Clear Sky Science · en
Integrating network toxicology, machine learning, and molecular dynamics simulations to reveal tanshinone iia’s dual mechanisms in TNBC and doxorubicin-induced cardiotoxicity
Why this research matters for patients
Doxorubicin is a workhorse drug for breast cancer, but it comes with a serious catch: it can damage the heart, sometimes years after treatment ends. At the same time, triple‑negative breast cancer—a hard‑to‑treat form that lacks common hormone and growth‑factor targets—still needs better therapies. This study explores whether tanshinone IIA, a compound extracted from the traditional Chinese herb Salvia miltiorrhiza, could both shield the heart from doxorubicin’s toxicity and help attack aggressive breast tumors, potentially making chemotherapy safer and more effective.
One compound, two big problems
Doxorubicin has saved countless lives, but its benefits are limited by dose‑dependent cardiotoxicity: above a certain lifetime dose, the risk of heart failure rises sharply. Clinicians often face a painful trade‑off between controlling the cancer and protecting the heart. Earlier laboratory work suggested that tanshinone IIA can boost antioxidant defenses in heart cells and directly kill cancer cells, but the underlying molecular reasons remained unclear. The authors set out to map, in a systematic way, how this single compound might dampen heart damage while also striking triple‑negative breast cancer, building a bridge between traditional medicine and modern precision oncology. 
Mining big biological data for heart protection
To understand heart protection, the team first gathered thousands of genes linked to cardiotoxicity from several large databases and combined them with known protein targets of doxorubicin and tanshinone IIA. Thirteen “overlap” targets emerged—proteins that were associated with heart damage, interacted with doxorubicin, and were also potentially modulated by tanshinone IIA. Using protein–protein interaction maps, the researchers narrowed these to six core players, including CCND1 (a cell‑cycle regulator), APAF1 (a trigger of programmed cell death), AR (androgen receptor), TERT (key to chromosome end maintenance), MMP2 (a tissue‑remodeling enzyme), and NQO1 (an antioxidant enzyme). Computer docking and molecular dynamics simulations suggested that both doxorubicin and tanshinone IIA can bind tightly to these proteins, but the tanshinone IIA–protein complexes are often more stable and energetically favorable, implying that the herbal compound may redirect or soften doxorubicin’s harmful signals in the heart.
Uncovering how it targets aggressive breast tumors
On the cancer side, the researchers focused on triple‑negative breast cancer gene‑expression datasets. They compared tumor samples with normal breast tissue to find hundreds of genes that were abnormally switched on or off. They then used a technique called weighted gene co‑expression network analysis to identify gene groups most tightly linked to the triple‑negative phenotype. By intersecting these cancer genes with predicted targets of tanshinone IIA, they distilled a small list of candidates and ran three independent machine‑learning methods to see which genes best distinguished tumor from normal tissue and were most likely to matter clinically. Converging evidence highlighted a single standout target: EZH2, a gene that helps package DNA and is often overactive in aggressive cancers.
