Clear Sky Science · en
Exceptionally selective voltage-sensor trapping of NaV1.5 channels by Mg-protoporphyrin impairs cancer cell migration
Plant pigment points to a new cancer strategy
Our hearts and our tumors may share an unexpected vulnerability: a special kind of electrical gate in the cell membrane called NaV1.5. This study shows that a natural cousin of the chlorophyll building blocks in plants, magnesium‑protoporphyrin IX (MgPpIX), can shut down this gate with extraordinary precision. By doing so, it sharply slows the movement of certain cancer cells—hinting that a molecule inspired by photosynthesis chemistry could help tame metastasis without heavily disturbing the brain or muscles.
A hidden electrical switch in cancer cells
Cells use voltage-gated sodium channels as tiny doors that open and close to let charged particles pass, generating electrical signals. NaV1.5 is best known for its role in heartbeats, where it helps trigger each contraction. But the same channel is also found in several cancers, including breast and colon tumors, where its activity has been linked to increased cell movement and invasiveness. Targeting NaV1.5 has been difficult because most sodium-channel drugs also hit related channels in nerves and skeletal muscle, risking side effects such as seizures, numbness, or muscle weakness. The authors set out to discover whether any small molecules could distinguish NaV1.5 from its relatives with much higher selectivity.
A plant-inspired blocker with record precision
The team examined a family of ring-shaped molecules called metal protoporphyrins, which resemble the cores of heme in blood and chlorophyll in plants but contain different central metal atoms. When they applied various versions to human NaV1.5 channels produced in cultured cells, one compound stood out: MgPpIX. It reduced the electrical current through NaV1.5 by about 99% at nanomolar concentrations, roughly 100 times more potent than an iron-based counterpart (hemin). Other metal versions, such as those containing nickel or copper, were essentially inactive. Remarkably, MgPpIX left several other human sodium channel types (used by the brain, peripheral nerves, and skeletal muscle) completely untouched at the same doses, making its selectivity among the sharpest ever reported for this channel family.
How the molecule jams a tiny voltage lever
To understand this unusual precision, the researchers combined genetic tinkering with computer simulations. Sodium channels have four repeating parts, each containing a “voltage sensor” that shifts position when the membrane potential changes. By swapping individual amino acids between NaV1.5 and its insensitive relatives, the team traced MgPpIX’s action to the voltage sensor in the second domain, especially two residues exposed to the outside of the cell. Molecular dynamics simulations in a realistic membrane environment suggested that MgPpIX nestles against this sensor when it is in its resting, “down” position. The positively charged metal center of MgPpIX is attracted to a negatively charged side chain on the channel, while the flat ring interacts with surrounding lipids. This binding appears to lock the voltage sensor in place, preventing it from moving into the active “up” state and thus keeping the channel closed. Interestingly, strong depolarizations—like those during a heartbeat—can temporarily dislodge the molecule, a behavior the authors describe as “reverse use-dependence.”
