Clear Sky Science · en
The metabolic dysfunction-associated steatohepatitis (MASH) drug resmetirom exhibits broad nuclear receptor activity with minimal functional impact
Why a new liver drug matters
Millions of people worldwide live with a dangerous form of fatty liver disease now called metabolic dysfunction–associated steatohepatitis, or MASH. It can quietly damage the liver for years and eventually lead to scarring, liver failure, or cancer. In 2024, a drug named resmetirom became the first medicine approved in the United States specifically for MASH. This study asks a crucial question for patients and doctors: does resmetirom act cleanly on its intended target in the liver, or does it also hit other molecular switches that might blunt its benefits or cause side effects?
A targeted way to burn liver fat
Resmetirom was designed to work like a focused version of natural thyroid hormone. Thyroid hormones control how fast our cells use energy, affecting heart rate, body temperature, and how fat and sugar are processed. They act through two related receptors, called alpha and beta. The beta form is most abundant in the liver, while the alpha form dominates in the heart and bone. In people with MASH, thyroid hormone signaling inside the liver is often sluggish, even when blood tests look normal. Resmetirom aims to "reawaken" the beta receptor in liver cells to burn more fat, break down cholesterol, and reduce fat buildup, while sparing the alpha receptor to avoid heart and bone side effects. 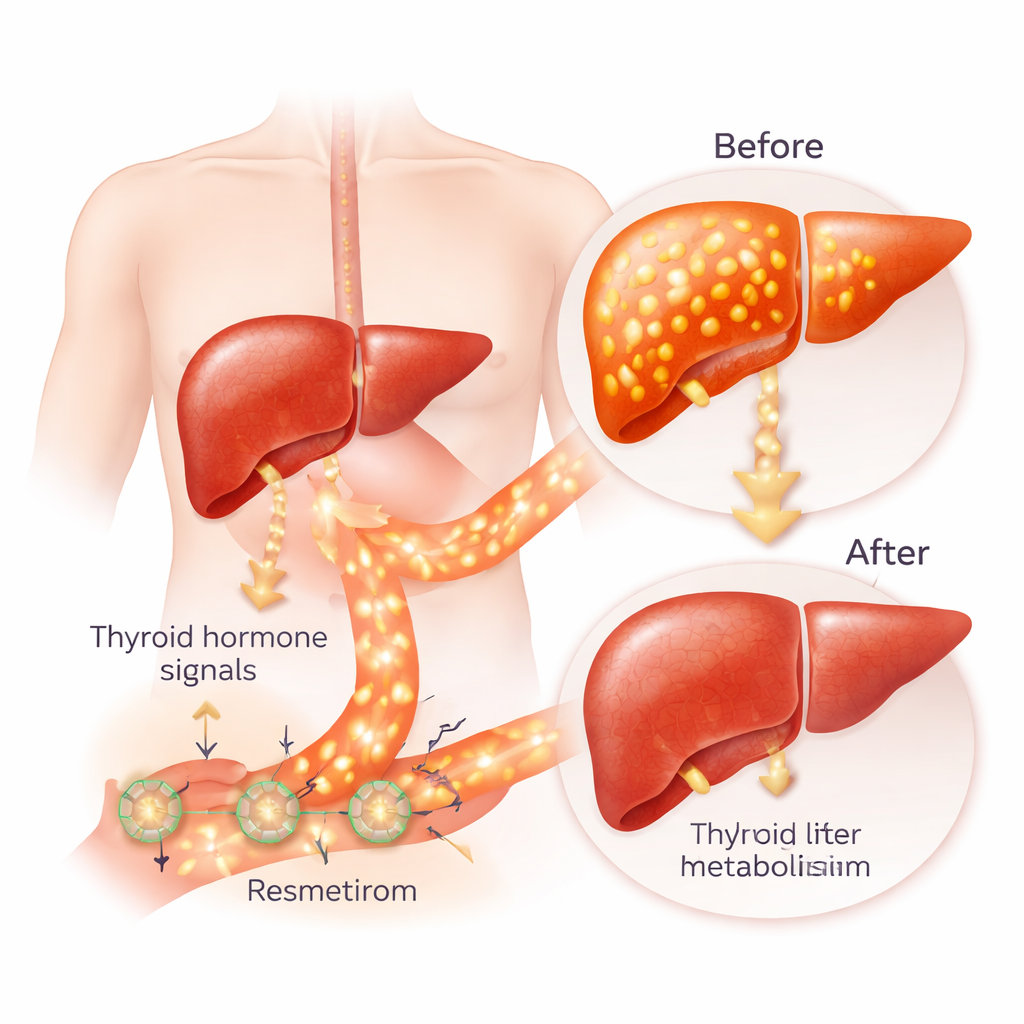
Checking for unwanted molecular targets
Because resmetirom must be given at relatively high doses to do its job, the authors wondered whether it might also latch onto other nuclear receptors—proteins in the cell nucleus that act as master switches for metabolism, detoxification, and daily body rhythms. Using computer modeling and a series of cell-based reporter tests, they first checked if resmetirom behaved like certain thyroid hormone breakdown products known to activate another receptor called PPARγ. The modeling suggested that, despite similar shapes, resmetirom would fit poorly into PPARγ’s binding pocket, and the cell tests confirmed that resmetirom did not turn this receptor on.
