Clear Sky Science · en
Therapeutic effects of IGF-2 on NMOSD by inhibiting astrocyte apoptosis through modulation of the IGF-1R/PI3K/AKT signaling pathway
Protecting the Brain in a Little-Known Autoimmune Disease
Neuromyelitis optica spectrum disorder, or NMOSD, is a rare but devastating autoimmune disease that attacks the optic nerves and spinal cord, often leaving patients with blindness, paralysis, or serious cognitive problems. Current drugs mainly calm the immune system, are expensive, and do not directly protect brain cells from damage. This study explores whether a natural growth-related molecule in the blood, called IGF‑2, might help shield brain cells from injury in NMOSD and offer a new way to treat the disease.
A Harmful Antibody and a Missing Protector
In NMOSD, the body makes antibodies against a water-channel protein called AQP4, which sits on star-shaped support cells in the brain called astrocytes. When these antibodies bind, they trigger immune attacks that damage astrocytes, strip away myelin (the insulating coating on nerves), and can harm neurons. The researchers compared blood from women with AQP4‑antibody–positive NMOSD to healthy volunteers. Using a broad protein survey, they found that IGF‑2 levels were sharply lower in NMOSD patients, while two IGF‑binding proteins that can trap IGF‑2 were higher. Importantly, patients with the lowest IGF‑2 tended to have worse disability and poorer scores on thinking and memory tests, linking this molecule to disease severity and cognitive health.
Standard Treatment Helps IGF-2 Recover
Many NMOSD patients receive rituximab, a drug that removes antibody-producing B cells. The team measured blood proteins before and after rituximab treatment. After therapy, IGF‑2 levels rose and the binding proteins fell, moving closer to the pattern seen in healthy people. This suggests that taming the immune system may allow the body to restore some of its own protective factors. While rituximab is not designed to act on IGF‑2, the shift in this molecule hints that it may be part of the body’s recovery response, and that monitoring IGF‑2 could help track how patients are doing over time.
Testing IGF-2 in a Mouse Version of the Disease
To see whether IGF‑2 can do more than signal disease severity, the scientists turned to a mouse model. They injected human AQP4 antibodies and human complement into the brains of mice, which reproduced key features of NMOSD: loss of AQP4 on astrocytes, inflammation, myelin damage, and limb weakness. Another group of mice first received a harmless virus that made their brain cells produce extra IGF‑2. These IGF‑2–boosted mice lost less weight, had milder movement problems, and showed less myelin loss and inflammation in brain and spinal cord tissue. Standard rituximab treatment also helped, but IGF‑2 overexpression appeared to protect brain tissue more directly, suggesting a neuroprotective role rather than only an immune-suppressing one.
How IGF-2 Calms Cell Death and Inflammation
The team then examined astrocytes in lab dishes exposed to NMOSD antibodies. Normally, these antibodies lowered levels of a key receptor called IGF‑1R and dampened a downstream safety pathway known as PI3K/AKT, while switching on “self-destruct” proteins and inflammatory signals. When the researchers forced astrocytes to make more IGF‑2, this protective pathway turned back on, markers of cell death dropped, and the cells released fewer inflammatory molecules such as IL‑1β, IL‑6, and TNF‑α. Blocking the IGF‑1R receptor partly erased these benefits, showing that IGF‑2 works mainly by activating the IGF‑1R/PI3K/AKT chain inside astrocytes. In living mice, boosting IGF‑2 also reduced activation of microglia, the brain’s immune sentinels, and lowered inflammatory signals in the bloodstream.
