Clear Sky Science · en
Sustainable wastewater treatment by banana peel/layered double hydroxide composite under ideal conditions using the Taguchi method
Turning Kitchen Waste into Clean Water
What if the banana peels left over from breakfast could help clean polluted rivers? This study explores exactly that idea. The researchers show that discarded banana peels, combined with a special layered mineral, can pull a toxic purple dye out of wastewater. By transforming agricultural waste into a powerful cleaning material, their work points toward cheaper, more sustainable ways to deal with industrial pollution, especially in regions where advanced treatment plants are too costly.

Why Colored Wastewater Is a Problem
Modern industries—from textiles and paints to biotechnology—use synthetic dyes to give products vivid and lasting colors. One of these dyes, crystal violet, is particularly troublesome. It does not break down easily in nature, blocks sunlight in rivers and lakes, and is known to be harmful to living cells. Traditional methods to remove such dyes, like advanced filters or chemical treatments, can be expensive, energy-hungry, and may generate new types of waste. This has pushed scientists to look for low-cost, nature-based materials that can soak up dyes before they reach the environment.
From Banana Peel to Cleaning Material
Banana peels are usually thrown away, even though they are rich in natural fibers and chemical groups that can stick to pollutants. In this work, the team first washed and treated banana peels with a simple alkaline solution, then dried and ground them into a powder. They also made a "layered double hydroxide"—a stack of ultra-thin mineral sheets containing nickel, calcium, and iron. Finally, they combined the two to form a banana peel/mineral composite. Microscopy and other tests showed that the mineral sheets spread across the rough banana surface, creating a highly textured material full of pores and active sites where dye molecules could attach.
Finding the Best Conditions
To understand how well these materials clean water, the researchers mixed them with water containing crystal violet and adjusted conditions such as acidity (pH), contact time, temperature, and how much adsorbent they added. They used a structured statistical approach, known as the Taguchi method, to squeeze the most information out of just nine carefully planned experiments. For both plain banana peel and the composite, the acidity of the water turned out to matter most: the dye was removed much more efficiently under mildly alkaline conditions, where the surface of the material carries a negative charge that strongly attracts the positively charged dye molecules. With an optimal pH of 9, a modest dose of material, and two hours of contact, the composite removed about 95% of the dye—much more than banana peel alone or the mineral alone.
