Clear Sky Science · en
Design and immunogenicity of a recombinant Saccharomyces boulardii secreting the P2-VP8 subunit rotavirus vaccine
Why a gut-friendly yeast could matter for child health
Rotavirus is a major cause of severe diarrhea in young children, especially in low‑income countries where medical care is harder to reach. Existing vaccines save many lives but work less well in these settings, and they must be kept cold from factory to clinic. This study explores a very different kind of vaccine: turning a common probiotic yeast into a tiny factory that releases a rotavirus protein in the gut, with the hope of triggering protection in an easy‑to‑deliver, stable form.
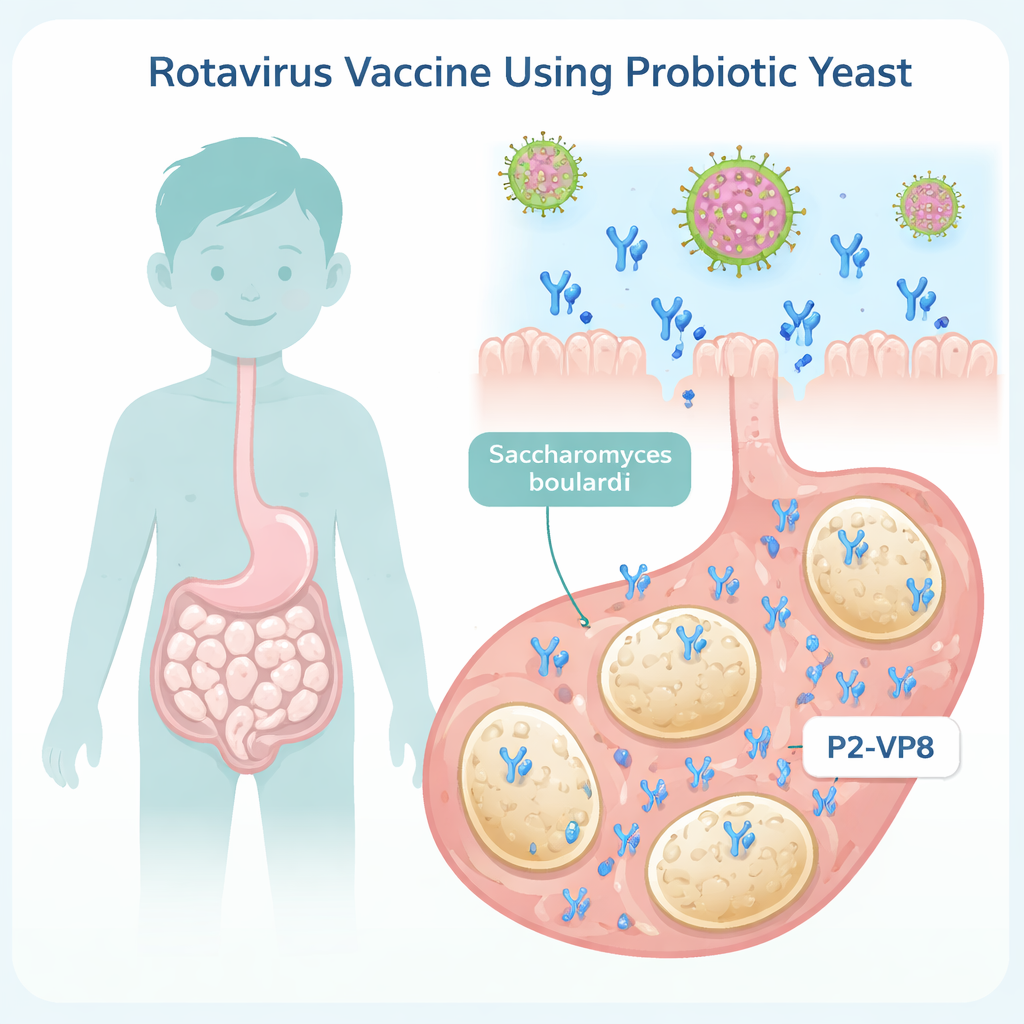
Turning a helpful microbe into a vaccine delivery vehicle
The researchers focused on Saccharomyces boulardii, a probiotic yeast already used to help prevent or treat diarrhea. Their idea was to engineer this yeast so that it secretes a fragment of a rotavirus surface protein, called VP8, which the virus uses to attach to human cells. They fused VP8 to a short helper piece of protein known as P2, previously shown to boost vaccine responses, creating a combined protein called P2‑VP8. Because only a small, non‑infectious part of the virus is used, this approach fits into the broader family of "subunit" vaccines that aim to be safer and easier to handle than whole‑virus vaccines.
