Clear Sky Science · en
Sexual dysfunction in Brazilian women undergoing adjuvant endocrine therapy for breast cancer: prevalence and associated factors
Why this topic matters beyond the clinic
For many women, surviving breast cancer is only the beginning of a long journey back to a full life. Medicines that help keep the cancer from returning can quietly erode sexual desire, comfort, and intimacy, straining relationships and self‑esteem. This large Brazilian study shines a light on how common sexual problems are among women taking long‑term hormone‑blocking drugs after breast cancer, and why doctors, patients, and families need to talk about this hidden side effect.
Life after treatment: cancer controlled, but at a cost
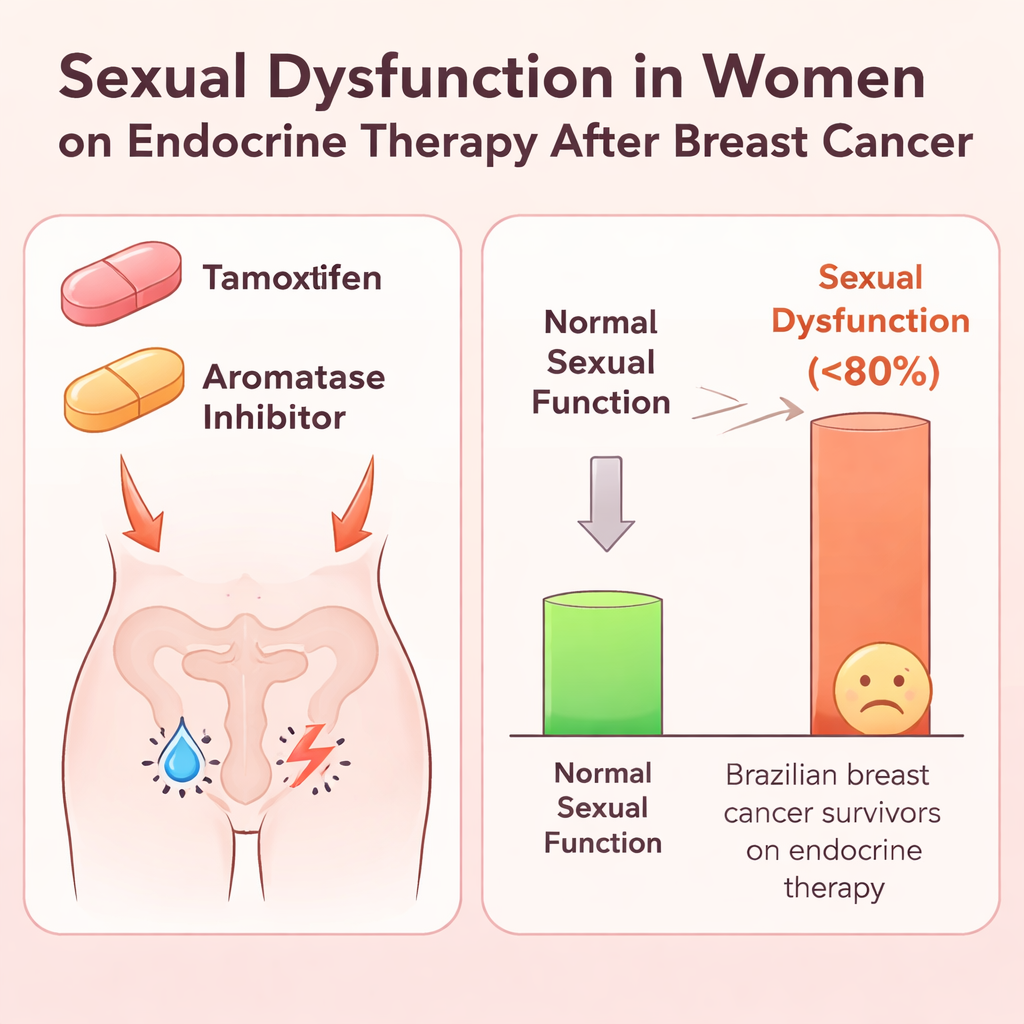
Most breast tumors are fueled by female hormones, especially estrogen. After surgery, chemotherapy, or radiation, many women take “endocrine therapy” pills such as tamoxifen or aromatase inhibitors for at least five years to cut the risk of the cancer coming back. These drugs save lives, reducing both recurrences and deaths. But by lowering or blocking estrogen, they can also trigger hot flashes, mood changes, joint pains – and sexual problems such as low desire, vaginal dryness, and pain during sex. Until now, there were few large, country‑wide data on how often Brazilian women on these treatments struggle with their sex lives.
