Clear Sky Science · en
Purple LED light and crude glycerol synergistically enhance astaxanthin production in Aurantiochytrium limacinum
Why glowing microbes could matter to you
Colorful compounds from the microscopic world are quietly reshaping how we make nutritional supplements, cosmetics, and even fish feed. This study explores how a tiny marine organism, Aurantiochytrium limacinum, can be coaxed with purple LED light and cheap industrial byproducts to produce high-value molecules like astaxanthin—a powerful red antioxidant also found in salmon and shrimp—alongside healthy omega-3 oils such as DHA. The work points toward more sustainable, less expensive ways to make these ingredients without relying on wild fish or energy-hungry chemical factories.
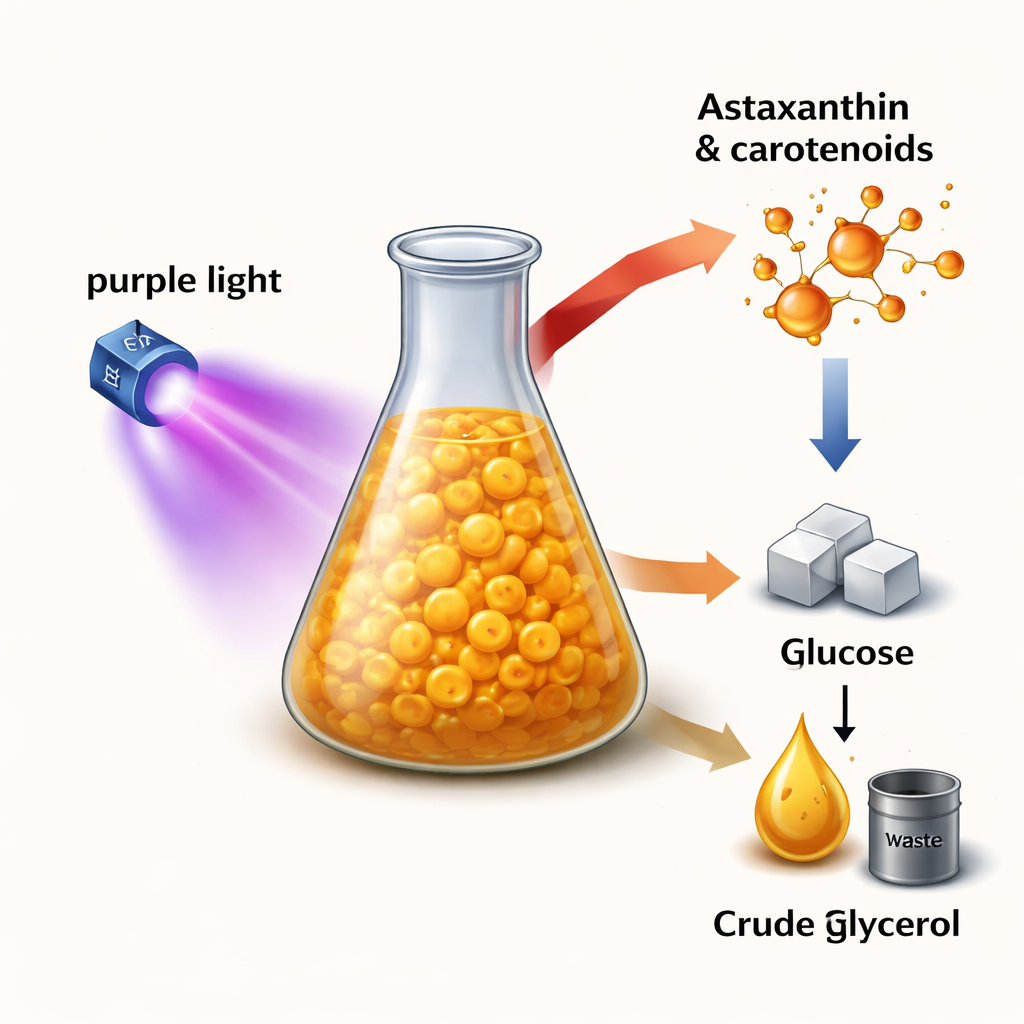
A tiny factory from the sea
Aurantiochytrium limacinum is a microscopic marine protist that thrives in the dark and feeds on organic carbon rather than sunlight. It is attractive to industry because it can make two valuable products at once: DHA, an omega‑3 fat important for brain and heart health, and carotenoids, the pigments that give many plants and animals their yellow, orange, and red colors. Among these carotenoids, astaxanthin stands out for its strong antioxidant and anti‑inflammatory properties, which have led to growing demand in supplements, functional foods, and skincare. Until now, most astaxanthin has come either from fish oil or from energy‑intensive chemical synthesis, both of which raise concerns about sustainability and safety.