Designing the vaccine candidate on the computer first
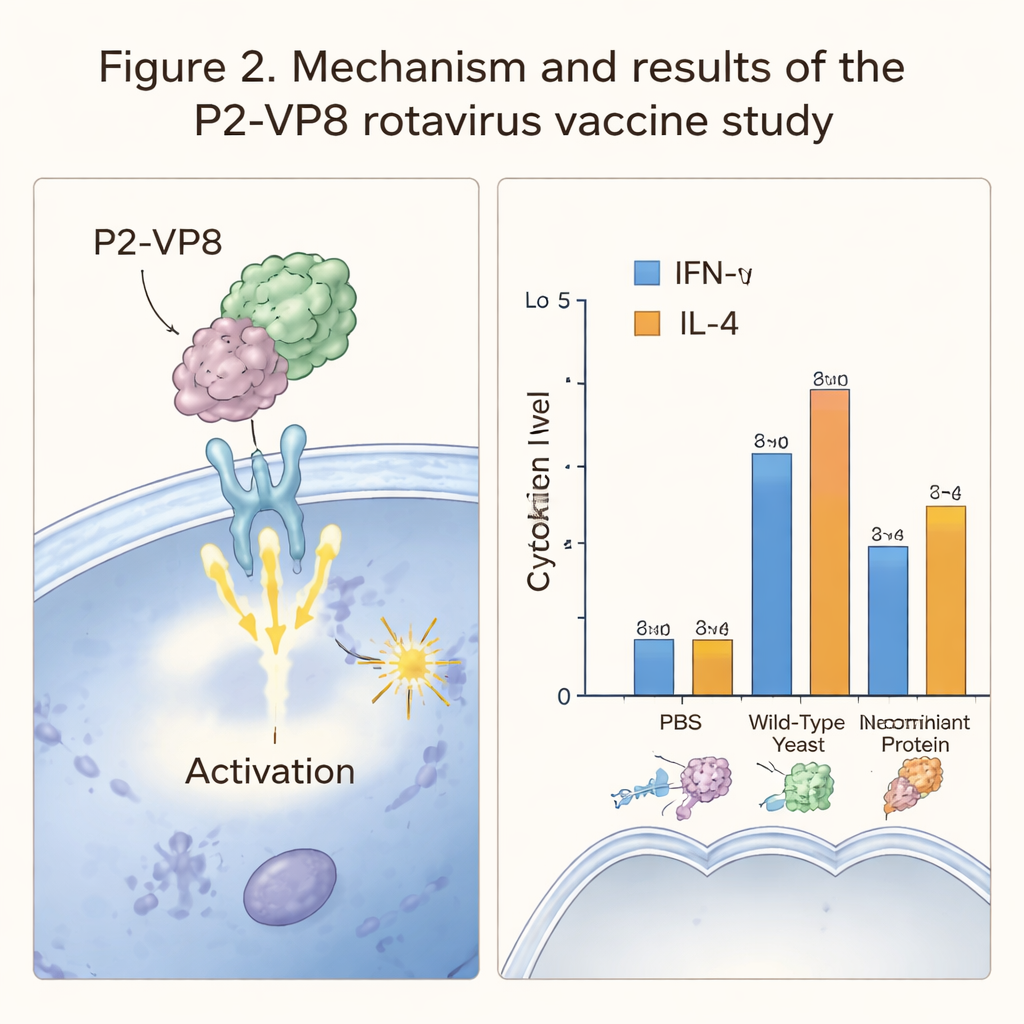
Before doing animal experiments, the team used a suite of computer tools to predict how the P2‑VP8 protein would behave. They mapped out sections of VP8 that immune cells are likely to recognize and checked whether these sections should be widely effective across different human genetic backgrounds. They then built a three‑dimensional model of P2‑VP8 and simulated its interaction with TLR3, a sensor on immune cells that helps detect viral infections. These simulations suggested that P2‑VP8 is stable, soluble, non‑toxic, and capable of forming a strong, sustained bond with the immune receptor, all encouraging signs for a vaccine candidate.
Engineering yeast to secrete the rotavirus protein
To make the probiotic yeast produce this protein efficiently, the researchers carefully rewrote the P2‑VP8 gene using codons—the three‑letter words of DNA—that Saccharomyces boulardii reads most easily. This optimization is like translating a recipe into the yeast’s native dialect so its machinery can follow it quickly and accurately. They then inserted the optimized gene into a yeast expression vector and transformed it into S. boulardii. In laboratory culture, they confirmed that the engineered yeast secreted a protein of the expected size using standard protein separation (SDS‑PAGE) and western blotting with custom‑made antibodies, demonstrating that the probiotic could indeed act as a living producer of the vaccine fragment.
Testing immune responses in mice
The team next looked at how this yeast‑based vaccine behaved in live animals. Mice received oral doses of encapsulated recombinant yeast over several weeks, while comparison groups received plain yeast, a simple salt solution, or purified P2‑VP8 protein by injection. After vaccination, the researchers measured key immune signaling molecules, IFN‑γ and IL‑4, which reflect different types of immune activation. Mice that got the engineered yeast or the injected protein showed higher levels of these cytokines than control animals, indicating that their immune systems had noticed and responded to the rotavirus fragment. However, they did not observe strong T‑cell proliferation or robust antibody levels in the yeast‑fed group, suggesting that the immune response to oral delivery was relatively weak.
What this work means and what still needs fixing
Overall, the study shows that a probiotic yeast can be turned into a factory that secretes a leading rotavirus vaccine fragment and that this construct behaves as expected in detailed computer tests. In mice, the engineered yeast triggered some immune activity but did not produce the strong, protective‑type responses that would be needed for a successful vaccine. For a lay reader, the takeaway is that the concept—using a friendly microbe as an edible vaccine—looks technically feasible, but the current design is not yet powerful enough. Future work will need to refine the protein design, dosing, and delivery strategy, and include direct tests of protection against rotavirus infection before such an approach could move closer to real‑world use.
Citation: Farhani, I., Yamchi, A., Nikoo, H.R. et al. Design and immunogenicity of a recombinant Saccharomyces boulardii secreting the P2-VP8 subunit rotavirus vaccine. Sci Rep 16, 6932 (2026). https://doi.org/10.1038/s41598-026-37374-5
Keywords: rotavirus vaccine, probiotic yeast, subunit vaccines, oral immunization, vaccine design