Clear Sky Science · en
Identification and characterization of fibroblast-related biomarkers and pro-inflammatory subpopulations in periodontitis by integrated transcriptomic and single-cell analysis
Why your gums matter beyond your smile
Bleeding gums and loose teeth may seem like small problems, but chronic gum disease—known as periodontitis—can damage the jawbone, cause tooth loss, and is linked to conditions such as heart disease and diabetes. This study looks closely at a usually overlooked group of cells in the gums, called fibroblasts, to see how they help drive or calm inflammation. By combining big genetic datasets with single-cell analysis, the researchers uncover new warning signals and cell types that could improve how dentists diagnose and treat gum disease.
Hidden players in sore gums
Fibroblasts are the workhorse cells that build and maintain the connective tissue holding teeth in place. For years they were viewed mainly as structural “scaffolding.” This research shows they are much more: fibroblasts can act like miniature control centers for inflammation. When bacteria disrupt the normal balance of microbes in the mouth, fibroblasts respond by releasing chemical messengers that attract immune cells and can either help repair tissue or intensify damage. The authors focused on finding fibroblast-related gene signals that differ between healthy gums and periodontitis, and on spotting fibroblast subgroups that become especially inflammatory in diseased tissue.
Scanning gum tissue cell by cell
To achieve this, the team mined several large gene-expression datasets from patients with healthy gums and periodontitis, along with a modern single-cell sequencing dataset that profiles thousands of individual cells. They first mapped the major cell types in gum tissue—such as immune cells, blood vessel cells, and fibroblasts—and confirmed that fibroblast numbers and activity were higher in diseased gums. They then searched for genes that were both altered in periodontitis and specifically linked to fibroblasts. Using a statistical method that narrows down the most informative features, they pinpointed six key fibroblast-related genes whose combined activity pattern reliably distinguished periodontitis from health across multiple patient groups.
Risk patterns and immune “personalities” of gum disease
With these six genes, the researchers built a diagnostic model that performed well in separating diseased from healthy samples, and they translated it into a simple scoring chart—called a nomogram—that could, in principle, estimate an individual’s risk. When they grouped patients according to how strongly these fibroblast genes were expressed, three distinct forms of periodontitis emerged. One form showed a highly inflammatory immune landscape, packed with aggressive T cells and “attack mode” macrophages. A second form was dominated by cells that dampen immune responses, suggesting a more suppressed environment. A third form fell in between, with a more balanced or resting mix of immune cells. These patterns hint that not all gum disease is the same—and that fibroblasts help shape these different “personalities” of inflammation.
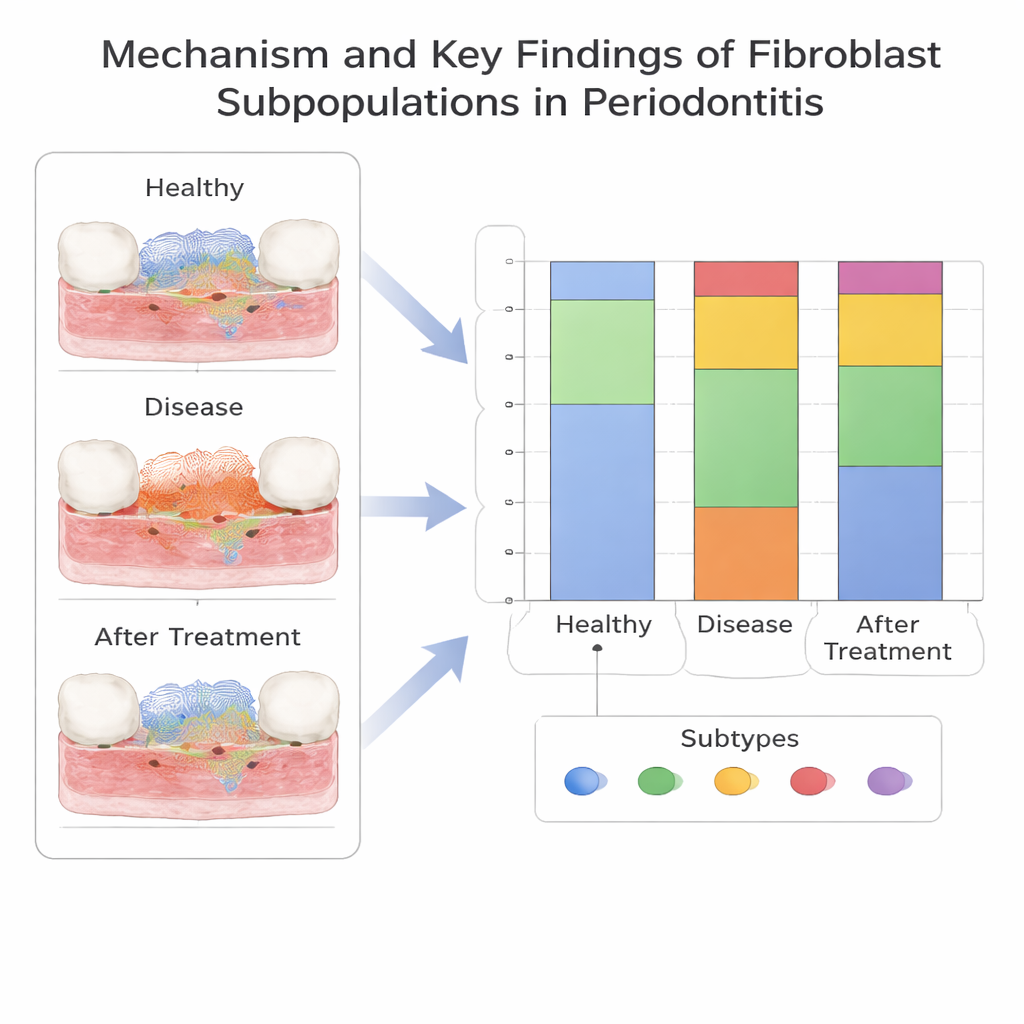
A new pro-inflammatory fibroblast subtype
Digging deeper at the single-cell level, the team divided fibroblasts into five subtypes based on their gene activity. One subtype, marked by a gene called CXCL13, stood out because it was much more common in periodontitis than in healthy gums. This CXCL13-positive fibroblast group appeared early along a developmental path and was linked with signals that attract immune cells and remodel the tissue matrix—features consistent with a strong pro-inflammatory role. In contrast, another fibroblast subtype was enriched in healthy tissue and may help keep the gum environment stable. The authors confirmed in patient samples that certain fibroblast-related genes, especially SELL, and the CXCL13-positive fibroblasts themselves, were indeed higher in diseased gums, reinforcing the computational findings.
What this means for future gum care
In plain terms, the study reveals that some fibroblasts in your gums behave like accelerators of inflammation, while others may act more like brakes or repair crews. By identifying precise gene markers and a distinct pro-inflammatory fibroblast subgroup, the work opens the door to more accurate tests that flag periodontitis earlier and more reliably. It also suggests that future treatments might target these specific fibroblast-driven pathways—dialing down harmful subsets like CXCL13-positive fibroblasts or boosting protective ones—to control gum inflammation more effectively and possibly reduce its impact on whole-body health.
Citation: Huang, M., Lin, Y., Wu, Z. et al. Identification and characterization of fibroblast-related biomarkers and pro-inflammatory subpopulations in periodontitis by integrated transcriptomic and single-cell analysis. Sci Rep 16, 6223 (2026). https://doi.org/10.1038/s41598-026-37385-2
Keywords: periodontitis, fibroblasts, gum inflammation, single-cell RNA sequencing, biomarkers