Clear Sky Science · en
Efficient apoptosis via double-strand breaks of DNA in human epithelial cells induced by non-thermal effect of high-field broadband terahertz waves
New Light on Killing Cancer Cells
Cancer treatments often walk a tightrope: they must destroy tumor cells without badly harming healthy tissue or the immune system. This study explores an unusual tool for that job—intense terahertz (THz) waves, a type of invisible light between microwaves and infrared. The researchers show that carefully tuned, high-field THz pulses can trigger cancer cells to commit “suicide” in a controlled way, without cooking the tissue or causing the messy cell death that leads to inflammation.
Why Gentle Cell Death Matters
Many common cancer therapies push cells toward death, but they often do so with collateral damage. Drugs and biological agents can lose effectiveness as tumors evolve resistance, and they may weaken the immune system. Physical methods like X-rays, radioactivity, or heat-based ablation can injure surrounding healthy tissue, frequently causing necrosis—a violent form of cell death that sparks inflammation and pain. A method that instead drives apoptosis, a tidy self-destruct process where cells quietly dismantle themselves, could offer a far less traumatic way to shrink tumors, especially those close to the skin.
Terahertz Waves Meet Breast Cancer Cells
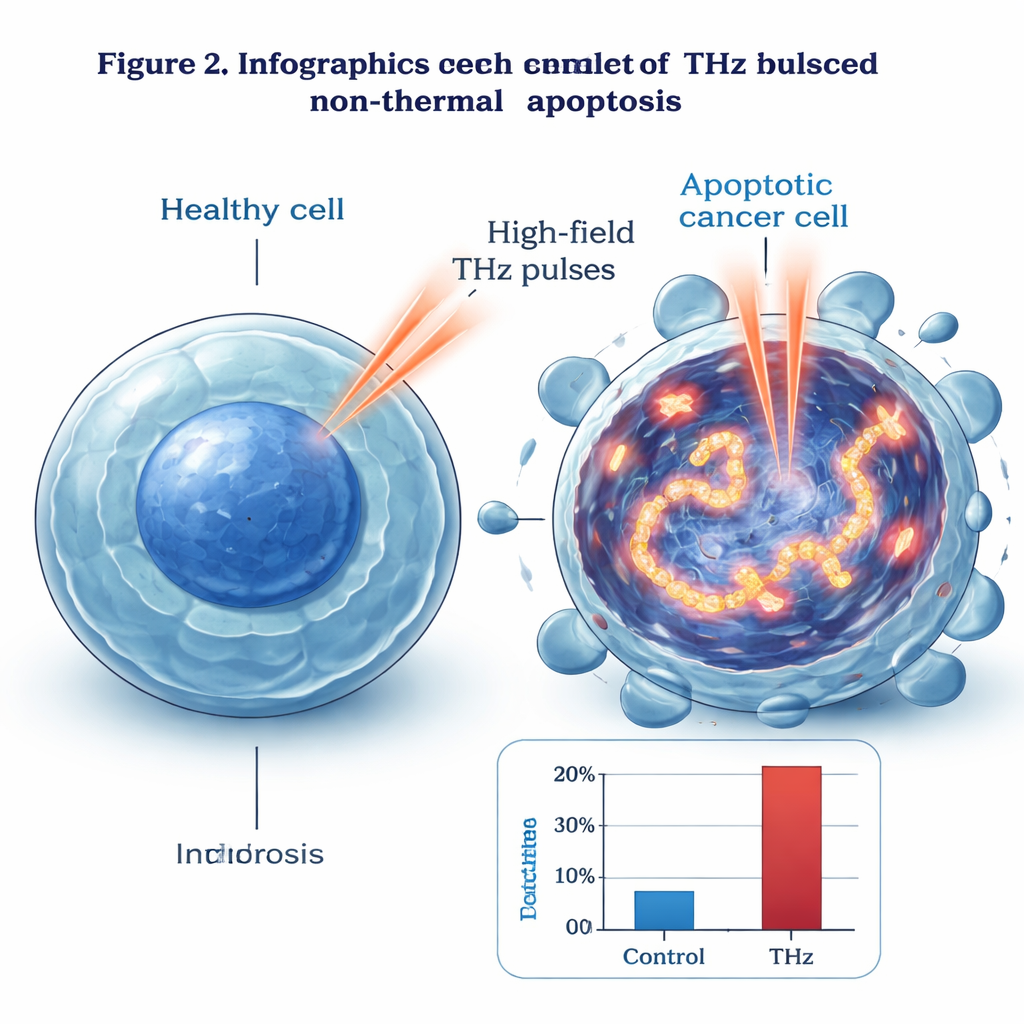
The team focused on human breast cancer cells (MCF‑7), a type of epithelial cell often used in lab studies. They exposed these cells to very strong, broadband pulsed THz waves for up to four hours while keeping them at normal body temperature. The field strengths reached several million volts per centimeter, far higher than in everyday devices. Under these conditions, the fraction of cells undergoing apoptosis climbed steadily with exposure time. With the highest THz intensity tested, nearly 99% of cancer cells entered apoptosis, while fewer than 1% died by necrosis. This massive, clean wave of programmed cell death suggests that the THz pulses are doing more than just warming the cells.
Non-Heating Effects and Safe Temperature Limits
Because water strongly absorbs THz radiation, the obvious concern is accidental heating. The scientists used thermal imaging to track temperature changes during irradiation. Even after four hours at moderate intensity, the cancer cells warmed only about 3 °C, and necrosis stayed below 4%. Separate control experiments in which cells were simply heated for four hours showed that temperatures below 41 °C produced almost no death, while a sharp jump in necrosis occurred above 43 °C. Even at their most intense THz settings, the researchers kept cell temperatures at or below this 43 °C threshold and still saw almost total apoptosis with minimal necrosis. By lowering the repetition rate of the pulses, they showed that temperature can be held in check while preserving strong anti-tumor effects, underscoring that a non-thermal mechanism is at work.
