Clear Sky Science · en
Analysis of systolic cardiac function in PERM1-knockout mice using large cohorts of animals
Why tiny mouse hearts matter to human health
Heart failure is often described as a tired pump, but what actually makes a heart weak is surprisingly complex. In this study, scientists used an unusually large group of mice to ask a focused question: does turning off a single gene called PERM1, known for helping heart cells make energy, also change how strongly the heart squeezes? By carefully scanning the hearts of more than 170 mice, they show that this gene subtly reshapes the heart and lowers its pumping strength, while the animals largely remain outwardly healthy. Their findings help settle a debate in the field and offer a roadmap for designing better heart experiments in the future.
A gene that links fuel use to heart squeeze
PERM1 was first identified as a switch that boosts energy production in muscle cells, including the heart. Earlier work showed that PERM1 levels drop in failing hearts in both mice and humans, raising the possibility that low PERM1 might actually contribute to weak heartbeats. But studies using PERM1-knockout mice—animals engineered to lack the gene—had produced conflicting answers about whether their hearts truly pump less effectively. Because PERM1 is being eyed as a possible target for new heart failure therapies, the authors set out to test its impact on heart function with enough animals and rigorous statistics to give a clear yes-or-no answer.
A big-number look at small hearts
The team gathered echocardiogram data—the same ultrasound technique used in cardiology clinics—from every PERM1-knockout mouse and its healthy littermate used in their lab over an 18‑month period. This retrospective sweep produced 84 normal (wild-type) and 88 knockout animals, a sample size far larger than is typical in mouse heart studies. From the ultrasound images, they measured standard indicators of how well the left ventricle, the main pumping chamber, works. The key number was ejection fraction, the percentage of blood pushed out with each beat, along with related measures such as how much the ventricle’s diameter shrinks when it contracts, how thick the walls are, and the total amount of blood pumped per minute.
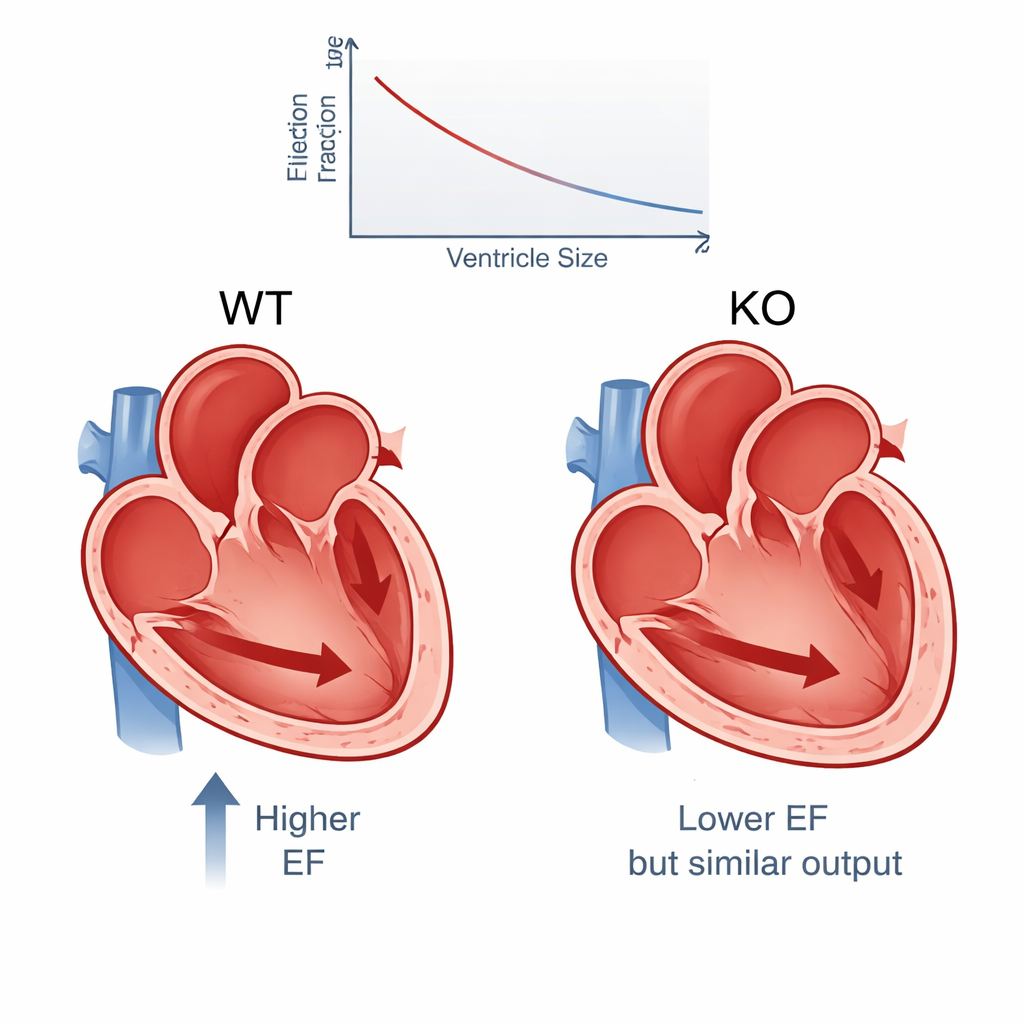
Weaker squeeze, bigger chamber, similar output
The results show a consistent pattern. On average, ejection fraction dropped from about 65% in normal mice to 54% in PERM1‑knockouts, a difference large enough to matter clinically in human patients and backed by extremely strong statistics. Other measures of contraction, such as fractional shortening and wall thickening, also declined. At the same time, the knockouts’ left ventricles were noticeably larger, especially at the end of contraction, indicating that more blood was left behind in the chamber. Despite this weaker squeeze, the total amount of blood pumped per minute fell only slightly—by roughly 6%—because the enlarged chambers could hold more blood to begin with. In other words, the heart partially compensated for poor contraction by stretching its main chamber.
Hidden diversity in “identical” hearts
Because the study included so many animals, the researchers could look beyond averages and study how individual hearts varied. Even among genetically identical, healthy mice, they saw a surprisingly wide spread in ejection fraction. Knocking out PERM1 shifted the entire distribution downward—more hearts with lower pumping strength—but did not make the spread itself wider. About 7% of knockout mice had ejection fractions below 40%, a level that would qualify as heart failure with reduced ejection fraction in people, yet these mice still maintained near-normal blood output. Statistical analyses showed that sex, age, and body weight explained only a small portion of the variability; most of the differences likely stem from harder-to-measure biological factors such as subtle environmental influences or epigenetic changes.
What this means for future heart research
To help other scientists design better experiments, the authors used their large dataset to calculate how many animals are typically needed to detect meaningful changes in each heart parameter. They found that the modest group sizes often used in the field can easily miss real but moderate effects, especially for subtle measures like stroke volume or cardiac output. Overall, the study makes a strong case that losing PERM1 causes a specific remodeling of the heart—larger chambers, weaker contraction—rather than outright failure, and that this remodeling is partly balanced by structural changes that help preserve blood flow. For non-specialists, the takeaway is that a single energy-regulating gene can nudge the heart into a new operating mode, one that looks healthy from the outside but carries quieter shifts in how the pump is built and how hard it works.
Citation: Zaitsev, A.V., Sreedevi, K., Goode, B. et al. Analysis of systolic cardiac function in PERM1-knockout mice using large cohorts of animals. Sci Rep 16, 7705 (2026). https://doi.org/10.1038/s41598-026-37420-2
Keywords: heart failure, cardiac contractility, mitochondrial energetics, mouse models, echocardiography