Clear Sky Science · en
TRMT6 mitigates susceptibility and progression of DSS-induced colitis multifacetedly via translational regulation
Why This Gut Study Matters
Inflammatory bowel disease (IBD) – which includes Crohn’s disease and ulcerative colitis – affects millions of people worldwide and can cause chronic pain, bleeding, weight loss, and fatigue. At the heart of these conditions is a fragile, one-cell-thick lining of the intestine that must constantly repair itself while holding back trillions of microbes. This study uncovers a previously underappreciated molecular “caretaker,” a protein called TRMT6, that helps intestinal cells grow, heal, and manage their metabolism. Understanding how TRMT6 works could open new doors to gentler, more targeted treatments for IBD.
A Microscopic Guardian of the Gut Wall
The inner surface of the intestine is lined with a single layer of cells that renews itself every few days. This living carpet acts as a barrier, separating gut bacteria from the body’s internal tissues. The researchers found that TRMT6, an enzyme present in these intestinal cells, is reduced in tissue samples from people with active IBD and in mice with chemically induced colitis. TRMT6 does not change the DNA sequence; instead, it subtly modifies RNA – the working copies of genetic information that guide protein production – in ways that influence how efficiently proteins are made. When TRMT6 levels drop, this protein-making process falters, and the intestinal lining becomes more vulnerable to damage.
What Happens When TRMT6 Is Switched Off
To see how important TRMT6 really is, the team engineered mice in which TRMT6 was specifically deleted in the cells of the small intestine. Under normal conditions, these animals looked healthy and had intestines that appeared largely normal under the microscope. However, when the mice were exposed to a chemical (DSS) that irritates the gut and is widely used to model colitis, the picture changed dramatically. Compared with their normal littermates, TRMT6-deficient mice lost more weight, had shorter and more swollen colons, and showed deeper injury in the small intestine. A second genetic model that removed TRMT6 only from the intestinal stem cells – the cells that continually replenish the lining – produced similarly severe disease, confirming that the enzyme is particularly critical in this renewing compartment.
Slowed Repair, Disturbed Fuel Use, and Unruly Microbes
Closer inspection of the gut tissue revealed why the absence of TRMT6 is so harmful during colitis. In the damaged intestine, there were more cells undergoing programmed cell death and fewer cells actively dividing, as shown by standard tissue stains. A key growth regulator, the protein MYC, was markedly reduced at the protein level, even though its RNA message declined only modestly. This mismatch suggests that TRMT6 helps intestinal cells translate MYC RNA into protein efficiently, enabling rapid repair of the lining. To go further, the researchers combined large-scale measurements of RNA, proteins, small molecules (metabolites), and bacterial DNA. These “multi‑omics” analyses showed that loss of TRMT6 disrupts proteins involved in fat handling and nutrient uptake, alters levels of many lipid-like molecules and organic acids in the intestine and blood, and shifts the balance of gut microbes away from several beneficial groups toward more inflammation-linked species.
