Clear Sky Science · en
Generalized blood vessel models for magnetic nanoparticle-based oncology: geometric and microfluidic properties
Guiding Tiny Magnets Through the Body
Imagine being able to pull cancer drugs directly into a tumor using a magnet, sparing the rest of the body from harsh side effects. This study explores how tiny magnetic particles travel through blood-vessel-like networks and asks a deceptively simple question: under realistic flow conditions, can magnets actually steer these particles where doctors want them to go?
Why Vessel Geometry and Flow Matter
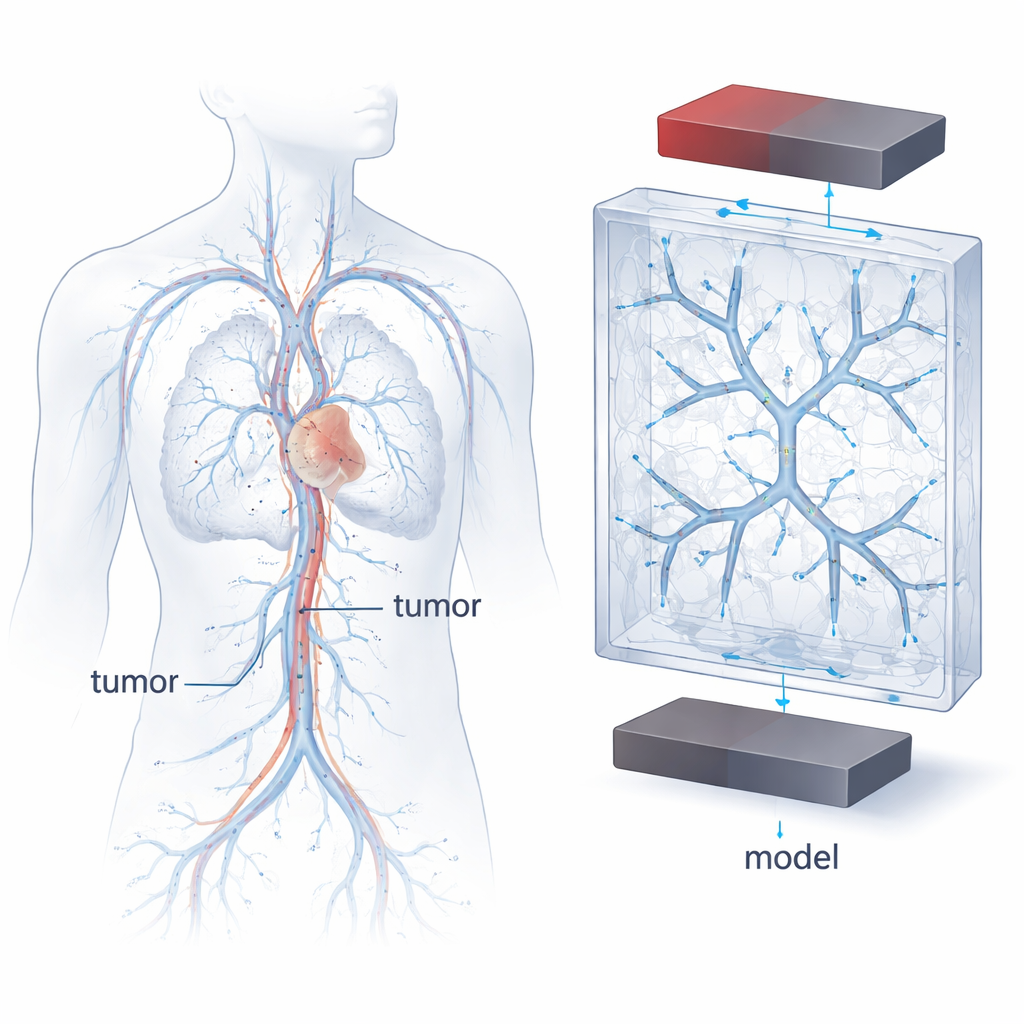
Magnetic cancer therapies rely on superparamagnetic iron oxide nanoparticles—SPIONs—that can be tugged by a magnetic field while they carry drugs through the bloodstream. But blood vessels are not simple straight tubes: they branch, narrow, curve and split, and blood speeds up or slows down in response. These changes in shape and speed affect how particles move, where they concentrate and whether they touch vessel walls long enough to stick. Real patient blood vessels are extremely variable, which makes it difficult to compare experiments or to predict how a new therapy will behave. The authors therefore set out to build a highly controlled, repeatable “model vasculature” that still captures the essential features of tumor-feeding blood vessels.
Building Idealized Tumor Blood Networks
To design these model vessels, the team drew on classic rules that describe how real arteries branch. One such rule, Murray’s law, links the diameters of parent and daughter vessels in an energy-efficient network. Using clinical measurements from breast tumors and these scaling laws, the researchers mathematically generated branching trees that mimic how tumor-feeding vessels shrink down toward the capillary bed. They then used high-resolution 3D printing to embed hollow channel networks with two, three or four branching levels inside clear plastic blocks. The channels followed a symmetric branching pattern and extended in three dimensions, offering a simplified but physiologically inspired landscape in which to study particle motion.
Watching Magnetic Particles in Action
Through these printed networks, the team pumped water as a stand-in for blood, then injected SPION-containing fluid upstream and applied a static magnetic field around the model. High-speed imaging provided a movie-like record of how the dark particle clouds passed through different branches, while computer analysis converted brightness changes into time-resolved signals that reflect local particle presence. By varying background flow rate, particle concentration, branching complexity and the presence or absence of a magnet, the researchers could tease apart how each factor shaped particle distribution, residence time and eventual deposition on channel walls.