Shining the right color of light
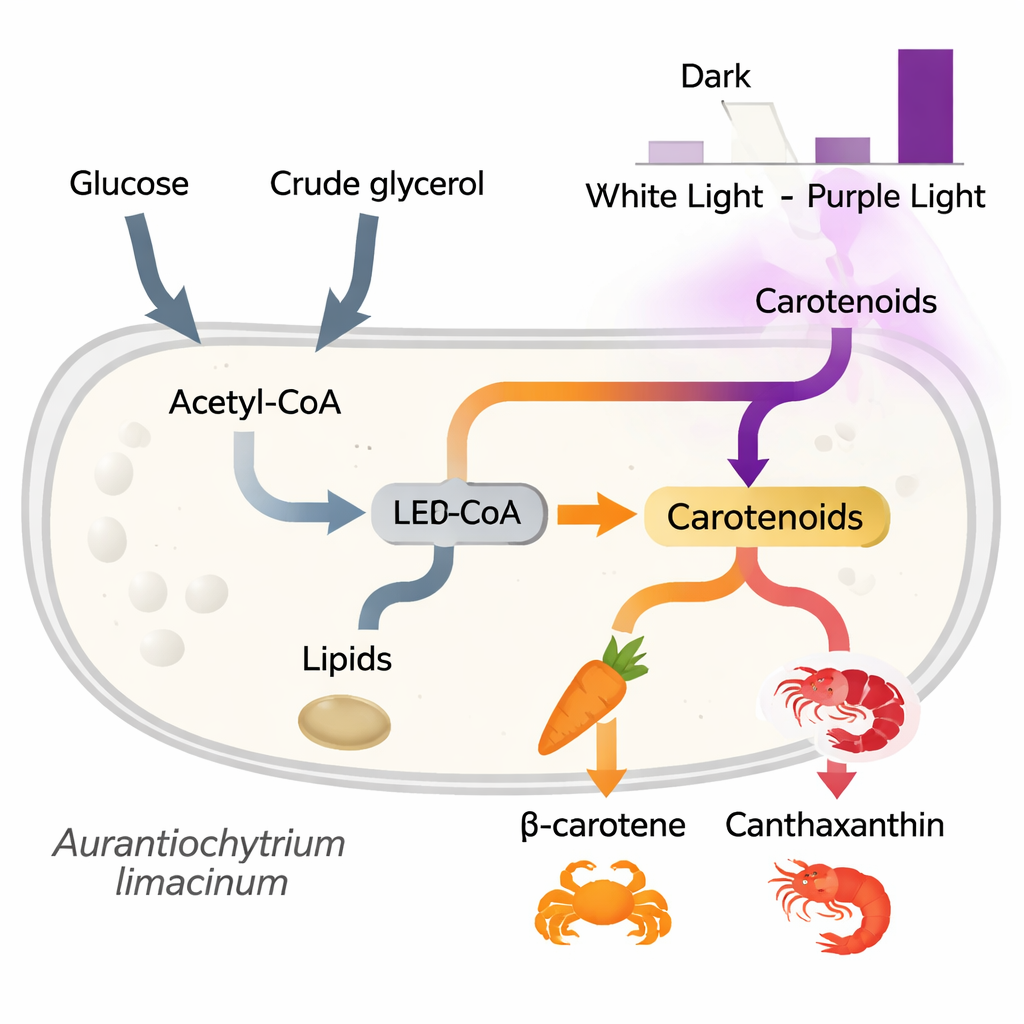
The researchers asked how different colors of light—darkness, normal white light, and narrow‑band purple LEDs (410–420 nm)—affect what this microbe makes. They grew cultures with either glucose (a simple sugar) or glycerol as the main food source and then measured cell growth, fat storage, and carotenoid levels. The cells grew just as well under all light colors, and their overall fat content stayed about the same. What changed dramatically was the pigment mix: purple light led to the highest levels of carotenoids, followed by white light, with darkness producing the least. Both β‑carotene and canthaxanthin, orange pigments that sit upstream in the astaxanthin pathway, rose sharply under purple light, especially when glycerol was used as the carbon source. Astaxanthin itself, however, was most abundant in cultures fed with glucose, showing that light color and food type subtly steer how far along the pigment pathway the cells go.
Turning waste into color and oil
A major challenge for bringing this microbe to market is the cost of feeding it. Glucose is relatively expensive at industrial scale, but biodiesel production generates large volumes of crude glycerol, a low‑value byproduct that can be difficult to dispose of. The authors tested whether this impure glycerol could be cleaned up just enough to serve as a useful feedstock. After simple treatments—dilution, acidification to remove soaps and salts, and in some cases an extra activated‑carbon polishing step—the crude glycerol supported microbial growth comparable to that seen with pure glucose or glycerol. Under purple LED light, cultures grown on treated crude glycerol produced high levels of β‑carotene and canthaxanthin, and eventually reached astaxanthin amounts similar to those from standard media, all while maintaining neutral lipid (oil) production.
Peeking under the hood of the cell
To understand how light and food type reshape metabolism, the team also examined which genes were turned on or off under different conditions using RNA sequencing. They found that genes involved in taking up and processing glycerol were strongly activated when glycerol was the food source, confirming that the cells can efficiently route this waste‑derived carbon into central metabolism. Surprisingly, many genes linked to fat and carotenoid synthesis were less active under purple light at an early time point, even though pigment levels were higher later on. This pattern suggests that the cells may first damp down some processes under light stress and then ramp up pigment production later as a protective response, using carotenoids as natural “sunscreens” and antioxidants.
What this means for future products
For non‑specialists, the key takeaway is that careful tuning of both lighting and diet can turn a marine microbe into a flexible, low‑cost factory for health‑related ingredients. Purple LED light boosts colorful, protective pigments without sacrificing the production of beneficial oils, while modestly purified crude glycerol—essentially an industrial waste stream—can replace more expensive sugars as the main food source. Together, these strategies point toward greener, more economical production of astaxanthin and DHA, reducing pressure on wild fisheries and fossil‑based chemistry while harnessing the quiet power of microbial biotechnology.
Citation: Yamakawa, K., Kawano, K., Kato, S. et al. Purple LED light and crude glycerol synergistically enhance astaxanthin production in Aurantiochytrium limacinum. Sci Rep 16, 6623 (2026). https://doi.org/10.1038/s41598-026-37313-4
Keywords: astaxanthin, Aurantiochytrium, purple LED light, crude glycerol, microbial bioproducts