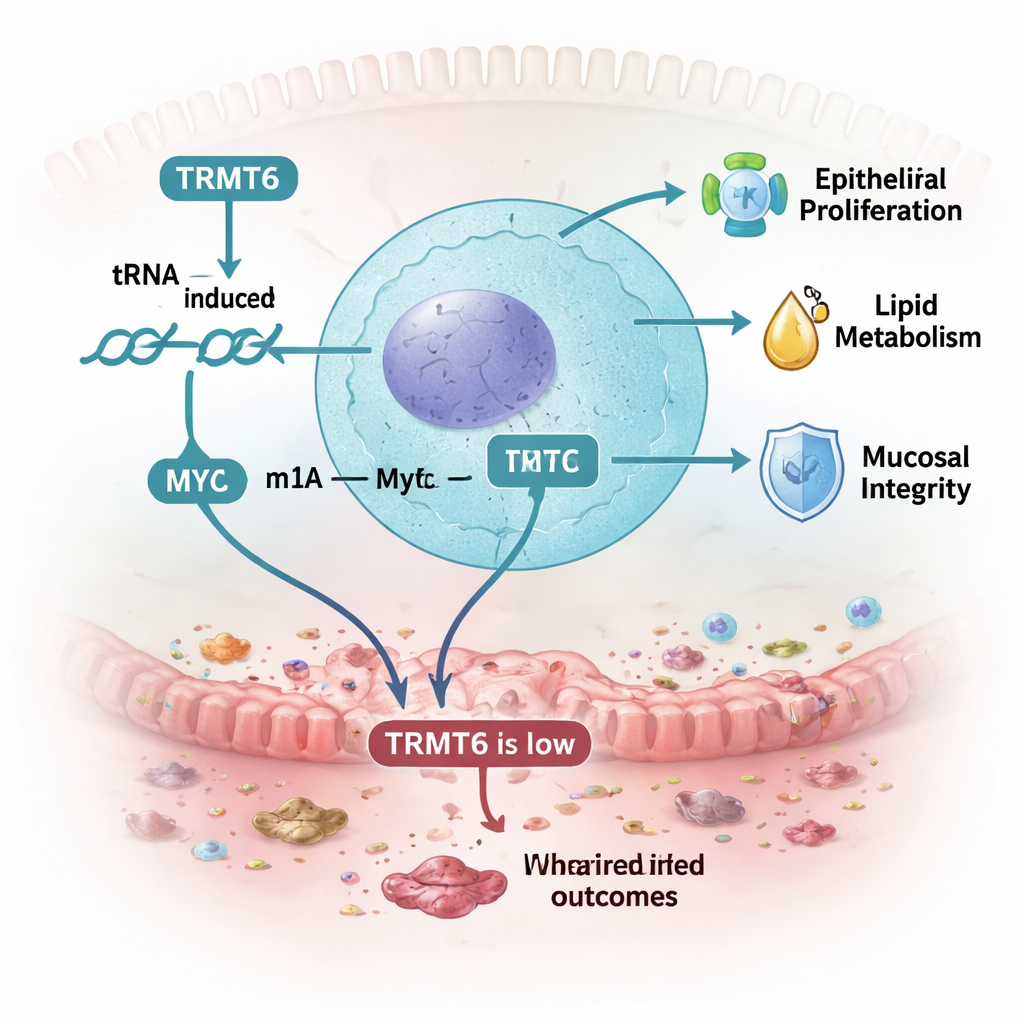
A Molecular Hub Linking Barrier, Metabolism, and Inflammation
Taken together, the findings portray TRMT6 as a central hub that helps the gut lining cope with stress. By fine-tuning a specific chemical mark on transfer RNA, TRMT6 supports the production of MYC and other proteins that drive cell renewal, regulate lipid processing, and maintain a tight seal between the body and its resident microbes. When TRMT6 is missing or reduced, the intestinal wall repairs itself more slowly, absorbs nutrients less efficiently, and creates conditions that favor a more harmful microbial community. Under the added pressure of an inflammatory trigger, these weaknesses translate into more severe colitis in mice, echoing the reduced TRMT6 seen in human IBD tissue.
What This Could Mean for Future Treatments
For non-specialists, the key message is that IBD is not only an immune problem; it also arises from failures in the everyday housekeeping of intestinal cells. TRMT6 emerges as one of the cell’s tools for keeping protein production, metabolism, and barrier repair in balance. While this research is still at an early, preclinical stage, it suggests that gently boosting TRMT6 activity, or mimicking its effects on RNA and protein synthesis, might one day help strengthen the gut lining and calm inflammation in people with IBD. Larger studies in different disease models and in patient samples will be needed, but this work points to a new, more precise angle for tackling chronic intestinal inflammation.
Citation: Zhang, X., Du, Y., Ye, Y. et al. TRMT6 mitigates susceptibility and progression of DSS-induced colitis multifacetedly via translational regulation. Sci Rep 16, 6809 (2026). https://doi.org/10.1038/s41598-026-37299-z
Keywords: inflammatory bowel disease, intestinal barrier, RNA modification, gut microbiome, intestinal metabolism