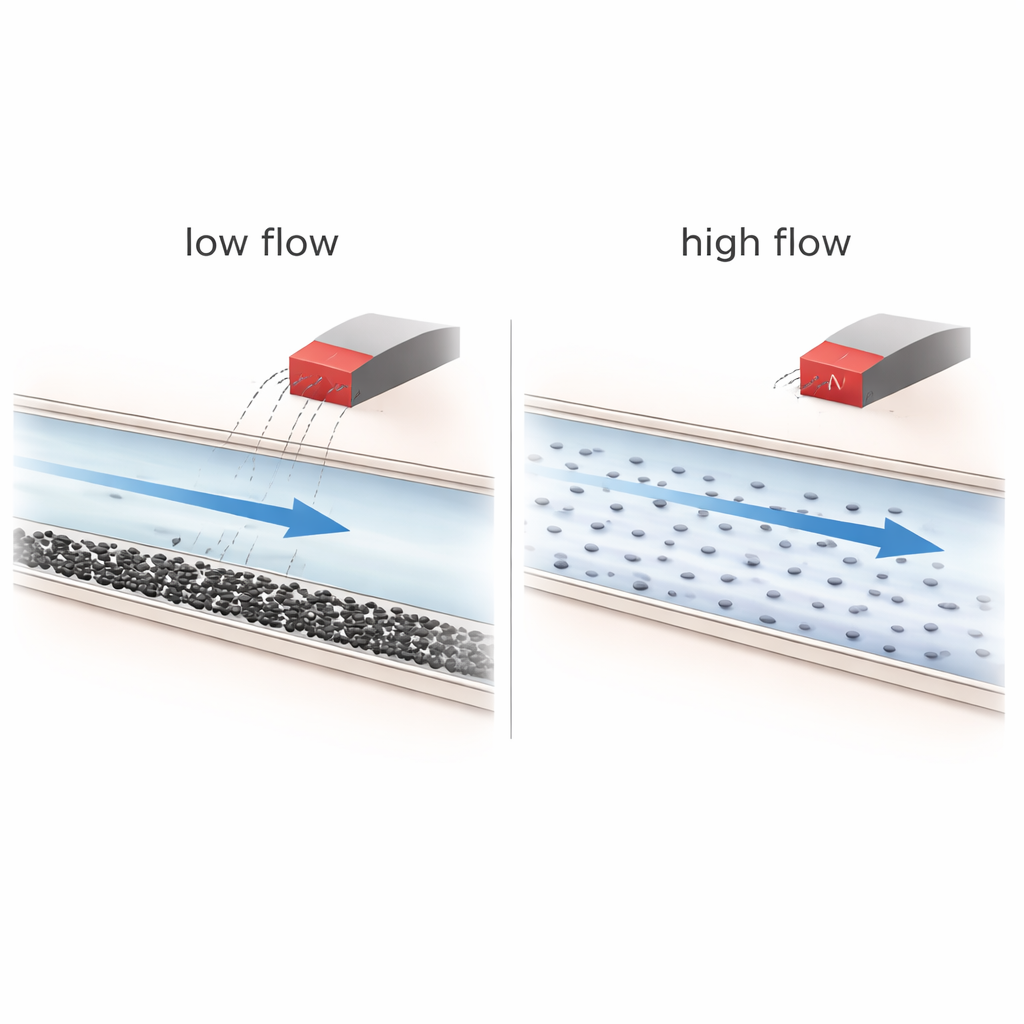
When Magnets Help—and When They Don’t
The experiments reveal that flow conditions dominate the story. At higher flow rates, the fluid becomes well mixed and the nanoparticles travel quickly; under these conditions, the magnetic field has almost no influence, and particles simply follow the streamlines through the network. At low flow rates, however, gravity and concentration effects cause particles to settle toward the bottom of the channels, forming stratified layers and sometimes loose clusters. In this already uneven situation, the magnet can nudge more particles into certain branches and slightly prolong their residence, particularly in more complex, highly branched networks. Yet calculations show that the magnetic force on a single 40-nanometer particle is many orders of magnitude weaker than the viscous drag from the flowing liquid, making true “single-particle steering” physically unrealistic under the tested conditions.
What This Means for Future Cancer Therapies
To a non-specialist, the main takeaway is that magnets alone cannot reliably pull freely suspended nanoparticles out of a fast-moving bloodstream. Instead, noticeable magnetic effects arise only when particles have already slowed, settled or clustered—situations that effectively magnify their magnetic response. The generalized, 3D-printed vessel models introduced here provide a standardized way to explore such effects across different network geometries and flow regimes. For future magnetic drug targeting, this work suggests that successful strategies will likely need to exploit collective behaviors—such as controlled aggregation or carrier systems—rather than relying on steering isolated nanoparticles through large, rapidly perfused vessels.
Citation: Fleischhauer, D., Schlicht, S. & Drummer, D. Generalized blood vessel models for magnetic nanoparticle-based oncology: geometric and microfluidic properties. Sci Rep 16, 3701 (2026). https://doi.org/10.1038/s41598-026-37348-7
Keywords: magnetic drug targeting, nanoparticles, tumor vasculature, microfluidic models, drug delivery