Clear Sky Science · en
3D heterotypic models of glioblastoma reveal the impact of microglia on cellular organization and the production of a distinct secretome
Why tiny brain tumors in a dish matter
Glioblastoma is one of the deadliest brain cancers, in part because it resists nearly every treatment doctors throw at it. A big reason for this failure is that standard lab tests don’t capture the full complexity of a real brain tumor. This study builds more life‑like, three‑dimensional “mini‑tumors” that include not only cancer cells but also microglia – the brain’s resident immune cells – to reveal how these helpers can quietly make tumors tougher, more invasive, and harder to kill.
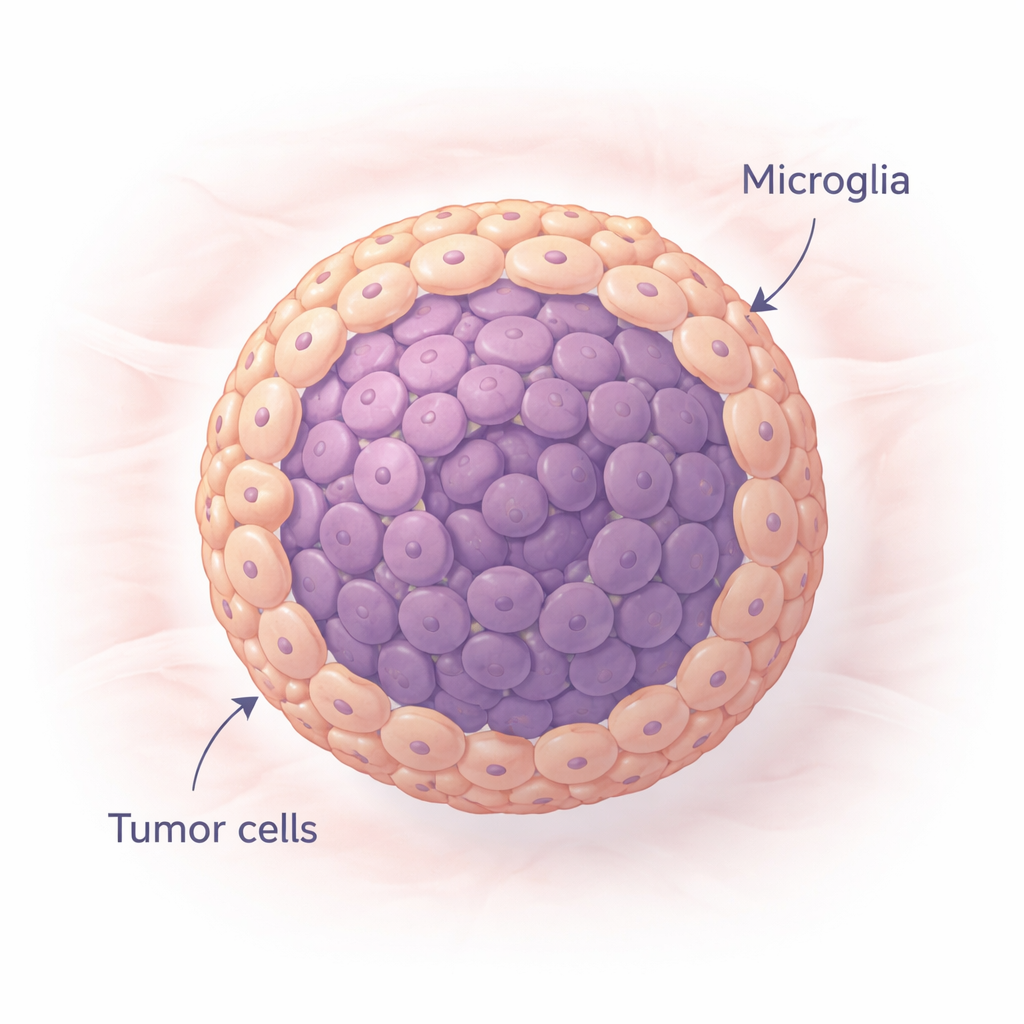
Building mini tumors that feel more like the real thing
The researchers created tiny spherical clusters of cells, called spheroids, using either a common glioblastoma cell line or stem‑like tumor cells taken from a patient. Some spheroids contained only tumor cells, while others mixed tumor cells with microglia in proportions similar to those seen in patients. Grown in special low‑stick dishes, these cells naturally clumped into compact balls over a week, staying largely alive throughout. By comparing “tumor‑only” and “tumor‑plus‑microglia” spheroids, the team could see how the added brain immune cells reshaped growth, structure, and behavior.
Microglia as growth boosters and invaders’ guides
Adding microglia changed the character of the mini‑tumors. Mixed spheroids grew larger and packed in more cells than tumor‑only spheroids, showing that microglia helped fuel overall expansion. In patient‑derived models, these mixed spheroids even developed multiple dense centers, a feature linked to aggressive disease. When the spheroids were placed onto a soft gel that mimics brain tissue, cells from the mixed spheroids spread out more persistently, particularly for one tumor cell line. Tracking which cells moved showed that microglia often led the way at the edges, echoing how they cluster around real glioblastoma masses and help open paths into nearby brain tissue.
