Clear Sky Science · en
Harmonization of IgG antibody values against the SARS-CoV-2 Spike protein
Why this matters for everyday protection
As the world continues to live with COVID-19, it is vital to understand how well vaccines protect people over time and how booster doses renew that protection. Many hospitals measure levels of antibodies—blood markers of the immune response—to track this. But because different hospitals often use different machines and tests, their results can be like temperatures reported in mixed scales: hard to compare and easy to misinterpret. This study shows a new way to “translate” these differing antibody results onto a common scale, allowing clearer answers about how long vaccine protection lasts and how strongly boosters reinforce it.
Following hospital workers over time
The researchers followed more than 3,000 healthcare workers in three Portuguese hospitals between 2020 and 2022. This group was chosen because hospital staff face frequent exposure to SARS-CoV-2 and were among the first to be vaccinated. Blood samples were collected at six key moments: before vaccination, soon after the initial vaccine series, 3, 6 and 12 months after the second dose, and after a booster dose. In each hospital, antibody levels against the virus’s Spike protein—a key target of COVID-19 vaccines—were measured using different commercial lab tests. These tests all look at the same kind of antibodies but report values on incompatible scales, which normally makes it difficult to combine their results into a single picture of immunity over time.
Why standard conversion was not enough
To help labs worldwide compare results, the World Health Organization (WHO) introduced a common unit for antibody measurements. In theory, each manufacturer provides a simple factor to convert their test’s result into this shared unit, much like converting inches to centimeters. The Portuguese team tried this standard conversion first. However, when they applied it to their data, the pattern of antibody levels over time looked odd. After the initial rise following vaccination, levels dipped at three months, then unexpectedly increased again at six months before dropping by twelve months. This behavior does not fit what is seen in most other studies, where antibody levels peak soon after vaccination and then steadily decline until a booster is given. The mismatch suggested that relying only on the WHO factors might blur or distort real biological trends.
A smarter way to line up different tests
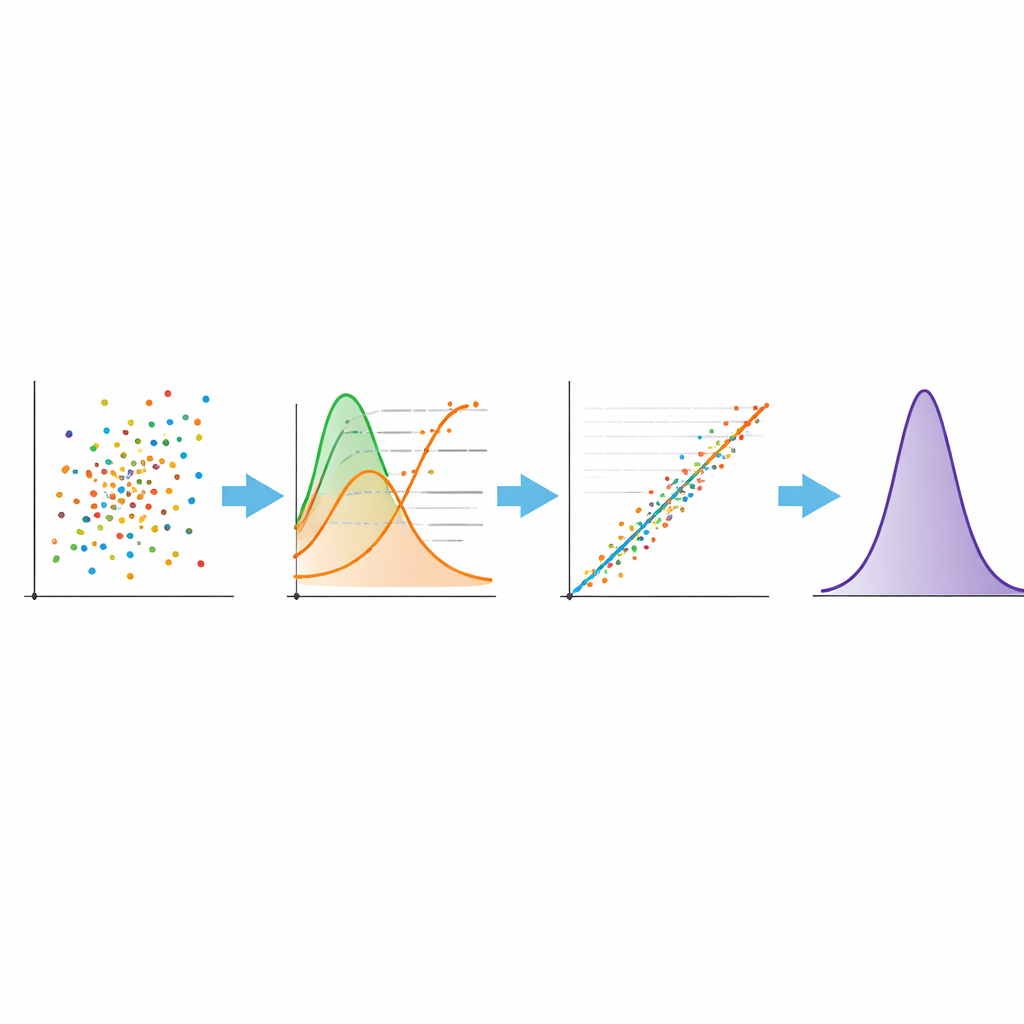
To address this, the team developed a more refined mathematical approach to “harmonize” the results from the three hospitals. First, they adjusted each hospital’s data so that the shapes of the distributions could be compared fairly, using transformations that make skewed values more evenly spread out. Then they ranked the measurements and matched them by quantiles—pairing, for example, the 10th, 50th or 90th percentile from one test with the same percentile from another. This step aligns the overall spread of values across tests rather than forcing a one-size-fits-all conversion factor. Finally, they applied a special kind of regression, called Deming regression, which takes into account that both tests have measurement error, to derive equations that map each hospital’s results onto the reference test used in one center. After reversing the transformations, they obtained formulas that can convert any raw value from one hospital into the comparable value on the chosen reference scale.