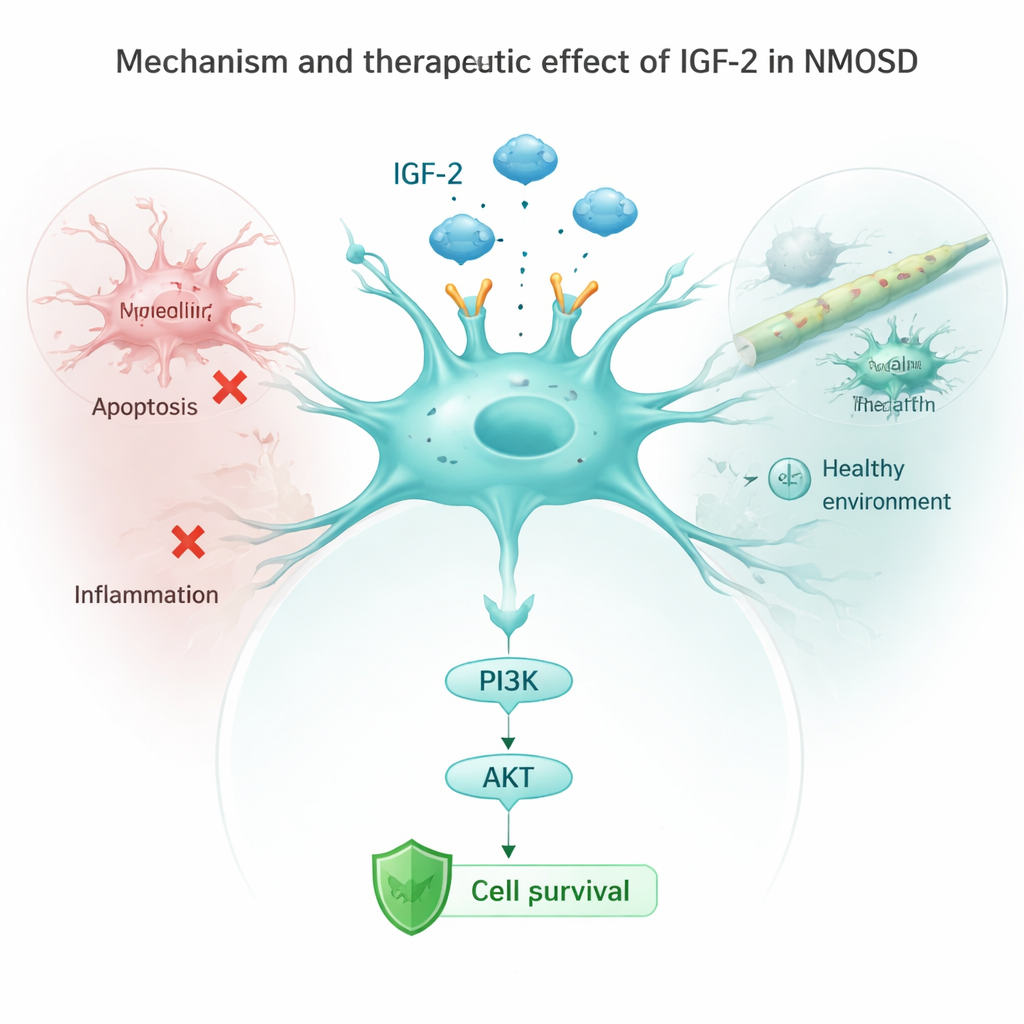
What This Could Mean for Patients
Taken together, these findings suggest that low IGF‑2 is both a warning sign and a missed opportunity in NMOSD. Patients with less IGF‑2 tend to be more disabled and think less clearly, while restoring or boosting IGF‑2 in experimental models helps astrocytes survive, reduces brain inflammation, and lessens nerve damage. Although this work is early and based on small patient numbers and animal studies, it points to IGF‑2 as a promising target for future treatments that would not only dial down the immune attack but also directly protect vulnerable brain cells.
Citation: Zhang, Y., Xu, N., Yu, K. et al. Therapeutic effects of IGF-2 on NMOSD by inhibiting astrocyte apoptosis through modulation of the IGF-1R/PI3K/AKT signaling pathway. Sci Rep 16, 6312 (2026). https://doi.org/10.1038/s41598-026-37456-4
Keywords: neuromyelitis optica, astrocytes, IGF-2, neuroinflammation, neuroprotection