Connecting tumor biology and the immune system
To understand why EZH2 matters in real patients, the team examined large cancer databases. They found that EZH2 is markedly more active in invasive breast tumors than in normal breast tissue. While its expression didn’t neatly track overall survival across all breast cancer cases, it was strongly linked to features of the tumor immune microenvironment. Higher EZH2 levels correlated with greater infiltration of several immune cell types—such as B cells and T cells—and with increased expression of molecules that both stimulate and suppress immune responses, including well‑known checkpoint proteins like CTLA‑4 and LAG‑3. These patterns suggest that EZH2 sits at a crossroads between tumor growth and immune control, and that a drug like tanshinone IIA, by influencing EZH2, might help reshape how the immune system recognizes and attacks triple‑negative breast cancer. 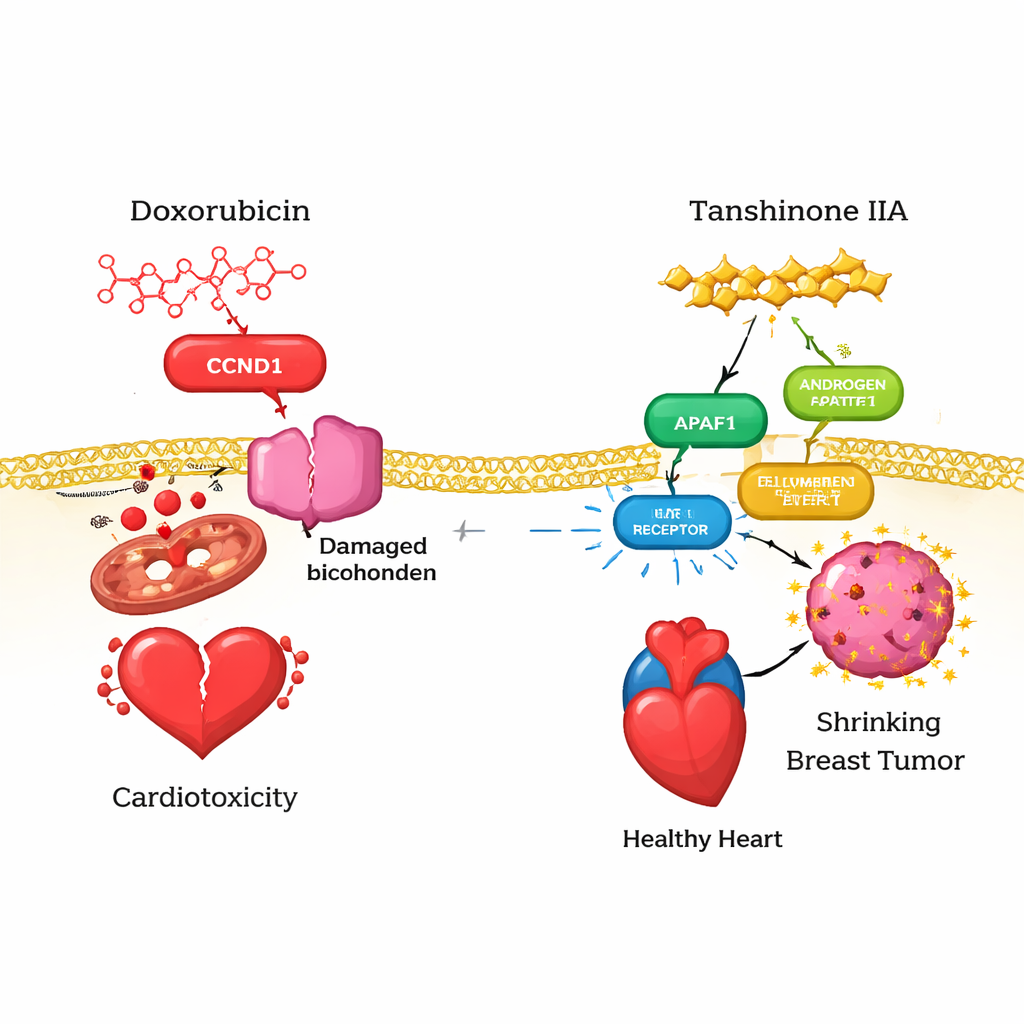
What this could mean for future treatment
In plain terms, this work proposes that tanshinone IIA could serve a dual role in breast cancer care: acting as a kind of “chemical shield” that reduces doxorubicin‑related heart damage by stabilizing key heart‑protective proteins, and doubling as a targeted weapon against triple‑negative breast cancer through its action on EZH2 and related immune pathways. The findings are based on computational and statistical analyses rather than animal or human trials, so they are an early blueprint rather than a ready‑to‑use therapy. Still, they offer a detailed molecular roadmap for designing combination treatments that preserve the life‑saving power of doxorubicin while curbing its long‑term risks—and for translating a traditional herbal component into a modern, mechanism‑guided cancer adjunct.
Citation: Wu, B., Lan, Xh., Chen, Xq. et al. Integrating network toxicology, machine learning, and molecular dynamics simulations to reveal tanshinone iia’s dual mechanisms in TNBC and doxorubicin-induced cardiotoxicity. Sci Rep 16, 6861 (2026). https://doi.org/10.1038/s41598-026-37428-8
Keywords: tanshinone IIA, doxorubicin cardiotoxicity, triple-negative breast cancer, EZH2, network pharmacology