Clear Sky Science · en
Smart, automated junctional tourniquets leveraging AI-driven ultrasound guidance
Stopping Bleeding Where Tourniquets Fall Short
Uncontrolled bleeding is one of the most preventable causes of death after serious injuries, both on the battlefield and in civilian life. Ordinary tourniquets work well on arms and legs, but they struggle at the body’s “junctions” – places like the groin, armpit, and neck where major blood vessels dive into the torso. This study explores a new kind of smart tourniquet that uses ultrasound imaging and artificial intelligence (AI) to find and squeeze these deep vessels automatically, aiming to give first responders a faster and more reliable way to save lives when every second counts.
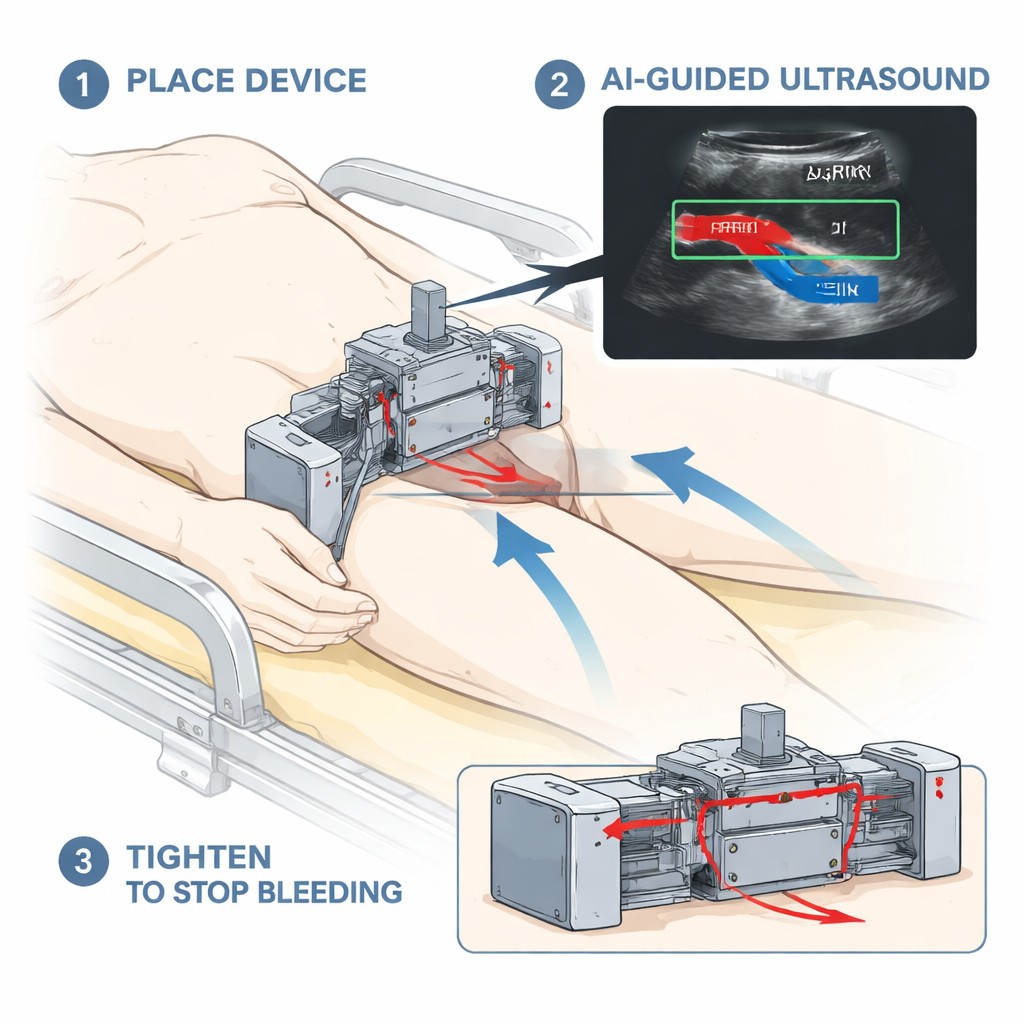
The Challenge of Hard-to-Reach Bleeding
Standard tourniquets are designed to wrap tightly around a limb, crushing the blood vessels against bone to stop blood loss. At junctions like the groin (femoral), under the collarbone (subclavian), or over the abdomen (aortic), the anatomy is more complex: vessels lie deeper, the surfaces are curved, and there is less structure to push against. Existing junctional tourniquets approved for medical use can help, but they are often bulky, slow to apply, and prone to slipping during movement. Studies show relatively high failure and malfunction rates, and using these devices correctly requires training and experience that might not be available in chaotic emergencies.