A protective shell that blunts chemotherapy
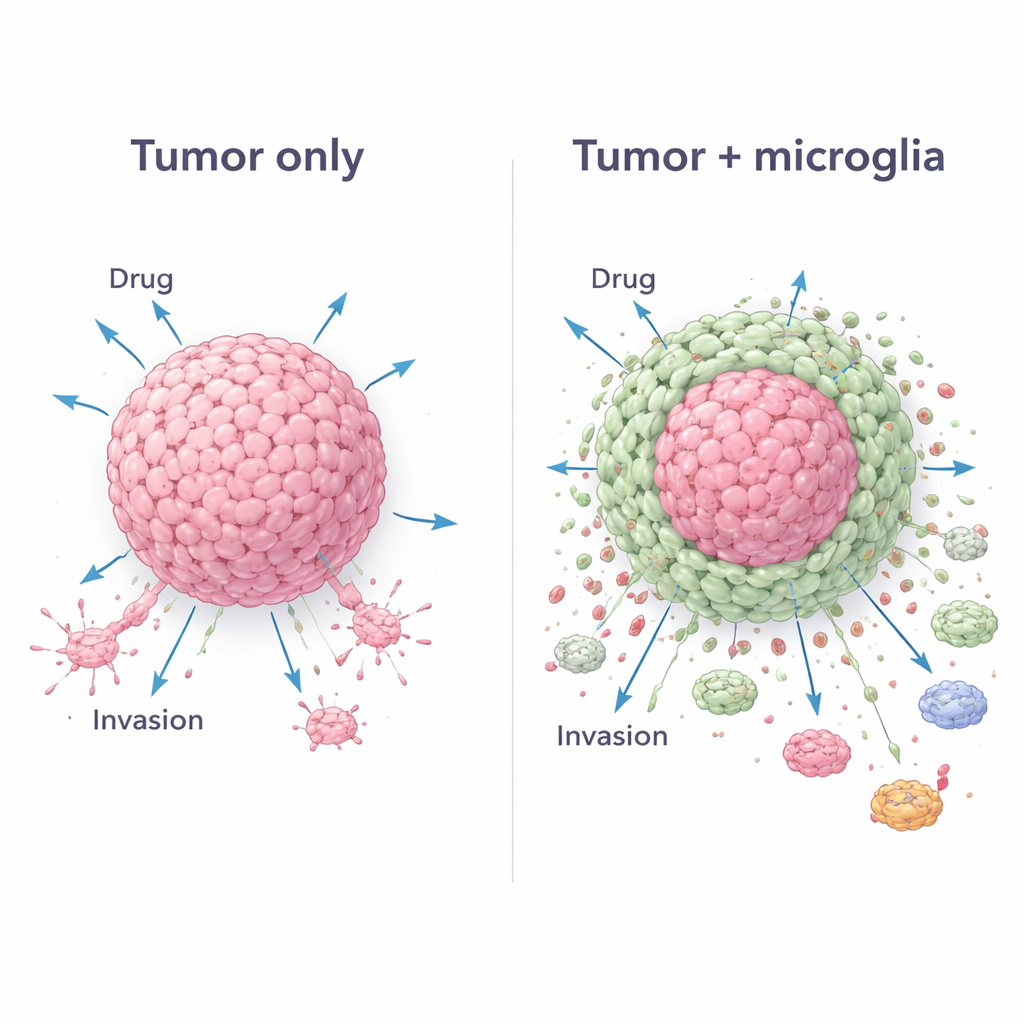
The team next tested the standard drug temozolomide, widely used in glioblastoma care. Tumor‑only spheroids lost many cells after drug exposure, indicating substantial damage. In contrast, mixed spheroids retained far more cells and in some cases even bounced back with higher cell numbers after 48 hours. High‑resolution imaging revealed why: in the mixed models, glioma cells concentrated in the center while microglia formed a surrounding shell. This core‑and‑shell architecture appeared to act like a living shield, making it harder for the drug to reach and kill the tumor core. Measurements of cell types before and after treatment showed that tumor cells were preferentially lost, while microglia survived and became relatively more abundant, reinforcing this protective effect.
Rewiring the body’s defenses in favor of the tumor
Glioblastoma also survives by hijacking the immune system. To explore this, the researchers exposed human blood‑derived immune cells to liquid collected from the spheroids. Fluid from mixed tumor‑microglia spheroids pushed many of these cells to settle down and adopt an “M2‑like” state, an anti‑inflammatory mode known to support tumor growth rather than attack it. In migration tests, more circulating immune cells moved toward mixed spheroids than toward tumor‑only ones, suggesting the presence of strong chemical lures. A broad survey of secreted proteins confirmed that mixed spheroids released a distinct cocktail of signals tied to invasion, treatment resistance, and immune suppression – a signature absent when either cell type was grown alone.
What this means for future brain cancer treatments
For non‑specialists, the key message is that glioblastoma cannot be understood by studying cancer cells in isolation. This work shows that when tumor cells and microglia grow together in 3D, they self‑organize into a tumor core wrapped in a microglia shell that boosts growth, aids spread, blunts chemotherapy, and tricks incoming immune cells into helping rather than fighting. These realistic mini‑tumors capture many hallmarks of real glioblastoma, especially when patient‑derived cells are used. As a result, they offer a powerful testbed for designing drugs that not only target tumor cells but also disrupt their dangerous alliance with microglia, potentially bringing more effective therapies closer to patients.
Citation: García-Sáez, C., Alonso-Marañón, J., García-Puga, M. et al. 3D heterotypic models of glioblastoma reveal the impact of microglia on cellular organization and the production of a distinct secretome. Sci Rep 16, 7246 (2026). https://doi.org/10.1038/s41598-026-37395-0
Keywords: glioblastoma, microglia, 3D tumor models, drug resistance, tumor microenvironment