Clear Sky Science · en
Longitudinal MR angiographic evaluation of circle of Willis morphologic remodeling and induced aneurysms in Hashimoto rat cerebral aneurysm model
Why tiny bulges in brain arteries matter
Most of us never think about the small arteries at the base of the brain—until one of them bursts. When a brain aneurysm ruptures, it can cause a sudden, often deadly stroke called a subarachnoid hemorrhage. Doctors would like to predict which aneurysms are likely to grow and rupture, but that requires watching them form and change over time, something that is impossible to do directly in people. This study uses high-resolution MRI in rats to follow brain arteries for weeks, offering a rare moving picture of how blood vessels remodel under stress and how aneurysms begin, grow, and sometimes rupture.
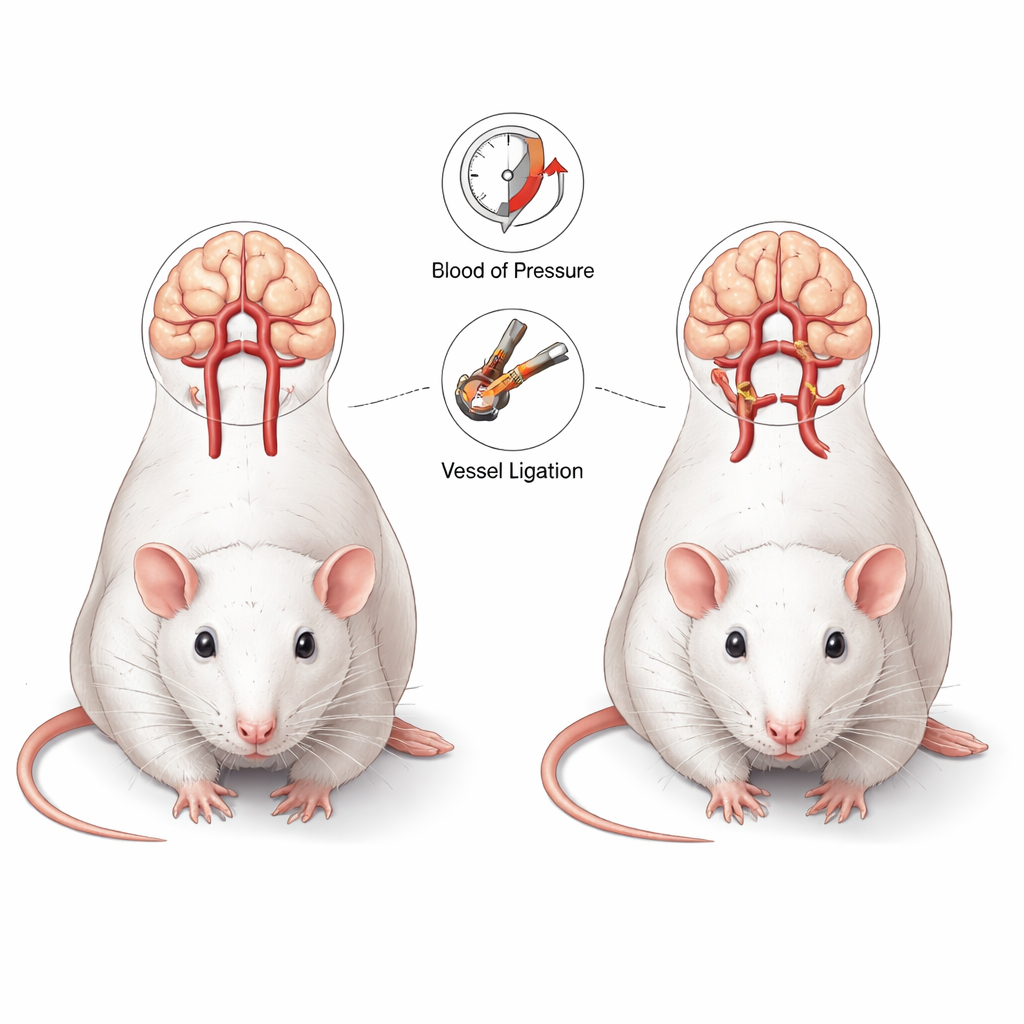
Building a living model of brain vessel stress
The researchers used a classic rat model that imitates important features of human brain aneurysms. In these animals, surgeons tied off one carotid artery in the neck and one kidney artery, then added a high-salt diet and a drug that weakens the vessel wall. Together, these changes raise blood pressure and make arteries more fragile, forcing blood to reroute through the Circle of Willis—a ring of arteries that feeds the brain. Thirteen rats underwent this “aneurysm induction” and six served as controls. All animals received repeated scans in a powerful 7‑Tesla MRI scanner before surgery and up to 12 weeks afterward, allowing the team to track each individual rat’s vessels over time rather than just taking one-time snapshots.
