Clear Sky Science · en
Liquid biopsy of plasma and urinary CfDNA differentiates glioma recurrence from radiation brain necrosis in preclinical models
Why a Simple Blood and Urine Test Could Help Brain Tumor Patients
For people treated for brain tumors, one of the scariest questions after radiation is whether a new spot on a scan means the cancer is back or the brain is simply scarred by the treatment itself. These two possibilities look almost identical on standard MRI images but call for very different decisions: more aggressive therapy versus careful observation. This study in rats explores whether fragments of DNA that circulate in blood and urine could offer a simple, non‑invasive way to tell the difference, potentially sparing patients risky brain biopsies and helping doctors choose the right next step.
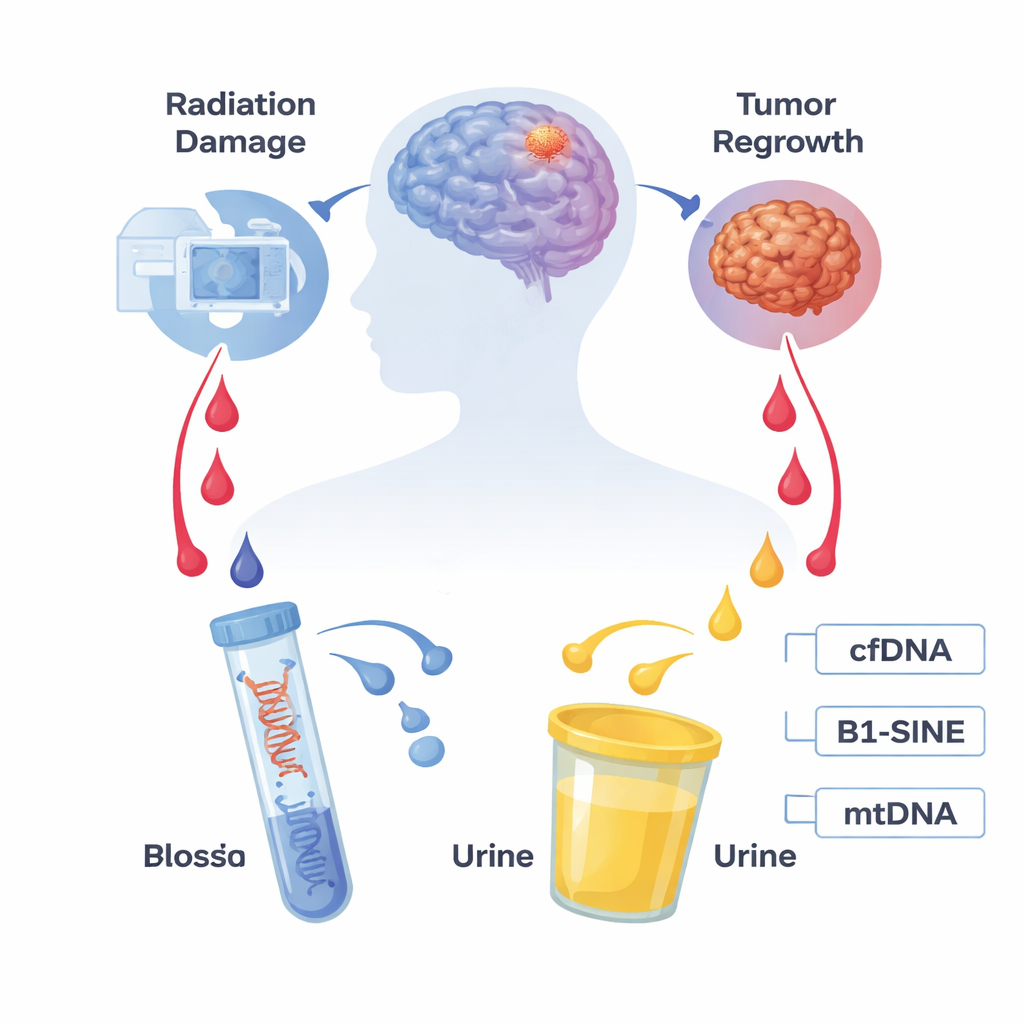
The Challenge of Reading Brain Scans After Treatment
Gliomas are among the deadliest brain tumors, with high recurrence rates even after surgery, radiation, and chemotherapy. When a treated area lights up again on MRI, it may signal either tumor regrowth or radiation brain necrosis, a delayed form of tissue death caused by high‑dose radiation. Both conditions can cause swelling, neurological symptoms, and similar scan patterns. Today, doctors often rely on long‑term imaging follow‑up or invasive surgical biopsies to figure out which process is occurring. These approaches are slow, risky, and sometimes inconclusive, creating an urgent need for a safer, earlier, and more reliable test.
Looking for Clues in Free‑Floating DNA
Our cells constantly shed tiny pieces of genetic material, called cell‑free DNA, into bodily fluids such as blood and urine. Tumors and damaged tissues release even more of these fragments. The researchers focused on two particular signals: B1‑SINE, a common repetitive DNA sequence that reflects overall cell‑free DNA in rodent blood, and mitochondrial DNA (mtDNA), which comes from the cell’s energy factories and can leak out when cells are stressed or die. Because blood draws and urine collection are far easier and safer than brain biopsies, these markers are promising candidates for a "liquid biopsy"—a test that reads molecular information from fluids instead of tissue.
