Clear Sky Science · en
Cafestol-derivatives as potential FXR agonists and CYP7A1 inhibitors and their impact on hypercholesterolemia: an in silico study
Why Your Morning Coffee Might Nudge Your Cholesterol
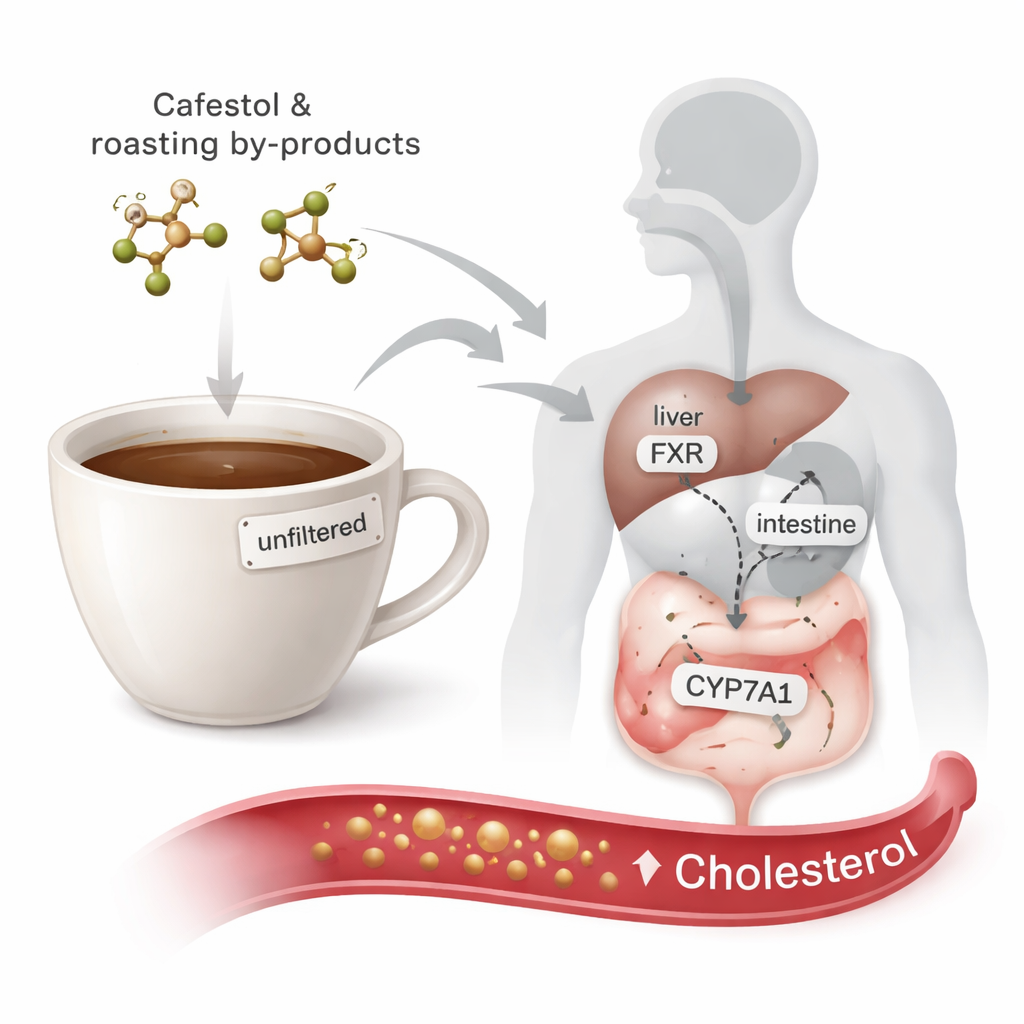
Coffee lovers often hear that unfiltered brews like French press or boiled coffee can raise cholesterol, but the reasons usually stay hidden in technical jargon. This study digs into the chemistry behind that warning. It looks not only at cafestol—a natural substance in coffee already known to raise cholesterol—but also at what happens to cafestol when coffee beans are roasted and when the body starts to break it down. Using powerful computer simulations, the researchers ask a simple, practical question: could these related compounds also be quietly pushing cholesterol levels upward?
