Clear Sky Science · en
“Sonochemically synthesized Ag(I) and Ni(II) schiff base complexes as efficient visible-light photocatalysts for dye degradation with DFT insights.”
Turning Colored Waste into Clear Water
From the clothes we wear to the food we eat, modern life depends heavily on synthetic dyes. But these bright colors leave a dark mark: dye-filled wastewater that is hard to clean and harmful to rivers, lakes, and the creatures that live in them. This study explores a new way to use light and tiny metal-based particles to break down a common blue dye in water, pointing toward cheaper, greener methods for cleaning industrial wastewater.
Why Dye Pollution Matters
Textile and other industries release large amounts of leftover dyes into water systems. These dyes block sunlight, lower oxygen levels, and are linked to serious health problems, including genetic damage. Traditional cleanup methods—such as filtering, adding chemicals, or burning off pollutants—can be costly, complex, or create new waste. A promising alternative is photocatalysis, where a solid material uses light to trigger chemical reactions that break pollutants down into harmless substances like carbon dioxide and water, without the need for extra chemicals.
Building Tiny Light-Driven Cleaners
The researchers created two new photocatalysts based on so‑called Schiff base molecules made from isatin and the sulfa drug sulfathiazole. These organic building blocks were linked to silver (Ag) or nickel (Ni) ions to form metal complexes. Importantly, they used an eco‑friendly sonochemical method, in which sound waves help drive the reaction in solution, to produce nanoscale particles—extremely small grains with a large surface area that can interact efficiently with dye molecules in water. A wide suite of techniques, including infrared and ultraviolet–visible spectroscopy, nuclear magnetic resonance, X‑ray diffraction, and thermal analysis, was used to confirm the structure, stability, and nanoscale size of the resulting silver and nickel complexes.
Seeing How They Respond to Light
To understand how these new materials interact with light and electrons, the team combined experiments with computer simulations based on density functional theory (DFT). Optical measurements showed that both complexes behave like semiconductors: their electrons can be excited by visible light across relatively small energy gaps. The DFT calculations supported this picture, revealing that attaching silver or nickel to the Schiff base shrinks the gap between the highest occupied and lowest unoccupied molecular orbitals, making it easier for light to create mobile electrons and “holes.” The simulations also mapped out regions of negative and positive charge across the molecules, helping to identify where dye molecules and reactive species are likely to bind on the catalyst surface.
Putting the Catalysts to the Test
The real test was whether these nanomaterials could actually destroy dye in water. The team chose Methylene Blue (MB), a widely used blue dye, and shone visible light from an ordinary 60‑watt tungsten lamp on dye solutions containing different amounts of the silver or nickel complexes. They varied three key conditions: how much catalyst was added, how concentrated the dye solution was, and the acidity or alkalinity (pH) of the water. Under the best conditions—moderately alkaline water at pH 11, 30 mg of catalyst in 100 mL of a 10‑parts‑per‑million MB solution—both materials performed impressively. The silver complex removed about 95.3% of the dye and the nickel complex about 91.7% within 100 minutes. The reaction followed so‑called pseudo‑first‑order kinetics, meaning the rate depended mainly on how much dye remained, and both catalysts could be recovered and reused at least four times with only a small drop in efficiency.