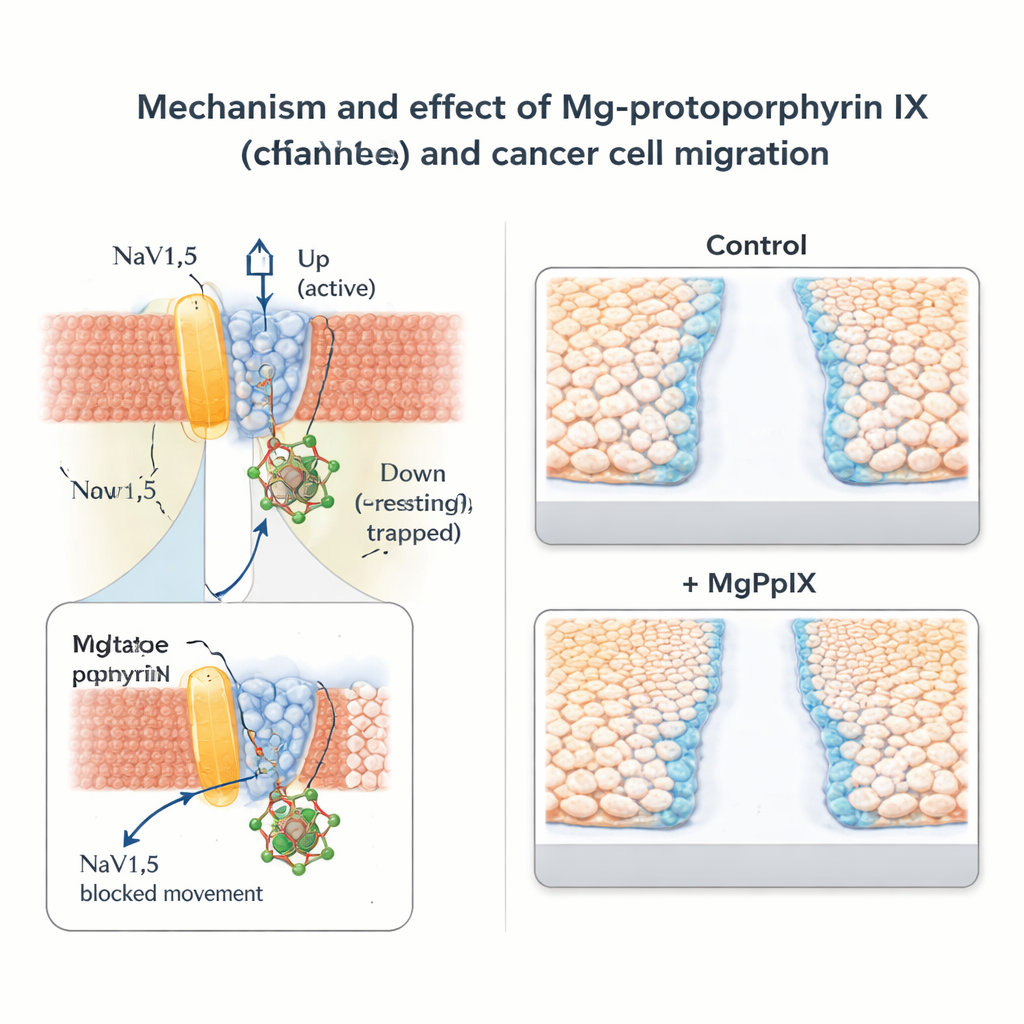
Slowing cancer cell movement without broad nerve block
The team then explored what this molecular brake means for cancer behavior. In breast cancer (MDA-MB-231) and colon cancer (SW-480) cell lines that naturally express NaV1.5, MgPpIX dramatically reduced sodium currents and slowed cell migration in standard “scratch” and transwell assays. The more a given metal protoporphyrin blocked NaV1.5, the more it hindered cell movement, strengthening the link between channel activity and motility. In contrast, cancer cell lines lacking NaV1.5 showed no response to MgPpIX either electrically or in migration tests, supporting the idea that the effect is highly specific rather than generally toxic. Compared with the classic sodium channel toxin tetrodotoxin, MgPpIX achieved stronger suppression of migration at far lower concentrations.
Promise and caution for future anti-metastatic drugs
For a non-specialist, the central message is that the researchers have found a plant-related molecule that acts like a precision clamp on a heart-type electrical gate hijacked by some cancers. By trapping the gate’s tiny voltage lever in its resting position, MgPpIX can strongly curb the mobility of cancer cells in the lab while sparing other sodium channels that are crucial for sensation and movement. Because the heart’s rapid electrical activity can partially relieve this block, there may even be a safety window where tumor cells are inhibited more than cardiac cells. While MgPpIX itself may not be ready as a medicine, its unique binding site and mechanism provide a powerful blueprint for designing future drugs aimed at stopping cancer spread by targeting NaV1.5 with unprecedented selectivity.
Citation: Jamili, M., Ahmed, M., Bernert, A. et al. Exceptionally selective voltage-sensor trapping of NaV1.5 channels by Mg-protoporphyrin impairs cancer cell migration. Sci Rep 16, 4085 (2026). https://doi.org/10.1038/s41598-026-37492-0
Keywords: sodium channels, Nav1.5, cancer cell migration, chlorophyll-derived molecules, targeted ion channel drugs