Coffee Oils and the Cholesterol Question
Cafestol is a fat-like molecule found in the oily fraction of coffee beans, especially in unfiltered coffee drinks. While cafestol has been linked to some potential health benefits, such as anti-cancer and anti-diabetes effects in laboratory studies, human trials have consistently shown that it can raise blood cholesterol. Just 10 milligrams a day for a few weeks is enough to measurably increase cholesterol in volunteers. Roasting and brewing methods matter: espresso, moka, French press, and boiled coffee retain more of these oily compounds than paper-filtered coffee. Yet, until now, almost nothing was known about whether the roasting products and early metabolites of cafestol might share its cholesterol-raising potential.
Two Molecular Switches That Control Cholesterol
The research focuses on two key proteins that help control how the body handles cholesterol: the farnesoid X receptor (FXR) and an enzyme called CYP7A1. FXR acts as a sensor for bile acids—compounds made from cholesterol in the liver—and helps regulate both bile acid production and fat balance. When FXR is switched on, it sends signals that can ultimately reduce the activity of CYP7A1. CYP7A1, in turn, performs the first and rate-limiting step in converting cholesterol into bile acids for excretion. If FXR is overactivated or CYP7A1 is directly blocked, the body may convert less cholesterol into bile acids, potentially leaving more cholesterol circulating in the blood. Cafestol is already known to act on these switches, but the behavior of its roasting products and early metabolites had not been mapped.
Testing Coffee Compounds in a Virtual Laboratory
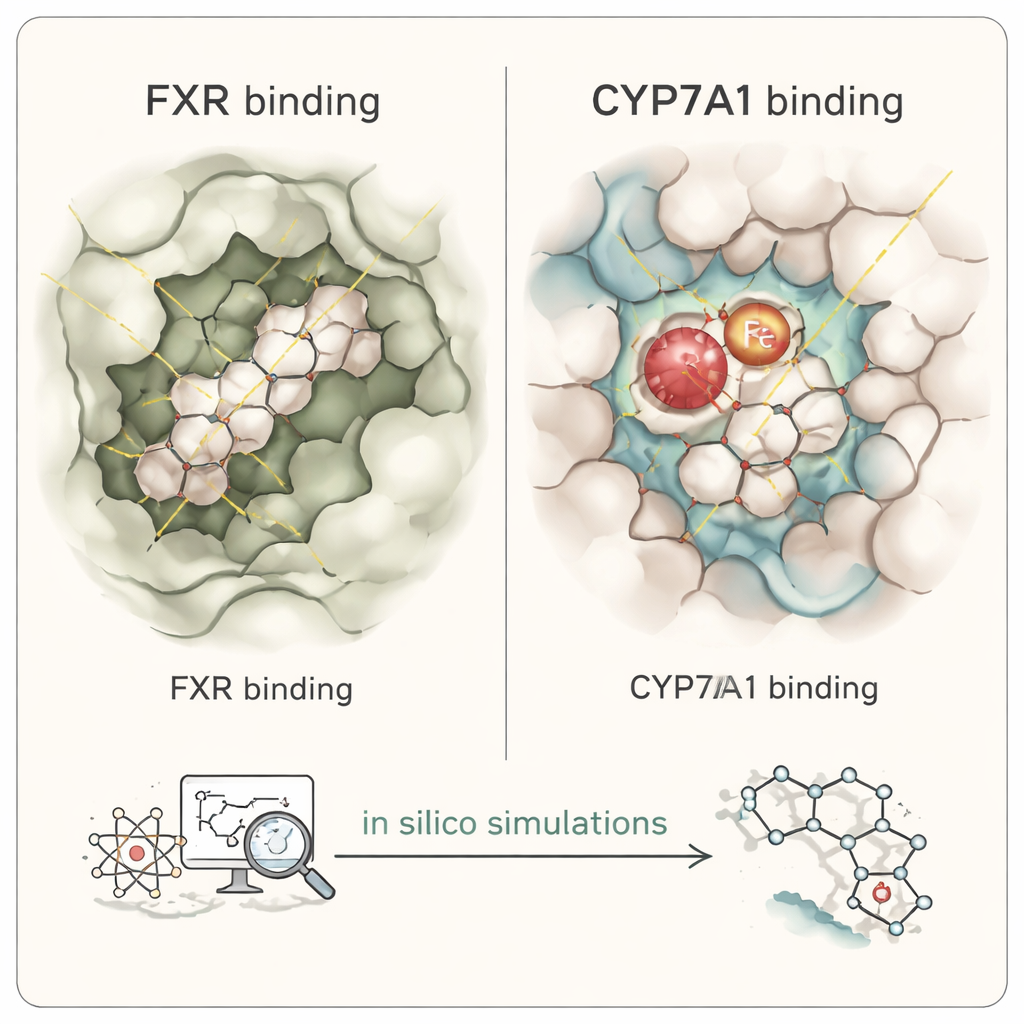
Because many of these cafestol-related molecules are difficult to isolate and test experimentally, the team turned to in silico methods—computer-based experiments. They built three-dimensional models of human FXR and CYP7A1 and then simulated how cafestol, its roasting derivatives, and its phase I metabolites might fit into these proteins, much like trying different keys in a lock. This involved molecular docking, which predicts how tightly and in what orientation each molecule can bind, and molecular dynamics simulations, which watch these complexes “wiggle” and stabilize over time in a virtual watery environment. They also created pharmacophore maps, abstract patterns that highlight which features—such as oily regions and hydrogen-bonding groups—are most important for binding, and ran absorption and metabolism predictions to estimate how the roasting products might behave in the body.