Shaking DNA Apart and Switching On Death Genes
To find that mechanism, the group looked inside the cells. Under an electron microscope, THz-treated cells showed classic early signs of apoptosis: condensed genetic material, altered nuclei, and intact outer membranes. Using a fluorescent marker called γ‑H2AX, they detected extensive double-strand breaks in the DNA—damage far more frequent in irradiated cells than in untreated or mildly heated controls. The damage pattern followed the intensity profile of the THz beam, becoming stronger toward its center. Gene and protein analyses revealed that a set of stress- and damage-responsive molecules, such as GADD45B and c‑Jun, were strongly activated, while well-known players in traditional cell-death routes, including p53 and many caspases, did not change much. The authors propose that intense THz fields may drive violent vibrational “resonances” in DNA, physically straining and breaking the double helix and then triggering a distinct, p53- and caspase-independent apoptosis pathway.
From Lab Dish to Future Therapies
While these experiments were performed in cell cultures, they hint at a new style of physical cancer therapy. High-field THz waves seem able to penetrate several millimeters into moist tissue—enough to reach superficial tumors—yet lose strength quickly, potentially sparing deeper organs. Because the effect is mainly non-thermal, it could, in principle, kill cancer cells layer by layer without burning surrounding tissue or provoking strong inflammation. Much work remains: the proposed DNA “resonance” mechanism must be confirmed, the safety for normal cells needs thorough testing, and animal and clinical studies are required. Still, this research opens the door to the idea that carefully engineered light pulses, tuned to the natural motions of biomolecules, might one day provide a precise and relatively gentle way to ablate certain cancers.
Citation: Zhou, H., Wei, X., Peng, XY. et al. Efficient apoptosis via double-strand breaks of DNA in human epithelial cells induced by non-thermal effect of high-field broadband terahertz waves. Sci Rep 16, 6163 (2026). https://doi.org/10.1038/s41598-026-37373-6
Keywords: terahertz cancer therapy, non-thermal apoptosis, DNA double-strand breaks, breast cancer cells, physical tumor ablation