Watching brain arteries reshape themselves
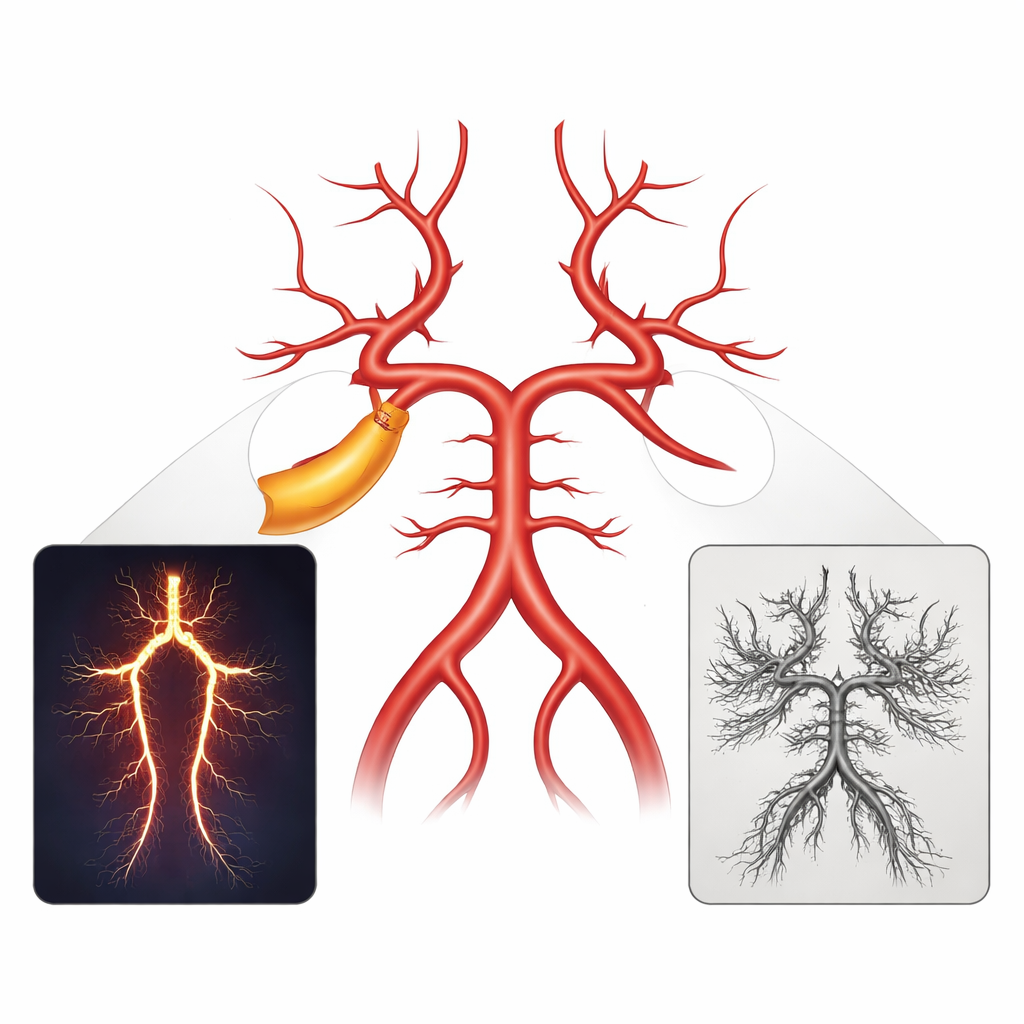
The MRI scans showed that even one week after surgery, the Circle of Willis was already changing shape in the stressed rats. Some arteries became wider, and their paths grew more twisted, especially on the side where the carotid artery had been tied off. One key artery in the back of the brain, the left posterior cerebral artery, enlarged much more than its partner on the right, reflecting a shift in blood flow. Other vessels at the front of the brain also widened as they tried to share and reroute blood. In contrast, the control rats, which did not undergo the full stress procedure, kept symmetric, stable vessel shapes throughout the 12‑week period. By measuring diameters and a “tortuosity index” that captures how winding a vessel is, the team showed that these remodeling patterns were not random but followed clear, time‑dependent trends.
From remodeling to dangerous bulges
As the weeks passed, some of these remodeled arteries developed small bulges—aneurysms—while others went on to rupture, causing bleeding around the brain. Using MRI, the researchers saw signs of aneurysm-related events in nearly half of the stressed rats, including three cases of overt brain hemorrhage. However, when they later created detailed casts of the blood vessels and examined them under a scanning electron microscope, they found more aneurysms than the MRI had revealed. Many of these were extremely small, often just tenths of a millimeter across, and tended to cluster at vessel branch points. Two lesions in a particular back‑of‑the‑brain segment ballooned into large, elongated (fusiform) aneurysms that ultimately ruptured. This pattern suggests that not only where a vessel sits in the network, but also how it carries the extra blood load, influences whether it quietly adapts or fails catastrophically.
How well can MRI see the smallest threats?
Because the study combined live imaging with microscopic postmortem analysis, it could directly test how good MRI is at spotting aneurysms in this small‑animal setting. The answer was mixed. The MRI sequences were excellent for following overall vessel widening and bending, and for spotting larger aneurysms and hemorrhages over time. But they missed many microaneurysms that were below the scanner’s practical resolution. In this experiment, MRI correctly identified only about 40 percent of confirmed aneurysms and produced some false alarms, often when a very twisted artery or overlapping tiny branches mimicked a bulge. These findings highlight both the power and the current limits of noninvasive imaging when structures approach the size of a grain of sand.
What this means for future stroke prevention
For a lay reader, the key message is that brain arteries are dynamic: under sustained high blood pressure and altered flow, they do not simply stretch uniformly, but remodel in complex, uneven ways. This rat model, paired with high‑resolution MRI, gives scientists a way to watch those changes unfold in living brains, linking vessel shape, blood pressure, and eventual aneurysm behavior. Although today’s scanners cannot reliably see the tiniest danger spots, this work shows how improved imaging and careful animal models can help identify which vessel segments are most at risk and how early changes might foreshadow rupture. In the long run, insights from such studies could guide better screening, more precise risk estimates, and targeted therapies to prevent devastating brain hemorrhages before they occur.
Citation: Kim, Y.S., Hwang, S., Kim, M.H. et al. Longitudinal MR angiographic evaluation of circle of Willis morphologic remodeling and induced aneurysms in Hashimoto rat cerebral aneurysm model. Sci Rep 16, 7094 (2026). https://doi.org/10.1038/s41598-026-37369-2
Keywords: cerebral aneurysm, brain blood vessels, MRI angiography, vascular remodeling, stroke risk