Roasting Products That Still Look Like Trouble
The simulations suggest that several cafestol derivatives formed during roasting, as well as some early breakdown products formed in a zebrafish model, can latch onto FXR and CYP7A1 in ways that resemble known steroid-based drugs and inhibitors. The core ring system of these coffee molecules mimics the rigid steroid backbone of natural bile acids and oxysterols, allowing them to sit snugly in the same pockets as the body’s own signaling compounds. In FXR, cafestol and its close relatives showed stable, predominantly oily (hydrophobic) contacts in the main binding site, with predicted binding strengths on par with each other, though weaker than a potent pharmaceutical FXR agonist. In CYP7A1, many of the roasting derivatives placed their furan ring near the enzyme’s heme center—the metal-containing core that performs chemistry—mirroring how a known cholesterol-derived inhibitor binds. Some metabolites lost this critical contact, hinting they may be less able to block the enzyme directly.
What This Means for Coffee Drinkers
To a layperson, the message is not that coffee is unsafe but that certain styles and doses deserve respect. This work supports the idea that cafestol is not acting alone: closely related molecules created during roasting and early metabolism may also influence the same protein switches that control how rapidly the body disposes of cholesterol. Because these conclusions are based on computer models rather than human trials, they are best viewed as an early warning flag, not a final verdict. Still, they underscore why unfiltered, high-oil coffee—French press, boiled coffee, or very strong espresso—can contribute to higher cholesterol in some people, and why moderation and brewing method matter when balancing the pleasures of coffee with long-term heart health.
Citation: da Silva, M.A.E., Camargo, P.G., da Silva Lima, C.H. et al. Cafestol-derivatives as potential FXR agonists and CYP7A1 inhibitors and their impact on hypercholesterolemia: an in silico study. Sci Rep 16, 7102 (2026). https://doi.org/10.1038/s41598-026-37519-6
Keywords: coffee diterpenes, cafestol, unfiltered coffee, cholesterol metabolism, FXR and CYP7A1