layered material that can absorb visible light but suffers from rapid loss of excited charges. The other, tin disulfide (SnS2), is a narrow‑band material that captures sunlight efficiently and sticks well to dyes but by itself mainly acts like a sponge, holding the dye instead of destroying it. Using a simple one‑pot thermal process, the team anchored tiny SnS2 particles onto thin sheets of g‑C3N4 at different loadings, forming tightly linked “heterostructures” that were confirmed by X‑ray, electron microscopy, and spectroscopy measurements to be well-coupled and structurally clean. 
From Colored Waste to Clear Water
When these materials were tested under real sunlight against water containing indigo carmine, one sample stood out: the composite with 5 percent SnS2 (called GS5). It removed all visible dye within 30 minutes at a modest amount of catalyst, and mineralized about three‑quarters of the organic carbon, showing that most of the dye was actually destroyed, not just hidden. Even at five times higher dye concentration, the same material still removed nearly 89 percent of the pollution, clearly outperforming the individual ingredients alone and similar systems reported in other studies. The catalyst also worked across a broad pH range, tolerated repeated reuse over five cycles, and kept its structure intact, suggesting it could be practical for continuous water treatment.
How the Catalyst Works at the Atomic Scale
At the heart of the improvement is how the two components share and separate electric charges when sunlight hits them. In a Z‑scheme arrangement, electrons from one material recombine with holes in the other, leaving behind especially strong oxidizing “holes” and reducing electrons on opposite sides. These drive the formation of highly reactive oxygen-based species in the water, which then attack and fragment the dye molecules. Scavenger tests—where specific reactive species are selectively blocked—showed that superoxide radicals (a reactive form of oxygen) play the leading role, with hydroxyl radicals playing a smaller part. Detailed chemical analysis of the treated water using gas chromatography–mass spectrometry revealed a sequence of intermediate molecules shrinking step by step into smaller, less harmful fragments, and finally simple acids close to full mineralization. 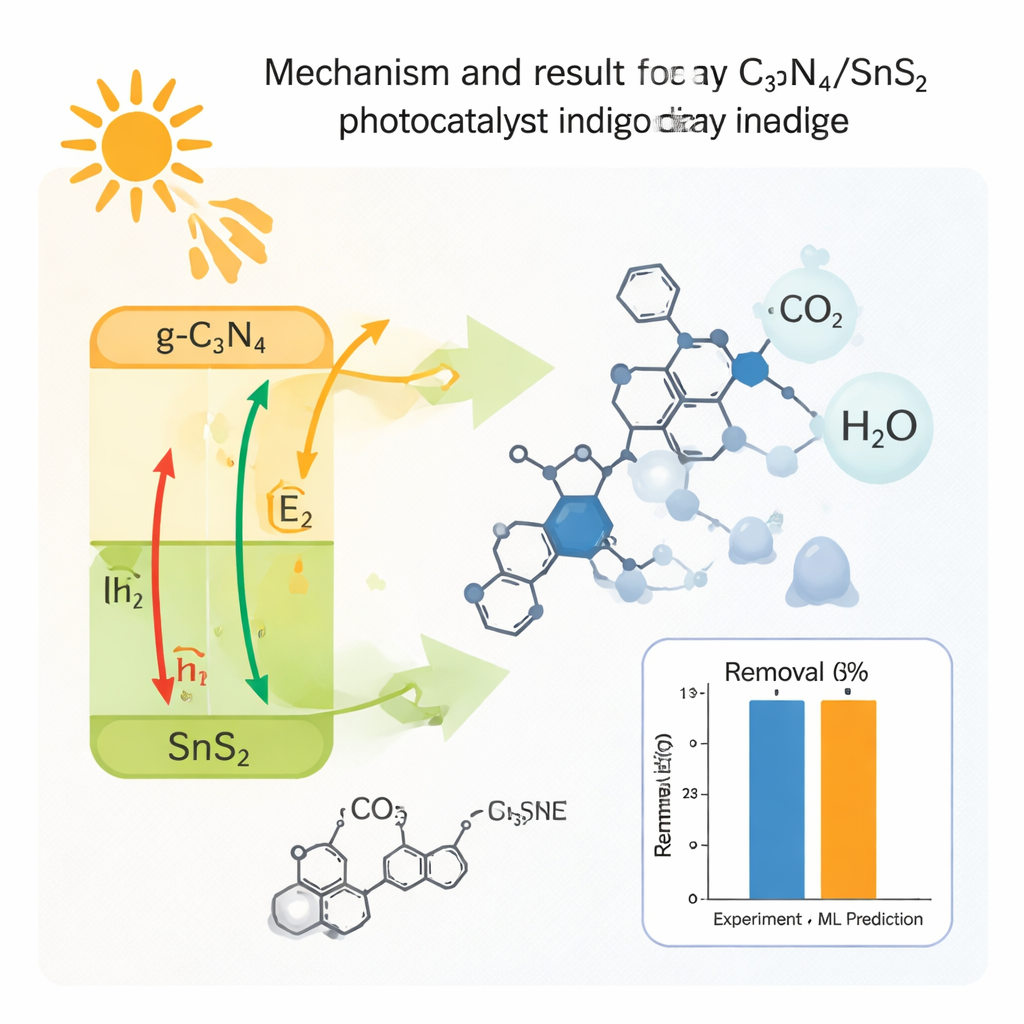
Letting Algorithms Predict Performance
To bridge laboratory findings with practical use, the team trained several machine learning models on their experimental data. These models took in variables such as exposure time and dye concentration and learned to predict how much dye would be removed under each condition. Among the tested approaches—Random Forest, support vector machines, neural networks, and gradient boosting—Random Forest delivered the most accurate and stable predictions, closely matching the measured removal efficiencies. This means that, once trained, such models can quickly forecast how well the catalyst would work in new scenarios without having to run countless new experiments, guiding engineers toward optimal treatment conditions.
What This Means for Cleaner Water
For non-specialists, the message is straightforward: this work demonstrates a low-cost, sunlight‑driven material that can rapidly clean up a tough industrial dye and largely destroy it rather than just hiding it. The catalyst is simple to make, needs only small amounts to work, and can be reused multiple times, making it attractive for real wastewater treatment. By pairing careful experiments with modern machine learning tools, the study also shows how we can design and fine‑tune future water-cleaning technologies more intelligently, speeding up the path from lab discovery to practical, cleaner rivers and lakes.
Citation: Gaur, R., Parmar, H., Patel, J. et al. Machine learning-driven performance prediction of Z-scheme g-C3N4/SnS2 heterostructure photocatalyst for complete mineralization of indigo carmine and elucidation of degradation pathways. Sci Rep 16, 6403 (2026). https://doi.org/10.1038/s41598-026-37528-5
Keywords: water purification, photocatalysis, wastewater treatment, machine learning, industrial dyes