Clear Sky Science · en
Alterations in peripheral blood B cell subsets and their clinical significance in systemic lupus erythematosus
Why the Body Turns Against Itself
Systemic lupus erythematosus, or lupus for short, is an autoimmune disease in which the body’s defense system mistakenly attacks its own tissues, from the skin and joints to the kidneys and brain. Doctors know antibodies are involved, but they still need better ways to spot lupus early, track how active it is, and tailor treatment. This study looks closely at one key group of immune cells, B cells, in the blood of people with lupus to see how their balance changes and whether these changes could improve diagnosis and guide care.
The Immune Cells at the Center of Lupus
B cells are white blood cells that normally help protect us from infections. When they first encounter a germ, many B cells are still “naïve,” not yet trained for a specific target. Some of these mature into “memory” B cells that provide long‑lasting protection, while others become plasmablasts and plasma cells that churn out antibodies. In lupus, B cells start making antibodies against the body’s own DNA and other components, fueling widespread inflammation. The researchers set out to measure how these three major B cell types are distributed in the blood of different groups: newly diagnosed lupus patients, patients with stable disease, patients with serious kidney involvement called lupus nephritis, and healthy volunteers.
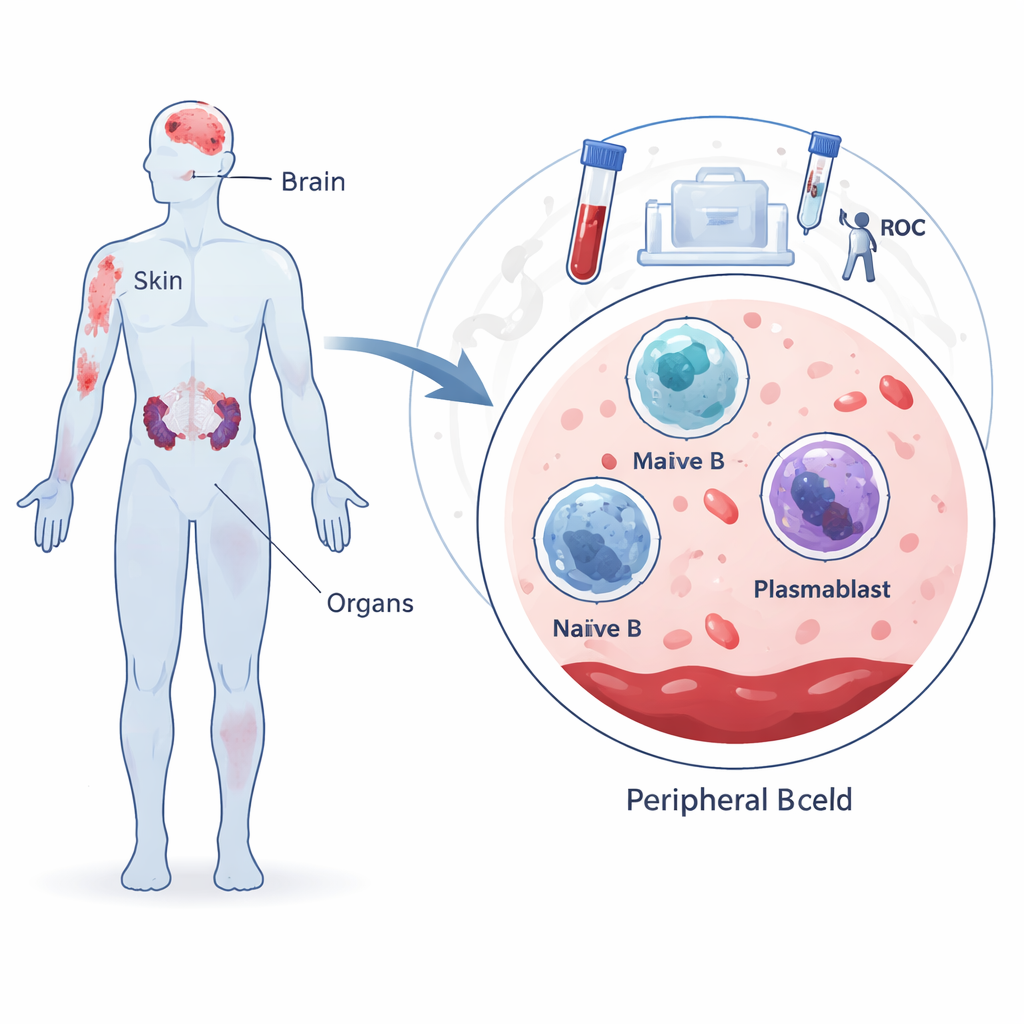
Comparing Patients and Healthy Volunteers
The study included 64 people with lupus and 20 healthy controls. Using a laboratory method called flow cytometry, the team identified naïve B cells, memory B cells, and plasmablasts in blood samples and compared their proportions across groups. They found a striking and consistent pattern: memory B cells were sharply reduced in all lupus patients compared with healthy individuals, regardless of whether their disease was newly diagnosed, stable, or involved the kidneys. Naïve B cells were also lower in many patients, especially those with more long‑standing or severe disease. In contrast, newly diagnosed patients showed an expansion of plasmablasts, reflecting an immune system in overdrive at the moment the disease is first recognized.
Turning Cell Counts into Diagnostic Clues
To test whether these changes could help doctors identify lupus, the authors used a common statistical approach called ROC analysis, which weighs how often a test is right or wrong. The single best performer was the share of memory B cells among all white blood cells. At a certain cutoff, this measure correctly flagged 80 percent of lupus patients and correctly reassured 80 percent of healthy people, an unusually strong result for a simple blood marker. Ratios involving plasmablasts and naïve B cells also provided useful information, particularly for ruling out people who do not have lupus, though they were somewhat less powerful than memory B cells.
Links to Autoantibodies and Organ Damage
Lupus is famous for its autoantibodies, especially anti‑nuclear antibodies and anti‑double‑stranded DNA (anti‑dsDNA), which are used in diagnosis and are tied to organ damage. The study showed that higher antibody levels went hand in hand with more disturbed B cell patterns. Patients with strong anti‑nuclear antibody signals had fewer naïve and memory B cells and a higher fraction of plasmablasts, suggesting more intense ongoing immune activation. Those with anti‑dsDNA antibodies showed a similar profile, again with a particular rise in plasmablasts. Patients whose blood contained anti‑Sm antibodies, another hallmark of lupus, also had a notably higher proportion of plasmablasts. These findings hint that different antibody patterns may reflect distinct “flavors” of B cell disruption within lupus.
What This Means for People Living with Lupus
In plain terms, this work shows that the balance of B cells in the blood is clearly and consistently altered in people with lupus: long‑term memory cells are depleted, while antibody‑producing plasmablasts swell in active disease and in patients with key autoantibodies. Measuring these cell types could help doctors recognize lupus earlier, judge how active it is, and perhaps choose treatments that better match each patient’s immune profile. Although the study was relatively small and done at a single center, it strengthens the idea that tracking B cell subsets may become part of a more precise, personalized approach to lupus care in the future.
Citation: Huang, J., Xu, Z., Zhang, X. et al. Alterations in peripheral blood B cell subsets and their clinical significance in systemic lupus erythematosus. Sci Rep 16, 6293 (2026). https://doi.org/10.1038/s41598-026-37415-z
Keywords: systemic lupus erythematosus, B cells, autoantibodies, immune biomarkers, lupus nephritis