Clear Sky Science · en
Uncovering the potential MTAs, candidate genes and microRNAs regulatory networks involved in salinity stress tolerance triggered in Iranian Aegilops tauschii
Why salty soils threaten our daily bread
As climate change spreads drought and irrigation water becomes saltier, vast areas of farmland are turning too salty for ordinary wheat. This matters for everyone who relies on bread as a staple food. The study summarized here looks to a wild grass, Aegilops tauschii, which is a direct ancestor of modern bread wheat, to uncover the hidden genetic tools—and tiny regulatory molecules—that help plants cope with salty conditions. By mapping these natural defenses, the researchers hope to give breeders new ways to develop wheat varieties that stay productive even when the soil turns brackish.
Wild wheat cousin as a hidden resource
Aegilops tauschii grows naturally across the Fertile Crescent, including Iran, where it has adapted over thousands of years to harsh, dry, and often salty environments. This species donated the “D” portion of the genome that modern bread wheat carries today. Because today’s high-yield wheat varieties were bred from a relatively narrow genetic base, they often lack the full range of stress-tolerance traits that still exist in their wild relatives. The authors assembled 77 Iranian Aegilops tauschii ecotypes (local forms) and grew them under both normal and salty conditions at the seedling stage, measuring features like root and shoot length, fresh and dry weight, and leaf and root area. Salinity sharply reduced all of these traits, confirming that high salt levels are indeed damaging for young plants.
Reading the DNA fingerprints of salt tolerance
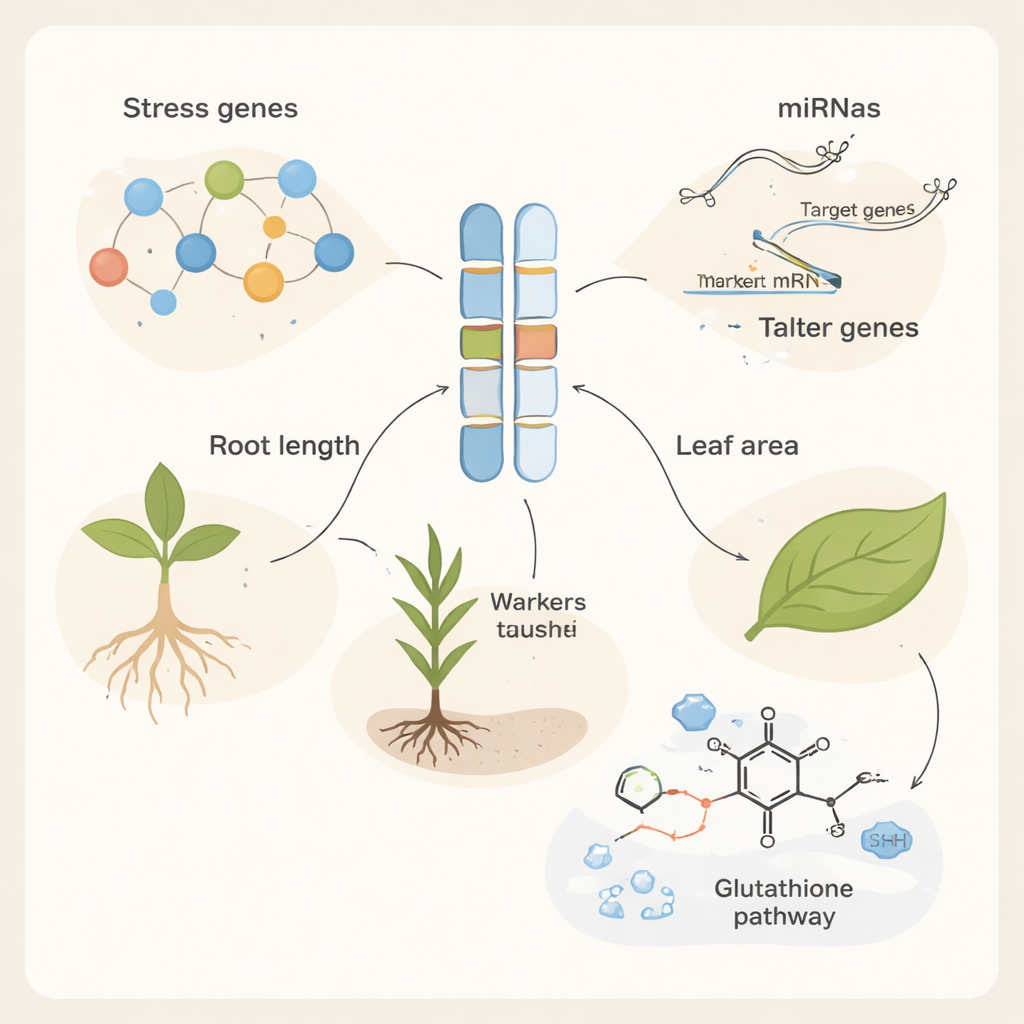
To understand why some wild plants handled salt better than others, the team turned to DNA markers—short, easily measured sequences scattered across the genome that act like barcodes for nearby genes. Using a combination of “random” markers and semi-random RAMP markers, they scored hundreds of DNA bands and assessed how diverse the collection really was. They found high levels of genetic variation, with certain marker systems, such as ISJ9 and OPE03-Xgwm44-7DF, providing especially strong power to distinguish one ecotype from another. This rich diversity means Iranian Aegilops tauschii still harbors many unique genetic variants that breeders can tap into. By statistically linking particular DNA bands to seedling traits under salt stress, the researchers identified 115 marker–trait associations, pointing to genomic regions that influence how well roots, shoots, and leaves develop in salty environments.
From markers to working stress-defense genes