Bringing Ultrasound and AI to the Front Line
Ultrasound imaging can “see” beneath the skin in real time, showing where arteries, veins, and bones lie, and whether blood is still flowing. The catch is that interpreting these gray-scale images usually takes a skilled clinician. The authors set out to remove that barrier by pairing a portable, wireless ultrasound probe with AI models that automatically recognize key structures and determine when a vessel is effectively closed. Their vision is a device that a relatively inexperienced user could place in the general area of bleeding; the AI would then guide them to the right spot and signal when enough pressure has been applied to control the hemorrhage.
Building and Testing Smart Tourniquet Prototypes
The team created two mechanical prototypes. One, called the Frame Reinforced Junctional Tourniquet (FRejT), uses a rigid metal frame to position the ultrasound probe and a motorized actuator that presses straight down. The second, the Base and Tightening Straps (BaTS) design, uses adjustable straps anchored to a table or stretcher to adapt better to curved body surfaces. Both clamp a small ultrasound probe against the skin and can tighten automatically under computer control. To test them safely, the researchers built life-like silicone “phantoms” of the chest, abdomen, and groin with artificial arteries and veins that circulate fluid under pressure, mimicking a real bleed.
How the Smart System Sees and Squeezes
Two kinds of AI models drive the system. First, an object-detection model analyzes each ultrasound frame and draws digital boxes around the artery, vein, and underlying bone. This helps the device “know” when it is lined up correctly over the vessel and a firm surface. Second, a classification model judges whether the artery is still open or has been essentially shut. The researchers tried several versions and found that simple rules worked best: rather than estimating exact percentages of blood-flow reduction, the most accurate approach was a yes-or-no decision based on whether the dark, hollow center of the artery (“lumen”) had disappeared in the image. When this model signaled that the lumen had collapsed, the motor continued to tighten for a few more seconds to ensure at least a 90% drop in flow in the test setup.
