Clear Sky Science · en
A tri-omics and machine learning framework identifies prognostic biomarkers and metabolic signatures in sepsis
Why this matters for people with severe infections
Sepsis is a life-threatening reaction to infection that can send the immune system into overdrive and cause organs to fail. Doctors know that catching sepsis early and tailoring treatment to each patient can save lives, but today’s blood tests are blunt tools: they often say little about who will get better and who is at highest risk. This study uses a powerful combination of three types of molecular measurements and modern machine learning to search for more precise warning signals in the blood of sepsis patients.
Looking at blood through three different lenses
Instead of focusing on a single kind of molecule, the researchers profiled the same patients in three ways at once. They measured which genes were switched on or off (transcriptomics), which proteins were actually present and active (proteomics), and which small metabolic molecules were circulating (metabolomics). They collected blood from 21 sepsis patients and 10 healthy volunteers and used advanced statistics to see how these three layers changed together in disease. This "tri-omics" view helps overcome a key problem: in sepsis, gene activity and protein levels can become uncoupled, so looking at only one layer can be misleading.
Teaching algorithms to spot high‑risk patterns
From thousands of genes and proteins, the team first used a network method to find groups that moved together in sepsis. They then cross-checked those groups with proteins that clearly differed between patients and healthy controls, ending up with 32 strong candidates. To narrow this list further, they turned to machine learning, using two complementary algorithms to strip away weaker signals and keep only the most informative ones. When they tested how these remaining genes related to survival in a large public sepsis dataset, two stood out: TPR and ERN1. Patients with higher TPR levels tended to survive longer, while higher ERN1 was linked to worse outcomes.
Linking immune cells and disturbed metabolism
The study did not stop at genes and proteins. By scanning thousands of metabolites in patient blood, the researchers found 136 small molecules that tracked closely with TPR and ERN1. Many belonged to pathways that handle cell membrane fats and fatty acids, which are key to how immune cells signal and how inflammation spreads. At the same time, a single-cell analysis — which examines individual blood immune cells rather than blending them together — showed that TPR and ERN1 are especially active in monocytes, macrophages, and natural killer cells. Together, these results suggest that the two markers sit at the crossroads between the body’s infection-fighting cells and the way those cells use and reshape fats and energy during sepsis.
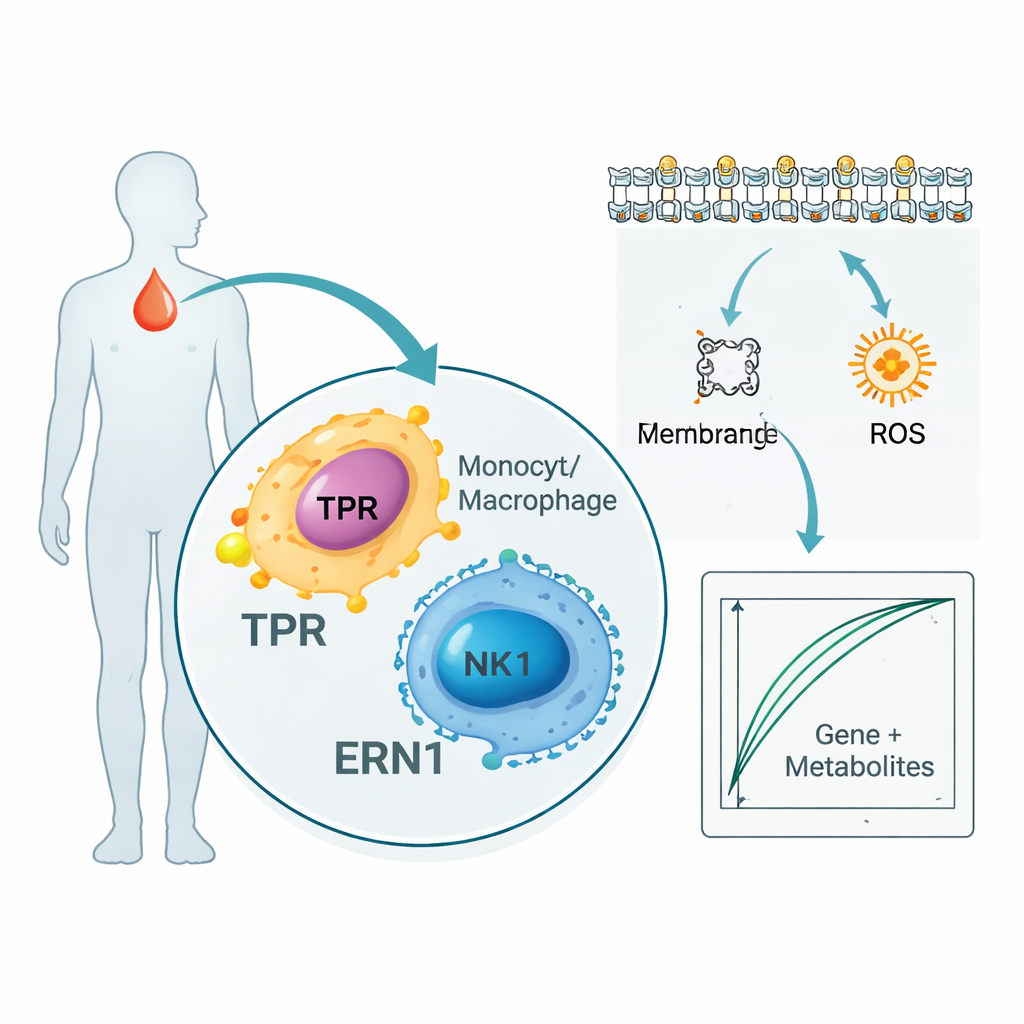
Building a proof‑of‑concept blood test
To explore how these discoveries might translate into practice, the authors combined the two genes with five of the most informative metabolites to train simple computer models that separated sepsis patients from healthy people. In their small internal dataset, these combined "gene-plus-metabolite" signatures could almost perfectly tell who had sepsis. The researchers also checked large public databases that link blood proteins to disease risk in tens of thousands of people and found that TPR and ERN1 protein levels were consistently associated with sepsis-related conditions, adding another layer of support. Still, the authors stress that these models are early-stage tools designed to generate hypotheses, not ready-made bedside tests.
Plant compounds as early leads, not cures
In a final step, the team asked whether any natural molecules might influence TPR or ERN1. They searched a specialized database of nearly 500 purified compounds from traditional Chinese medicines, each with its own gene activity profile. Several compounds appeared to strongly push these two genes up or down in lab-grown cells, hinting that they might one day help researchers probe the biology of sepsis or design new drugs. However, these findings come from computer matching only: they do not show that any of these substances are safe or effective for people with sepsis.
What this work really tells us
This study offers a detailed map rather than a finished solution. By weaving together three molecular layers, single-cell data, and machine learning, the authors highlight TPR and ERN1 — and their connected metabolic changes — as promising signposts of how the immune system and metabolism become unbalanced in sepsis. For a layperson, the key message is that sepsis is not a single disease but a shifting pattern of immune and metabolic states, and that smarter blood tests might one day help doctors see which state a patient is in and adjust treatment accordingly. Before that happens, these early signals must be tested and confirmed in much larger, diverse patient groups and in laboratory experiments that can prove cause and effect.
Citation: Li, X., Ke, G., Hu, Y. et al. A tri-omics and machine learning framework identifies prognostic biomarkers and metabolic signatures in sepsis. Sci Rep 16, 6648 (2026). https://doi.org/10.1038/s41598-026-37342-z
Keywords: sepsis biomarkers, multi-omics, machine learning in medicine, immune metabolism, precision diagnostics