Clear Sky Science · en
Decreased N-acetylaspartate plus N-acetyl-aspartyl-glutamate levels in the caudate of schizophrenia patients with tardive dyskinesia
Why this matters for everyday life
Tardive dyskinesia is a troubling side effect of long-term antipsychotic treatment that can cause uncontrollable facial grimacing, tongue movements, or limb jerks. For people living with schizophrenia and their families, these movements can be embarrassing, disabling, and difficult to treat. This study asks a simple but important question: is tardive dyskinesia just a drug side effect, or does it reflect deeper, long-lasting changes in the brain’s movement circuits?
A closer look at an invisible movement hub
The researchers focused on the caudate nucleus, a small but crucial structure deep inside the brain that helps coordinate movement, motivation, and habit formation. Earlier brain scans had hinted that this region may be altered in people with tardive dyskinesia, but its chemistry had not been carefully measured. Using a noninvasive technique called proton magnetic resonance spectroscopy—essentially a chemical "listening device" built into an MRI scanner—the team measured key brain chemicals in the caudate of three groups: patients with schizophrenia and tardive dyskinesia, patients with schizophrenia but no abnormal movements, and healthy volunteers.
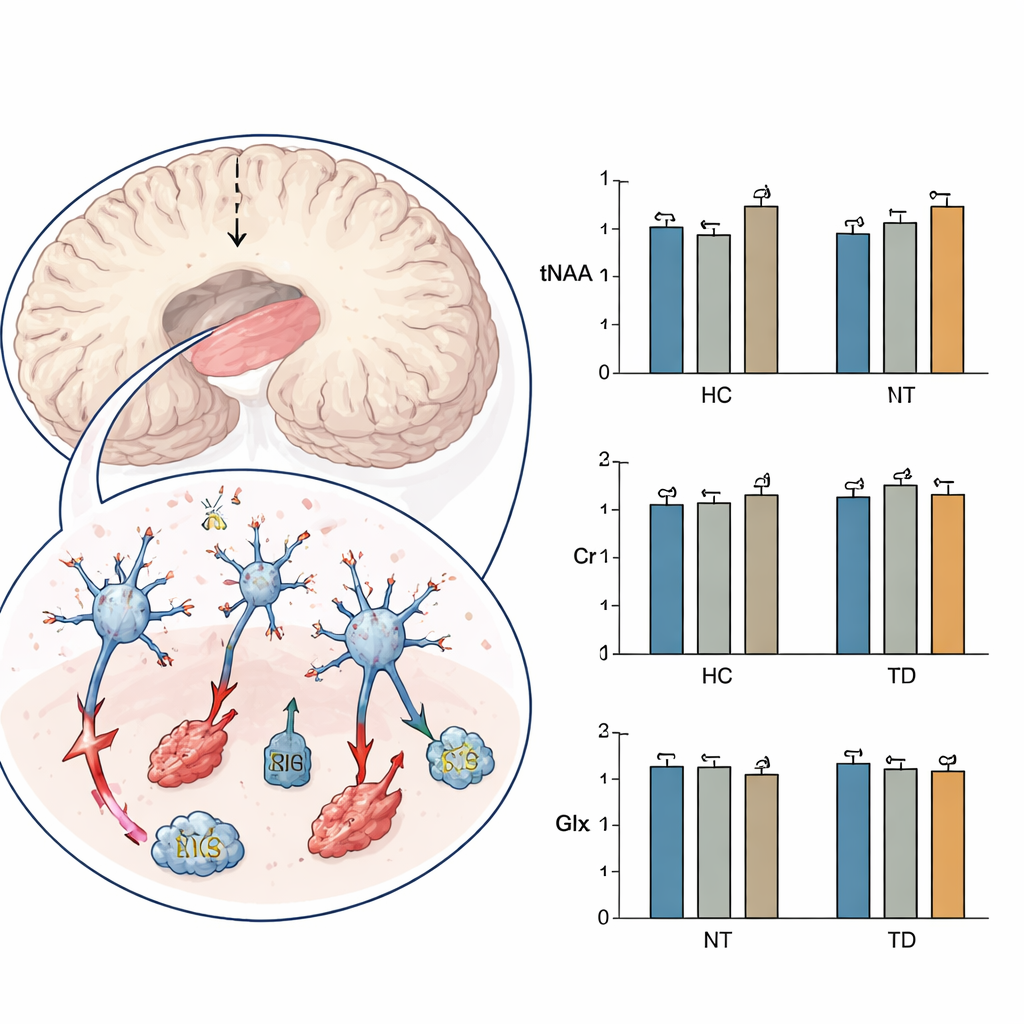
What the brain chemicals revealed
The main chemical of interest was a compound called tNAA, which reflects the health and energy status of nerve cells. The team also measured creatine, involved in cellular energy supply, and a combined signal from glutamate and glutamine, which are linked to excitatory brain signaling. Overall, the three groups were similar in age, sex, education, symptom severity, and antipsychotic dose, though patients with tardive dyskinesia had lived with schizophrenia for longer. When the researchers compared the brain data, one signal stood out: tNAA levels in the caudate were significantly lower in patients with tardive dyskinesia than in those without these movements, while creatine and glutamate-related levels did not differ meaningfully among the groups.
Signs of stressed and vulnerable brain cells
Lower tNAA suggests that nerve cells in the caudate may be under chronic stress or partially damaged. The authors discuss several possible culprits: long-term oxidative stress (a kind of “rusting” in the brain), mitochondrial strain (the tiny power stations inside cells), low blood flow, and overexcitation by neurotransmitters such as glutamate. They note that similar drops in tNAA have been seen in other movement disorders, including Huntington’s disease and certain inherited ataxias, where nerve cells slowly degenerate. In this study, tNAA was lowest in patients with tardive dyskinesia, somewhat reduced in patients without it, and relatively higher in healthy people—hinting at a gradient of vulnerability centered on the caudate.
Not just the movements you see
Interestingly, the severity of involuntary movements, measured by a standard rating scale, did not track directly with tNAA levels. In other words, people with more obvious movements did not necessarily have lower tNAA than those with milder symptoms. This suggests that reduced tNAA is less a day-to-day marker of how bad the movements are and more a background sign of an underlying brain state. The authors propose that this chemical change may reflect a long-term, trait-like vulnerability that makes some people more likely to develop tardive dyskinesia when exposed to antipsychotic drugs, rather than a simple consequence of current symptom intensity.
What this means going forward
For patients, families, and clinicians, these findings strengthen the idea that tardive dyskinesia is not just an unfortunate side effect but may be tied to subtle, long-lasting changes in brain circuits that control movement. A lower tNAA signal in the caudate appears to mark reduced nerve cell health in this region, helping explain why some individuals develop persistent abnormal movements even after medication changes. While more long-term and treatment-focused studies are needed, this work moves the field closer to biological markers that could one day help identify at-risk patients early, guide safer prescribing, and inspire new therapies aimed at protecting or restoring vulnerable brain cells.
Citation: Yu, T., Li, Y., Li, N. et al. Decreased N-acetylaspartate plus N-acetyl-aspartyl-glutamate levels in the caudate of schizophrenia patients with tardive dyskinesia. Sci Rep 16, 6773 (2026). https://doi.org/10.1038/s41598-026-37396-z
Keywords: schizophrenia, tardive dyskinesia, brain imaging, movement disorders, antipsychotic side effects