What the researchers set out to learn
Researchers from 14 hospitals across all five regions of Brazil invited adult women with non‑metastatic, hormone‑sensitive breast cancer who had been on endocrine therapy for at least six months to complete detailed questionnaires. The team focused on those who had been sexually active in the previous four weeks, asking about desire, arousal, lubrication, orgasm, satisfaction, and pain using a standard tool called the Female Sexual Function Index. They also collected information from medical records, including age, cancer stage, type of surgery, type and duration of hormone treatment, other illnesses, and use of medications. Quality of life was measured with international cancer questionnaires that assess overall well‑being, body image, and the impact of treatment side effects.
How common were sexual problems?
Out of 774 women surveyed, about three in four said they had been sexually active before their cancer diagnosis, but less than half had had intercourse in the month before the study. Among the 346 women who were sexually active in that period, nearly four out of five – 79.8% – met the cut‑off for sexual dysfunction. Compared with women who did not meet that threshold, they were slightly older and scored lower on every part of the sexual function scale, from desire and arousal to orgasm and comfort. On average, their overall sexual function score was well below the level considered healthy, echoing international studies showing that breast cancer survivors face more sexual difficulties than women without cancer.
Which treatments and factors made a difference?
When the team used statistical models to sift through many possible influences, one treatment stood out. Women taking aromatase inhibitors – a type of hormone pill commonly prescribed after menopause – had about a 13% higher prevalence of sexual dysfunction than those on tamoxifen. Women with more years of formal education were also more likely to report sexual problems, which may reflect greater awareness and willingness to discuss intimate issues rather than a true biological difference. Importantly, women with sexual dysfunction reported worse scores for global health, social life, body image, sexual enjoyment, and side effects from systemic therapy. By contrast, factors such as type of breast surgery, use of chemotherapy or radiotherapy, menopausal status, and presence of other illnesses were not clearly linked to sexual dysfunction in this study.
What can be done to help
The authors emphasize that sexual health is often overlooked in routine cancer follow‑up, even though simple measures can make a real difference. They highlight counseling frameworks such as the PLISSIT and BETTER models, which guide health professionals on how to give patients “permission” to talk about sex, offer clear information, suggest practical strategies, and refer women for more intensive therapy when needed. Examples include regular use of non‑hormonal vaginal moisturizers and lubricants, pelvic floor exercises, and early referral to gynecologists, psychologists, or sexual health specialists. Survivorship programs that systematically ask about sexual concerns and provide support could be especially important for women on aromatase inhibitors, who appear to be at higher risk.
Bringing intimacy back into survivorship
This nationwide study reveals that nearly 80% of sexually active Brazilian women taking long‑term hormone‑blocking drugs after breast cancer experience significant sexual difficulties, with clear ripple effects on quality of life. For patients and their partners, these problems are not trivial; they touch identity, emotional connection, and day‑to‑day happiness. The message is straightforward: controlling cancer should not mean sacrificing a satisfying sex life. By normalizing conversations about sexuality, training oncology teams to address it, and building sexual health support into follow‑up care, clinicians can help survivors reclaim an important part of their lives.
Citation: Assad-Suzuki, D., Laperche-Santos, D., Resende, H. et al. Sexual dysfunction in Brazilian women undergoing adjuvant endocrine therapy for breast cancer: prevalence and associated factors. Sci Rep 16, 6173 (2026). https://doi.org/10.1038/s41598-026-37429-7
Keywords: breast cancer survivorship, endocrine therapy side effects, female sexual dysfunction, aromatase inhibitors, quality of life