Clear Sky Science · en
Integrated safety and microbiota profiling of fulvic acid formulations across in vitro and in vivo models
Ancient Soil Compounds with Modern Health Promise
What if a substance formed from decaying plants over millions of years could quietly support your gut, your immune system, and even help tissues repair themselves? This study explores fulvic acid—an organic component of humic substances found in soils and peat—as a potential “multi-tool” supplement. Using lab-grown human and animal cells, as well as guinea pigs, the researchers asked a simple but crucial question: can carefully prepared fulvic acid liquids be both safe and genuinely helpful for our cells and our gut microbes?
What Fulvic Acid Is and Why It Matters
Fulvic acid is a mix of small, water‑soluble molecules produced as microbes slowly break down plant and animal material. Because of its rich chemistry, earlier work hinted that it can act as an antioxidant, help control inflammation, aid wound healing, and influence microbes in soil, animals, and people. However, past studies often looked at just one effect at a time, using different, poorly comparable preparations. In this work, scientists focused on two standardized, fossil‑derived liquid formulations, called MLG‑50 (acidic) and MLG‑A50 (alkaline). Both come from an ancient organic‑rich deposit in the southern United States and contain fulvic acid along with minerals like magnesium, calcium, sodium, potassium, and sulfur. The team set out to test these formulations in a coordinated way: checking safety for mammalian cells, potential DNA damage, effects on inflammation, tissue repair, and changes in gut bacteria in vitro and in vivo.
Safety First: Effects on Cells and Their DNA
To understand safety, the researchers exposed mouse skin cells and human intestinal and liver cell lines to a wide range of fulvic acid concentrations. At very strong (barely diluted) doses, the acidic formulation MLG‑50 reduced cell viability, but at higher dilutions—within ranges that would be relevant for supplements—both MLG‑50 and MLG‑A50 were well tolerated, and in some cases slightly boosted cell activity. They then checked for DNA damage using a sensitive “comet” assay that detects broken DNA strands in individual cells. While the fulvic acid preparations produced a tiny increase in measurable damage compared to untreated cells, levels stayed far below standard thresholds used to flag a compound as genotoxic. In fact, common reference compounds such as vitamin C and aspirin caused much higher DNA damage under the same conditions, suggesting that these fulvic formulations sit comfortably within typical safety margins.
Helping Cells Grow, Close Wounds, and Calm Inflammation
Beyond simple survival, the team examined whether fulvic acid could help intestinal cells grow and repair. In proliferation tests, diluted MLG‑50 and especially MLG‑A50 temporarily increased the number of intestinal epithelial cells over the first several days, compared with untreated cultures and with reference fulvic standards that showed little effect. In a “scratch” wound‑healing assay, where a gap is made in a sheet of cells to mimic tissue injury, MLG‑A50 stood out: it sped up early wound closure and maintained this advantage over 72 hours, pointing to a pro‑regenerative effect. The researchers then turned to the immune system, using monocyte‑like cells that switch on a key inflammatory switch called NF‑κB when exposed to bacterial endotoxin (LPS). On their own, the fulvic formulas did not trigger this alarm system—which is good news from a safety standpoint. But when cells were treated with LPS plus fulvic acid, NF‑κB activation dropped by around 60%, and production of powerful inflammatory messengers TNF and IL‑6 fell by about half, while levels of the calming cytokine IL‑10 were preserved or even increased. This pattern suggests that fulvic acid does not shut the immune system down; instead, it seems to dampen excessive reactions while leaving protective regulation intact.
