Clear Sky Science · en
Interaction of pulsed low frequency electromagnetic field (PEMF) with mitochondria
Gentle Fields and the Cell’s Powerhouses
Pulsed electromagnetic field (PEMF) therapy is already used in clinics to help mend broken bones, ease joint pain, and speed recovery after surgery. Yet we still do not fully understand how these gentle, low‑frequency fields act inside our bodies. This study asks a basic but important question: do PEMF signals directly influence mitochondria, the tiny power plants inside our cells that generate most of our energy, and if so, how?
Why Mitochondria Matter for Everyday Health
Mitochondria turn food and oxygen into the molecule ATP, the universal “energy currency” of cells. When mitochondria falter, tissues heal more slowly, muscles tire more quickly, and diseases linked to poor metabolism can worsen. Because previous research hinted that PEMF treatments improve energy metabolism and support tissue repair, the authors focused on whether a specific PEMF device could fine‑tune mitochondrial activity. They used a well‑defined magnetic signal: short, millisecond‑long pulses of a 30 kHz sine wave with low input energy, applied to muscle cells in culture, to rat tissues, and to purified mitochondria.
Taking a Closer Look Inside Living Cells
In human muscle cells grown in the lab, the team measured three key indicators after PEMF exposure: the electrical potential across the mitochondrial inner membrane (a kind of tiny battery), levels of reactive oxygen species (high levels can be damaging), and nitric oxide, a gas that can temporarily block mitochondrial respiration. Around 90 minutes after treatment, the mitochondrial membrane potential was modestly lower, while nitric oxide levels were reduced; reactive oxygen species did not increase. A small drop in membrane potential can signal either damage or harder‑working ATP production. Because the cells did not show signs of stress, the authors suspected that mitochondria were simply running a bit faster and using up the voltage stored across their inner membrane.
Testing How Mitochondria Breathe
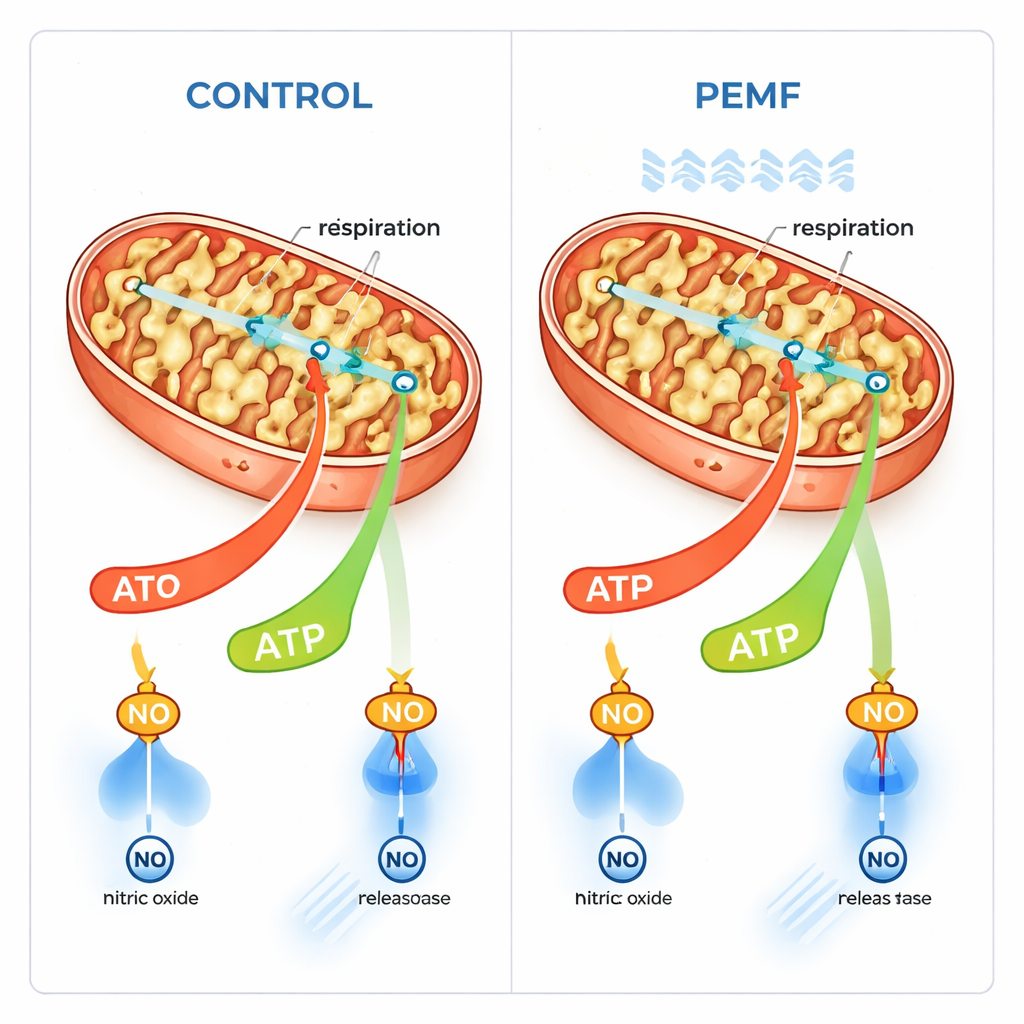
To test this idea more directly, the researchers gently perforated the outer cell membrane so that they could precisely feed different fuels to the mitochondria and measure oxygen use. They consistently saw a trend toward higher respiration linked to ATP production after PEMF, especially when glutamate, a common mitochondrial fuel, was present. Statistical analysis across many samples suggested that the combination of PEMF exposure and the choice of fuel significantly shaped how fast mitochondria consumed oxygen. Importantly, there was no sign that PEMF harmed the cells or their mitochondria. Instead, the fields appeared to selectively stimulate the “coupled” mode of respiration, where oxygen use is tightly tied to making ATP, without boosting the wasteful, uncoupled burning of fuel.