Finding a useful DNA marker is only the first step; next the authors asked which actual genes sit near those markers and might be doing the work. Using the wheat reference genome, they searched 500,000 base pairs around each associated marker and discovered 254 candidate genes. Many of these genes were independently confirmed by large RNA sequencing datasets to switch their activity up or down when plants faced environmental stress, including cold, heat, nutrient shortage, and disease. The candidate genes were enriched for roles in defense responses and protection from heat and oxidative damage. Several encode proteins such as disease resistance receptors, heat-shock chaperones, and so-called thaumatin- and osmotin-like proteins, which help stabilize cells when salt causes water loss and harmful reactive oxygen molecules to build up. Pathway analysis highlighted glutathione metabolism, a core chemical system plants use to detoxify stress-induced by-products.
Tiny RNA switches that fine-tune stress responses
Genes do not act alone; they are controlled by microRNAs, very short RNA molecules that can shut down or dampen gene activity. The researchers predicted which wheat microRNAs might target their candidate genes and uncovered 107 distinct microRNAs forming dense regulatory networks. Many of these tiny regulators were already known to respond to drought, heat, metals, or salinity. For example, specific microRNAs previously linked to salt tolerance, heat tolerance, or “stress memory” were found to control key defense and detoxification genes identified in this study. Some genes were targeted by multiple microRNAs, suggesting that plants layer several regulatory switches on top of one another to finely adjust their response as stress levels change.
Bringing wild resilience into farmers’ fields
Together, the catalog of diverse DNA markers, 254 candidate genes, and 107 regulatory microRNAs offers a roadmap for improving wheat performance on salty soils. Breeders can turn the most informative markers into simple lab tests to screen large numbers of plants for hidden salt-tolerance traits, a strategy known as marker-assisted selection. In the longer term, the most promising genes and their microRNA regulators could be deliberately introduced or edited into sensitive wheat varieties to strengthen their natural defenses. Although the study still calls for experimental validation under salinity in living plants, it clearly shows that the wild relative Aegilops tauschii holds powerful genetic tools that, if thoughtfully used, may help secure wheat harvests as salinity and climate pressures intensify.
Citation: Sabouri, H., Nikkhah, N., Kazerani, B. et al. Uncovering the potential MTAs, candidate genes and microRNAs regulatory networks involved in salinity stress tolerance triggered in Iranian Aegilops tauschii. Sci Rep 16, 6877 (2026). https://doi.org/10.1038/s41598-026-37365-6
Keywords: salinity tolerance, wheat breeding, Aegilops tauschii, stress-responsive genes, plant microRNAs