What the harmonized data reveal about boosters
Once this harmonization method was applied, the antibody patterns over time became much more biologically plausible. Levels rose sharply after the first vaccine series, then gradually declined over 3, 6 and 12 months, reflecting the natural waning of antibody responses seen in other research. Just before the booster, the typical (geometric mean) level on the reference scale was around 660 units, but after the booster it soared to roughly 26,000 units—far above the levels reached after the second dose. This large jump is consistent with other studies of vaccinated healthcare workers and confirms that booster shots can strongly re-energize immune memory, at least in the short term. Importantly, the refined harmonization preserved the characteristic behavior of each hospital’s data, rather than forcing them into an artificially smooth pattern.
Limits, lessons, and future use
The authors note some important caveats. They did not run the exact same blood samples on all three lab platforms, which would have provided a more direct test of how closely the harmonized values match true differences. They also had to choose one test as the reference scale even though no perfect “gold standard” exists. As a result, the harmonized numbers should be viewed as a way to compare trends and relative levels rather than as precise, absolute measures of protection for any single person. Still, by carefully checking that the method did not introduce new artifacts and that the overall antibody dynamics stayed consistent with known immunology, they showed that this approach is practical and robust for large real-world datasets.
What this means for public health
For non-specialists, the key message is that the way we handle numbers from different tests can profoundly affect what we think vaccines are doing. By developing a more reliable way to translate antibody measurements from many hospitals onto a common scale, this study helps researchers and health agencies build clearer, more trustworthy pictures of how immunity changes over time and how well booster doses are working. Such harmonized data are especially valuable for international collaborations, where studies must integrate results from many labs and countries. While further technical validation is still needed, the method presented here offers a promising tool for tracking COVID-19 vaccine performance and planning future booster campaigns.
Citation: Saraiva, A.L., Afreixo, V., Amaral, P. et al. Harmonization of IgG antibody values against the SARS-CoV-2 Spike protein. Sci Rep 16, 9522 (2026). https://doi.org/10.1038/s41598-026-37341-0
Keywords: COVID-19 antibodies, vaccine boosters, laboratory harmonization, healthcare workers, SARS-CoV-2 Spike protein