Inflammation, Nitric Oxide, and a Surprise from Blue Light
Because nitric oxide can build up during inflammation and block a key step in mitochondrial breathing, the team asked whether PEMF could help mitochondria recover from such inhibition. They added a chemical nitric oxide donor to muscle and liver tissue homogenates and to isolated liver mitochondria, strongly slowing respiration. Under these conditions, PEMF did not restore mitochondrial function, even when mitochondria were highly concentrated. To see whether the block was reversible at all, they shone blue light on the nitric‑oxide‑inhibited mitochondria, using a method known to dislodge nitric oxide from its binding site. Blue light partially rescued respiration, confirming that the system could recover, but PEMF did not add any further benefit. This showed that the positive effects of PEMF do not come from removing nitric oxide during inflammatory‑like conditions.
Membrane Gates and Energy Flow
Drawing an analogy between cells and electrical circuits, the authors suggest that PEMF may act mainly on biological membranes. Mitochondria are wrapped in an outer membrane that contains a key gateway protein called VDAC, which allows energy‑related molecules to pass in and out. Unlike the inner membrane’s very high voltage, the outer membrane has a smaller, more easily influenced voltage. The authors propose that PEMF subtly alters this outer membrane potential, shifting how often VDAC gates are open and thereby easing the flow of fuel and spent ATP across the membrane. This would naturally boost the coupled, ATP‑making mode of respiration without changing the maximum capacity of the electron transport machinery itself.
What This Means for Future Therapies
Overall, the study finds no evidence that this type of PEMF is harmful to mitochondria. On the contrary, PEMF gently enhances the part of mitochondrial respiration that produces ATP, while leaving maximum respiratory capacity and oxidative stress unchanged. However, it does not rescue mitochondria that are blocked by high nitric oxide, suggesting that its benefits in inflamed tissues must arise through other routes. For patients and clinicians, these results support the idea that carefully tuned electromagnetic fields can nudge the cell’s powerhouses into a more efficient, energy‑producing state—offering a plausible, mechanistic explanation for some of the healing effects reported in bone repair, wound care, and post‑surgical recovery.
Citation: Zavadskis, S., Gasser, A.S., Karas, M. et al. Interaction of pulsed low frequency electromagnetic field (PEMF) with mitochondria. Sci Rep 16, 6681 (2026). https://doi.org/10.1038/s41598-026-37527-6
Keywords: pulsed electromagnetic field therapy, mitochondria, cellular energy, nitric oxide, ATP production