Off-target hits, but muted effects
The researchers then screened resmetirom against a broader panel of nuclear receptors involved in lipid and glucose control. As expected, resmetirom activated the thyroid hormone beta receptor more strongly than the alpha version. However, it also interacted with several other receptors: it blocked a receptor called CAR, reduced the baseline activity of three ROR receptors, and modestly dampened a liver-protective factor known as HNF4α. On paper, these off-target actions could be worrying, because all of these switches help regulate liver metabolism and detoxification. Yet the strength of these effects was only moderate, and often partial, suggesting that resmetirom nudges these receptors rather than fully flipping them.
Putting the drug into more realistic liver cells
To see whether these off-target interactions matter under more life-like conditions, the team turned to human liver cancer cells commonly used as a stand-in for liver tissue. They measured the activity of key genes that are known to respond to thyroid hormone as well as to CAR, RORs, and HNF4α. Resmetirom boosted genes involved in glucose production and detoxification in a pattern matching thyroid hormone’s action, even though its off-target actions should have pushed some of those genes in the opposite direction. When the scientists triggered fat production in these cells, resmetirom and natural thyroid hormone both clearly reduced fat buildup, while drugs that specifically targeted ROR, CAR, or HNF4α alone had little effect. 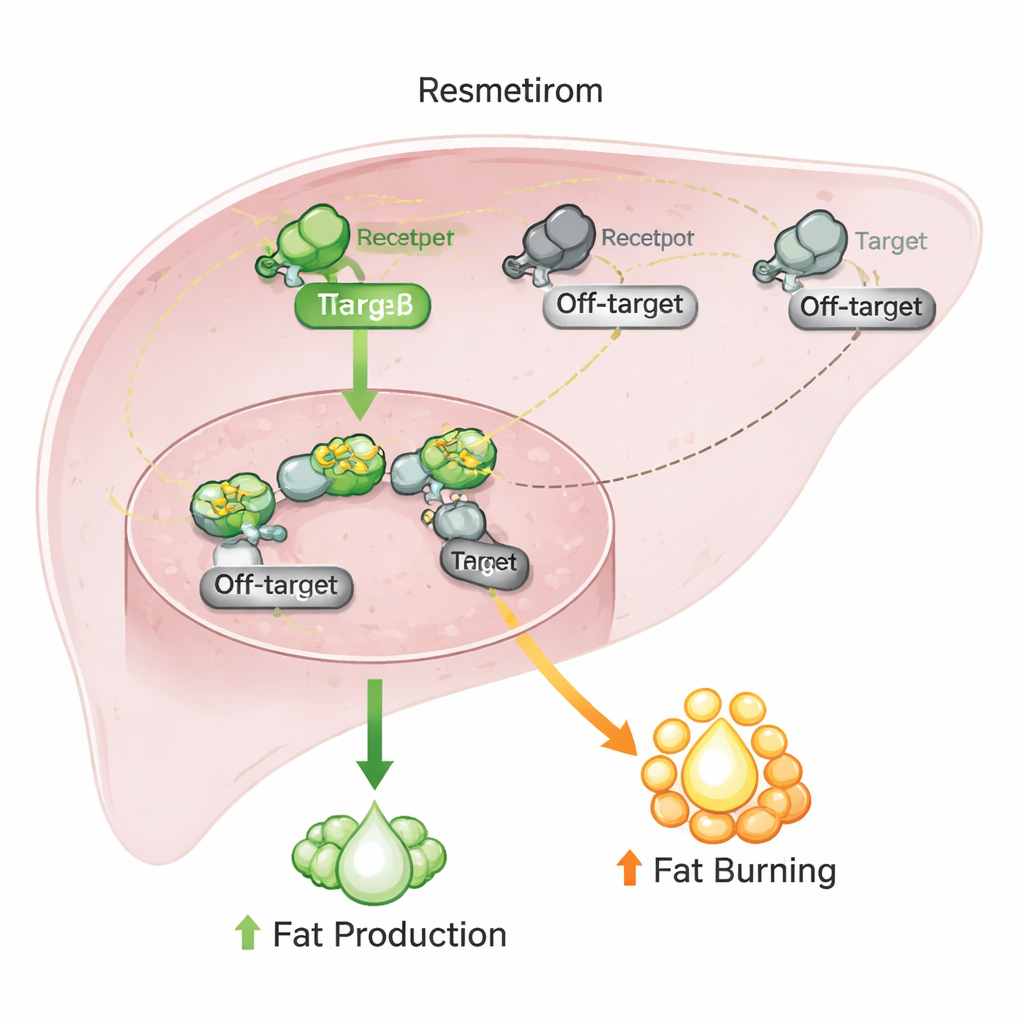
What this means for patients and future drugs
Taken together, the findings suggest that resmetirom does indeed brush against several other nuclear receptors, but its main effect in liver cells is still driven by the intended thyroid hormone beta receptor. In practical terms, that means the drug’s ability to lower liver fat and improve liver health in MASH is unlikely to be undermined by these side activities, at least in the experimental systems studied. The work also underscores a broader lesson: because resmetirom is only moderately potent, it needs fairly high concentrations to work, increasing the chance of subtle off-target effects elsewhere in the body. That makes it all the more important to develop future thyroid hormone–based medicines that grip the beta receptor more tightly and more selectively, delivering the same or better liver benefits with lower doses and even fewer unintended molecular interactions.
Citation: Kärcher, A., Isigkeit, L., Bandomir, N.C. et al. The metabolic dysfunction-associated steatohepatitis (MASH) drug resmetirom exhibits broad nuclear receptor activity with minimal functional impact. Sci Rep 16, 4083 (2026). https://doi.org/10.1038/s41598-026-37494-y
Keywords: fatty liver disease, MASH, resmetirom, thyroid hormone receptor, liver metabolism