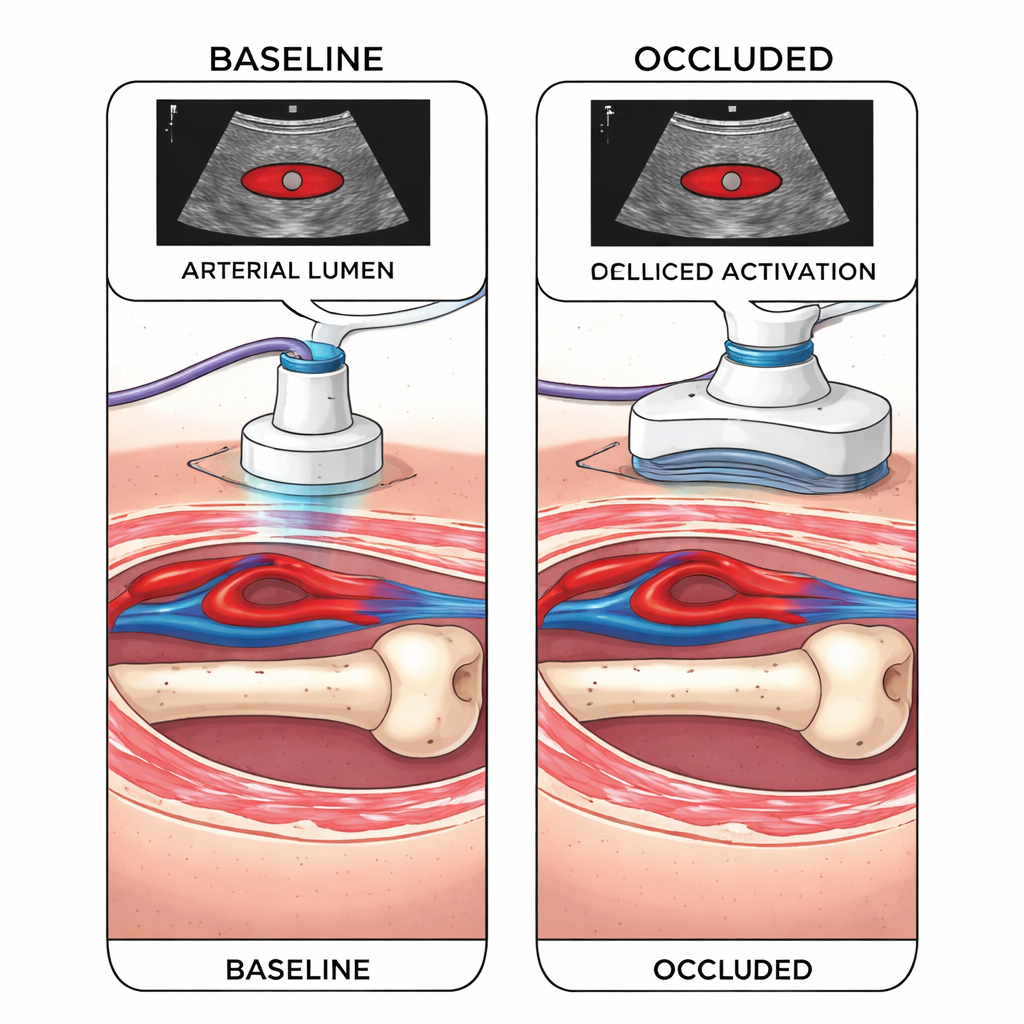
Performance Compared to Existing Devices
In timing tests, the new prototypes were put head-to-head against established commercial junctional tourniquets at the subclavian, aortic, and femoral sites. The FRejT design was especially promising: it consistently reached effective vessel occlusion as fast as or faster than current devices, and in some cases in about half the time. The BaTS design initially lagged and was more prone to slipping, but once the AI guidance and occlusion detection were fully integrated, both prototypes were able to find the target and stop flow in roughly 20 seconds in a femoral model. The guidance AI performed best when the ultrasound probe was close to the ideal location and the images were clear, and the researchers tuned their logic so that detecting just one major vessel plus bone was enough to trigger the occlusion sequence.
What This Could Mean for Future Trauma Care
To keep testing safe and controlled, all of these experiments were carried out in laboratory phantoms rather than in people or animals. That means there is still a long road ahead: the devices must be ruggedized, adapted for field conditions, tested on more realistic tissues, and evaluated with many different users. Even so, the work shows that it is technically feasible to combine ultrasound, AI, and automated mechanics into a single system that can quickly locate and compress deep blood vessels without expert interpretation. If further development is successful, such smart junctional tourniquets could give medics, police officers, or even trained bystanders a powerful new tool to stop otherwise untreatable bleeding long enough to get patients to definitive care.
Citation: Hernandez Torres, S.I., Winter, T., Mejia, I. et al. Smart, automated junctional tourniquets leveraging AI-driven ultrasound guidance. Sci Rep 16, 6865 (2026). https://doi.org/10.1038/s41598-026-37467-1
Keywords: hemorrhage control, junctional tourniquet, ultrasound, artificial intelligence, trauma care