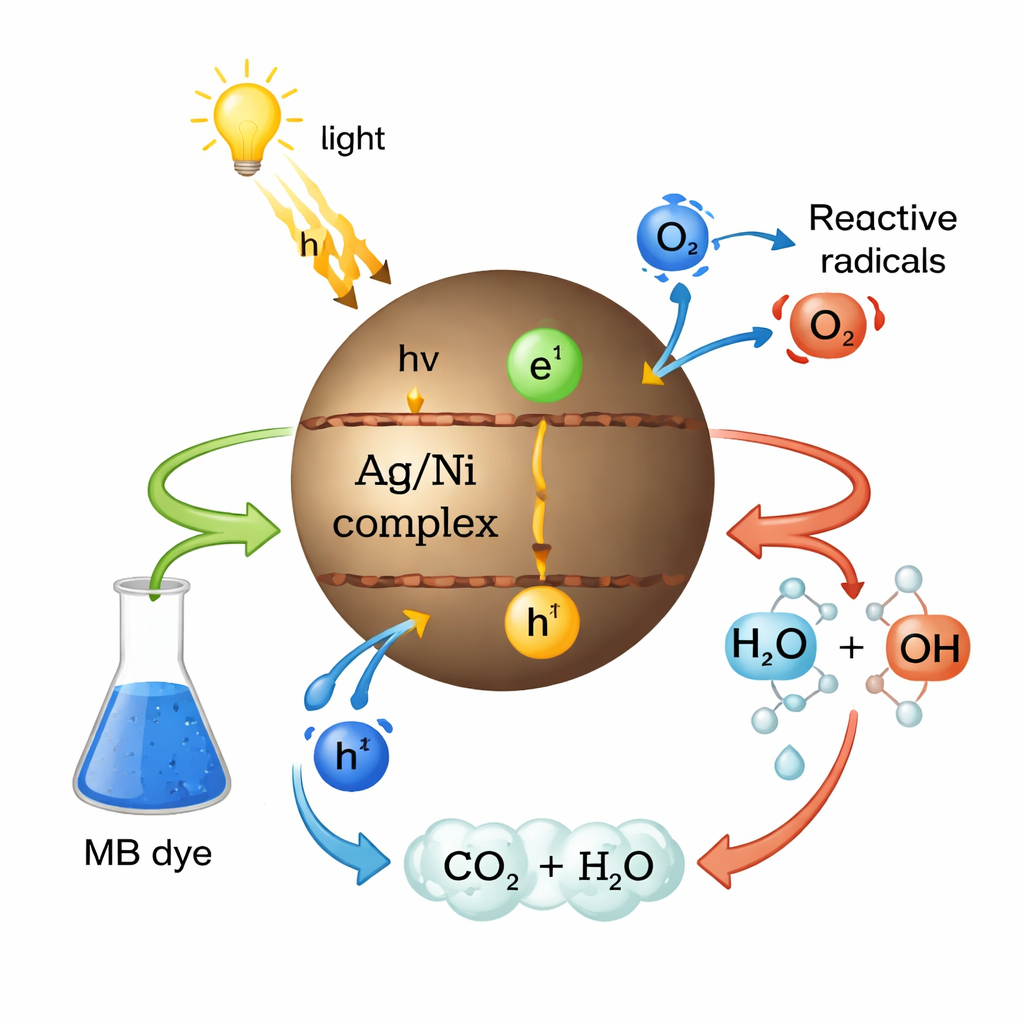
How the Breakdown Happens
The study outlines a step‑by‑step picture of how the dye is destroyed. When visible light hits the catalyst particles, electrons are pushed to higher energy levels, leaving behind positively charged “holes.” These electrons react with dissolved oxygen to form reactive oxygen species, while the holes react with water to form highly reactive hydroxyl radicals. These short‑lived radicals attack the dye molecules at many points, slicing their chemical bonds until they are fully broken down into carbon dioxide and water. The DFT results help explain why the silver complex performs slightly better: its smaller energy gap and favorable charge distribution allow it to absorb light more effectively and interact strongly with the positively charged dye.
What This Means for Cleaner Water
For a non‑specialist, the bottom line is that the researchers have demonstrated two new, stable, and reusable light‑activated materials that can almost completely remove a stubborn blue dye from water using only visible light and modest catalyst amounts, without added oxidizing chemicals. Because the particles are made by a relatively green, sound‑assisted process and can be recycled several times, they offer a promising route toward practical photocatalysts for treating dye‑contaminated wastewater. Further work will be needed to test them on real industrial effluents and other pollutants, but this study shows how smart molecular design, guided by theory, can turn everyday light into a powerful tool for cleaning our water.
Citation: Saleh, A.M., Mahdy, A.G. & Hamed, A.A. “Sonochemically synthesized Ag(I) and Ni(II) schiff base complexes as efficient visible-light photocatalysts for dye degradation with DFT insights.”. Sci Rep 16, 7181 (2026). https://doi.org/10.1038/s41598-026-37498-8
Keywords: photocatalysis, wastewater treatment, methylene blue, silver and nickel complexes, Schiff base nanomaterials