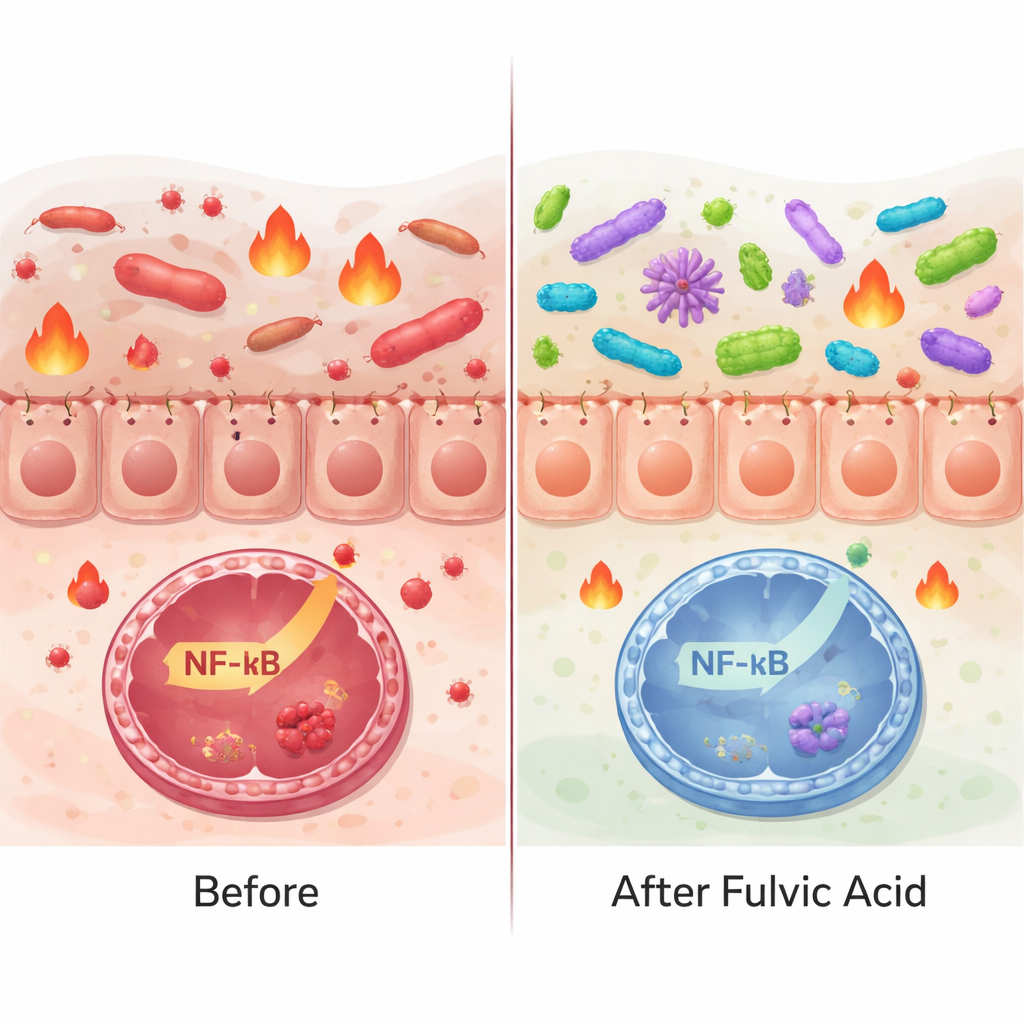
Feeding Friendly Microbes While Holding Pathogens in Check
Because fulvic acid can bind minerals and influence redox (electron‑transfer) reactions, it might also shape microbial communities. In test‑tube experiments, MLG‑50 markedly boosted the growth and metabolic activity of probiotic Lactobacillus strains, while leaving disease‑linked bacteria such as Escherichia coli and Salmonella either unchanged or slightly suppressed. MLG‑A50 showed a milder but still selective support for beneficial strains. To see what happens in a living gut, guinea pigs were given low doses of the two formulations for three weeks. DNA sequencing of fecal samples revealed that overall microbial diversity increased—a feature often associated with a more resilient gut. Some groups of bacteria linked to gut health, such as certain Clostridia and relatives of Lactobacillus, trended upward, whereas potentially problematic genera like Helicobacter and Campylobacter decreased significantly. These in vivo findings echo the selective “pro‑good, anti‑bad” effects seen in vitro and suggest that fulvic acid can gently remodel the gut ecosystem rather than simply acting as a broad antimicrobial.
What This Could Mean for Future Health Uses
Taken together, the study paints fulvic acid not as a miracle cure, but as a promising, multi‑purpose helper. At appropriate dilutions, the tested formulations showed low toxicity and minimal DNA damage, encouraged intestinal cell growth and wound closure, toned down excessive inflammatory signals while keeping protective ones, and nudged gut microbes toward a more diverse, probiotic‑friendly balance. For lay readers, this suggests that carefully characterized fulvic acid products could one day serve as part of “immunonutrition” strategies—supporting the gut barrier, the microbiome, and balanced immunity, especially in chronic inflammation or recovery settings. The authors stress that more mechanistic work and human trials are still needed, particularly to fine‑tune dosing and long‑term safety. But the integrated evidence from cells and animals makes a compelling case that certain fulvic acid formulations deserve serious consideration as future tools for gentle, whole‑system support.
Citation: Szwed-Georgiou, A., Płociński, P., Włodarczyk, M. et al. Integrated safety and microbiota profiling of fulvic acid formulations across in vitro and in vivo models. Sci Rep 16, 6166 (2026). https://doi.org/10.1038/s41598-026-37331-2
Keywords: fulvic acid, gut microbiome, anti-inflammatory, probiotics, wound healing