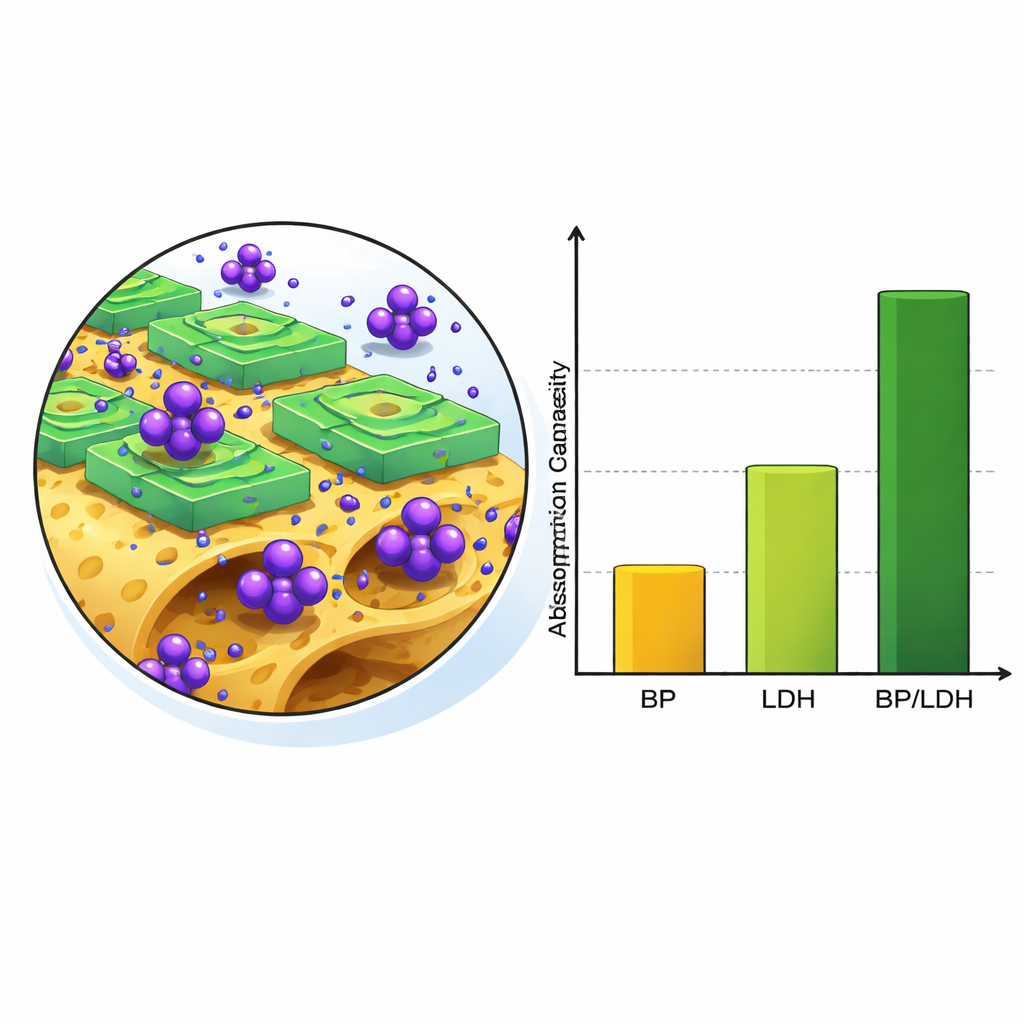
How the Composite Grabs the Dye
Closer inspection of the used material revealed how the new adsorbent traps the dye. After treatment, its once-open pores became filled or covered, and chemical fingerprints showed that new bonds formed between dye molecules and the surface groups of the composite. The dye sticks in several ways at once: through opposite electrical charges, through hydrogen bonds, and through stacking of flat dye rings against the aromatic structures in the banana peel fiber. The layered mineral portion contributes extra surface area and sites where the dye can lodge. Tests comparing different mathematical models of uptake over time suggest that the process is largely controlled by these strong, specific interactions rather than by simple loose sticking.
Reusable, Low-Cost, and Ready to Scale
An important question for any real-world water treatment is whether the cleaning material can be used more than once. The authors repeatedly loaded the composite with dye and then rinsed it with ethanol to release the color. After four cycles, it still removed about 80% of the dye, indicating solid structural stability and reasonable long-term performance. When they compared it to a range of other plant-based adsorbents reported in the literature, the banana peel composite offered one of the highest dye uptake capacities while relying on a nearly free raw material and simple preparation steps. A rough cost analysis suggests that it could compete with or even undercut activated carbon, the current workhorse of adsorption filters, especially where banana waste is abundant.
What This Means for Everyday Life
In practical terms, this study shows that something as ordinary as a banana peel can be engineered into a highly effective and reusable filter for toxic dyes. While the work was done in controlled lab solutions, it lays the groundwork for low-cost treatment units that could help small factories or communities clean their wastewater without sophisticated infrastructure. With further testing in real effluents and at larger scales, banana peel–based composites could become part of a broader toolkit that links waste reduction, resource recovery, and cleaner water into a single, sustainable loop.
Citation: Mohamed, H.F.M., Hafez, S.H.M., Abdel-Hady, E.E. et al. Sustainable wastewater treatment by banana peel/layered double hydroxide composite under ideal conditions using the Taguchi method. Sci Rep 16, 7188 (2026). https://doi.org/10.1038/s41598-026-37321-4
Keywords: wastewater treatment, banana peel adsorbent, crystal violet dye, low-cost water purification, sustainable materials