Building Rat Models to Imitate Human Disease
To test this idea under controlled conditions, the team created two separate rat models. In one group, they implanted glioma cells into the brain, allowed a tumor to form, surgically removed it, and then followed the animals until the tumor grew back—mimicking human glioma recurrence. In another group, they used carefully planned, high‑dose radiation aimed at one side of the brain to trigger delayed radiation necrosis, mirroring a serious complication seen in patients. Throughout the following weeks, they collected blood and urine, measured B1‑SINE and mtDNA levels using a sensitive DNA amplification technique, and confirmed brain changes with MRI scans and tissue analysis under the microscope.
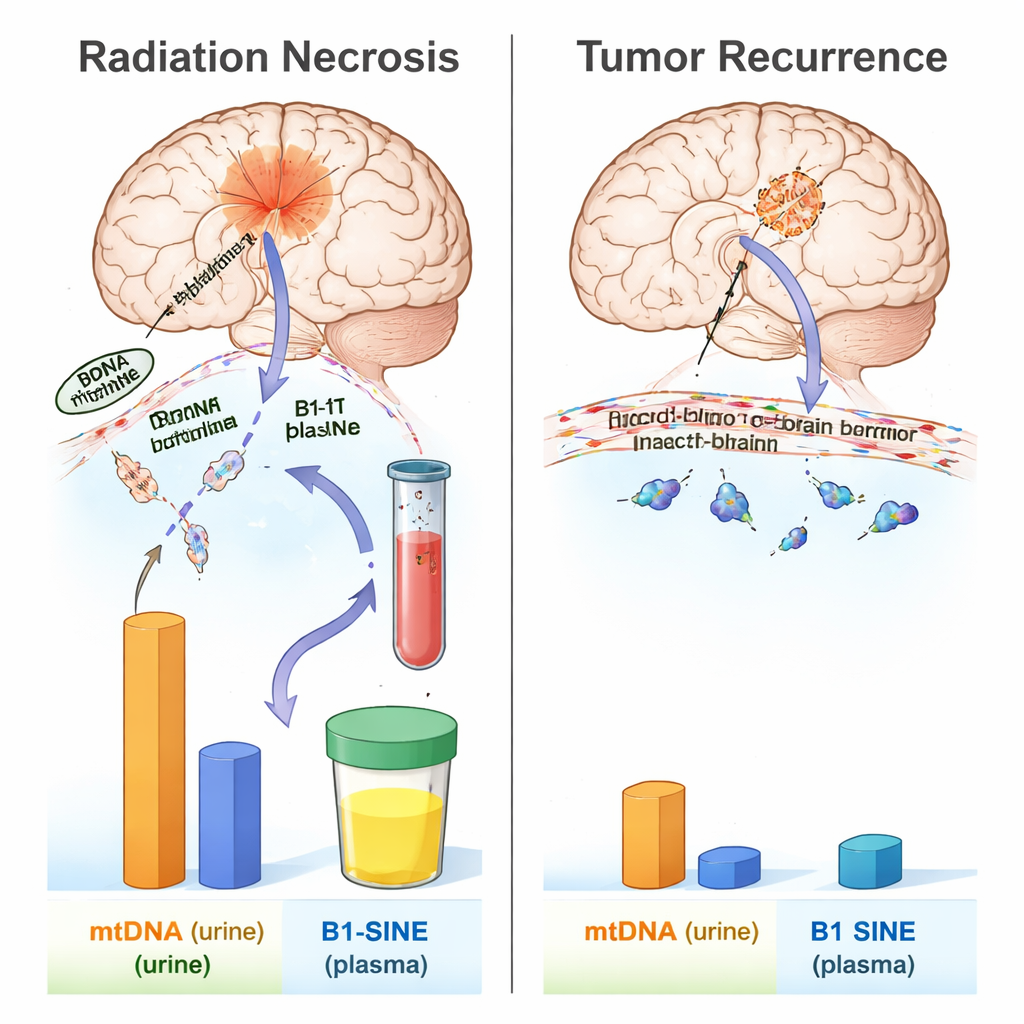
Distinct DNA Signatures for Radiation Damage
The patterns of DNA fragments in fluids turned out to be strikingly different between the two conditions. After radiation, urinary mtDNA jumped within 24 hours, briefly returned toward normal, and then rose again around week six, peaking at week eight and staying elevated as radiation necrosis developed. Plasma B1‑SINE levels also surged after radiation, with a clear peak at week eight and persistently high values from weeks eight to sixteen, the window when tissue death was evident in brain scans and pathology. In contrast, rats with recurring glioma showed no significant changes in either urinary mtDNA or plasma B1‑SINE compared with healthy and sham‑surgery controls over the same time frame. This suggests that radiation‑damaged brain tissue releases a characteristic wave of DNA fragments into the circulation that is not mimicked by tumor regrowth in this model.
What This Could Mean for Future Patients
To a layperson, the main message is that simple blood and urine tests may one day help doctors distinguish treatment‑related brain injury from returning cancer without opening the skull. In these rat experiments, two signals—mtDNA in urine and B1‑SINE in blood—rose sharply only when radiation necrosis was present, not when the tumor came back. Although more work is needed in larger studies and in human patients, and the exact thresholds and human‑specific markers still must be defined, this research points toward a future where a "liquid snapshot" of circulating DNA can guide brain tumor follow‑up, reduce uncertainty, and support more personalized, less invasive care.
Citation: Sun, J., Jin, C., Chen, Y. et al. Liquid biopsy of plasma and urinary CfDNA differentiates glioma recurrence from radiation brain necrosis in preclinical models. Sci Rep 16, 7123 (2026). https://doi.org/10.1038/s41598-026-37493-z
Keywords: glioma, radiation necrosis, liquid biopsy, cell-free DNA, brain tumor monitoring