Clear Sky Science · en
Anticipation of periodic events influences cell motility in amoeba proteus
Single Cells That Seem to “Know” What Comes Next
Most of us think of anticipation and memory as traits of animals with brains. This study, however, shows that even a single-celled creature, the amoeba Amoeba proteus, can change how it moves in a way that looks strikingly like it is expecting a future threat. Understanding how such a simple organism does this may reshape how we think about learning, behavior, and even disease in more complex life forms.
How a Simple Cell Gets Around
Amoeba proteus is a large single cell that crawls by constantly reshaping its body and streaming its internal fluid from one side to the other. This type of movement is common in many cells in our bodies, from immune cells that hunt germs to cancer cells that spread through tissues. Because movement is so crucial for survival, cells are highly sensitive to their environment and adjust their speed and direction when conditions change. Earlier work on another slime-like organism hinted that some single cells can “predict” repeating bad conditions, but it was unclear whether this ability exists more broadly in other species.
Testing Cell “Expectation” With Flashes of Light
To probe this question, the researchers exposed individual amoebas to short, regular bursts of blue light, a form of light known to be unpleasant and even damaging for many cells. The amoebas were kept in gentle infrared light most of the time—light they do not react to—and then received four blue light pulses of 10 or 20 seconds each, separated by one to nearly two minutes of darkness. The scientists filmed each cell through a microscope at 30 frames per second and tracked the movement of tiny crystals inside the amoeba. These crystals acted as natural markers of the cell’s internal flow, allowing the team to calculate how fast the interior “streamed” just before, during, and after each light pulse.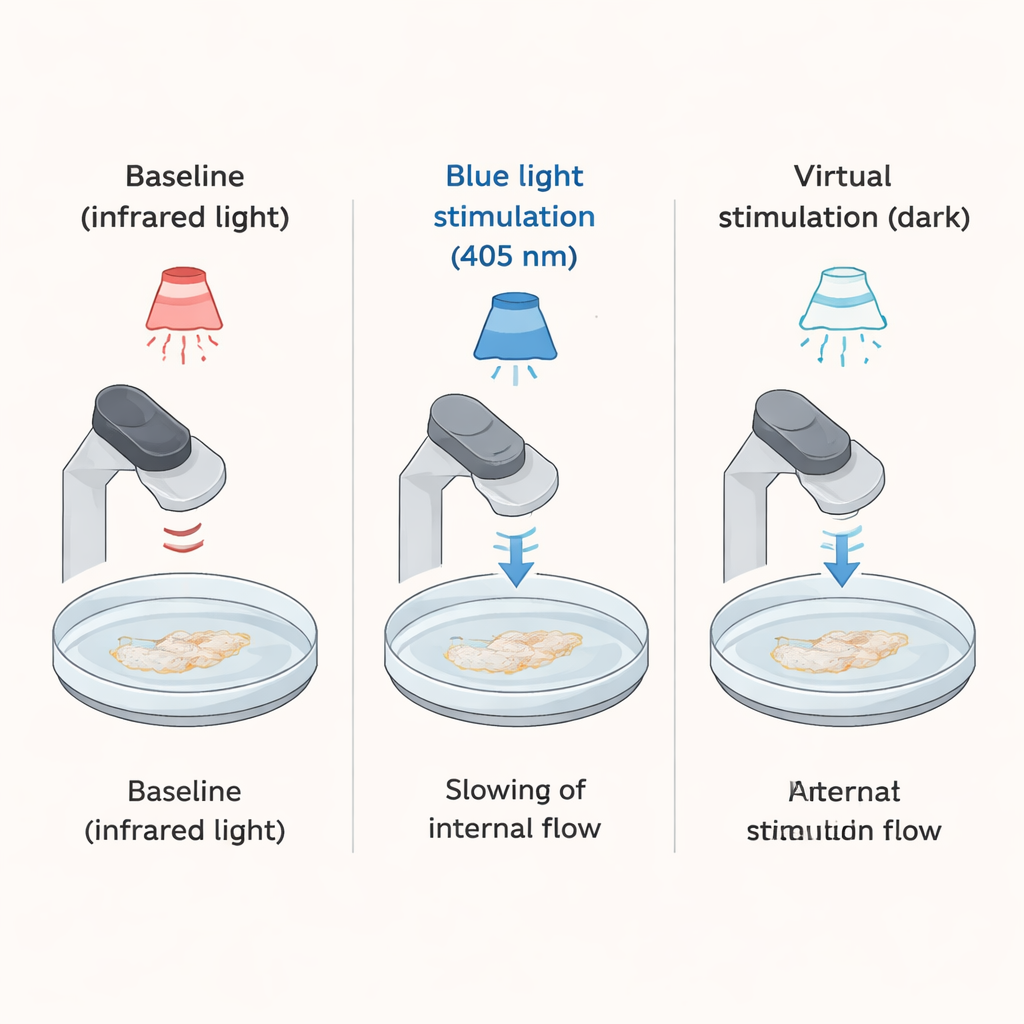
When Light Stops, the Cell Still Slows Down on Cue
As expected, every blue light pulse caused the amoebas’ internal streaming to slow sharply, sometimes almost to a halt, and then recover once the light was turned off. The key test came after the fourth real flash: the researchers continued filming for several more minutes but did not deliver any more blue light. Instead, they defined three “virtual” light moments—the times when the next flashes would have occurred if the pattern had continued. Remarkably, during the first of these virtual moments, most amoebas again showed a clear, in-phase slowdown of their internal flow, even though the cell was still sitting in harmless infrared light and received no new stimulus. About 90% of the cells slowed their streaming by more than 20% at that expected time point, and roughly one third repeated this anticipatory slowdown during all three virtual moments.
Comparing Real Light, Fake Light, and Quiet Periods
To be sure these changes were not just random wiggles, the team compared streaming speeds across many 20-second windows: before, during, and after each real and virtual light period, as well as during an undisturbed baseline phase. During baseline, speeds fluctuated only modestly. During true blue light pulses, speeds dropped drastically in all cells. During the first virtual pulse, speeds again fell significantly compared with the surrounding dark periods and with all baseline measurements, confirming that the slowdown was not just natural variability. Later virtual pulses showed weaker and less frequent slowdowns, suggesting that the “memory” of the pattern fades over a few minutes. Interestingly, the effect did not depend strongly on the exact length of the dark gaps between flashes: amoebas anticipated across a range of intervals between 60 and 100 seconds.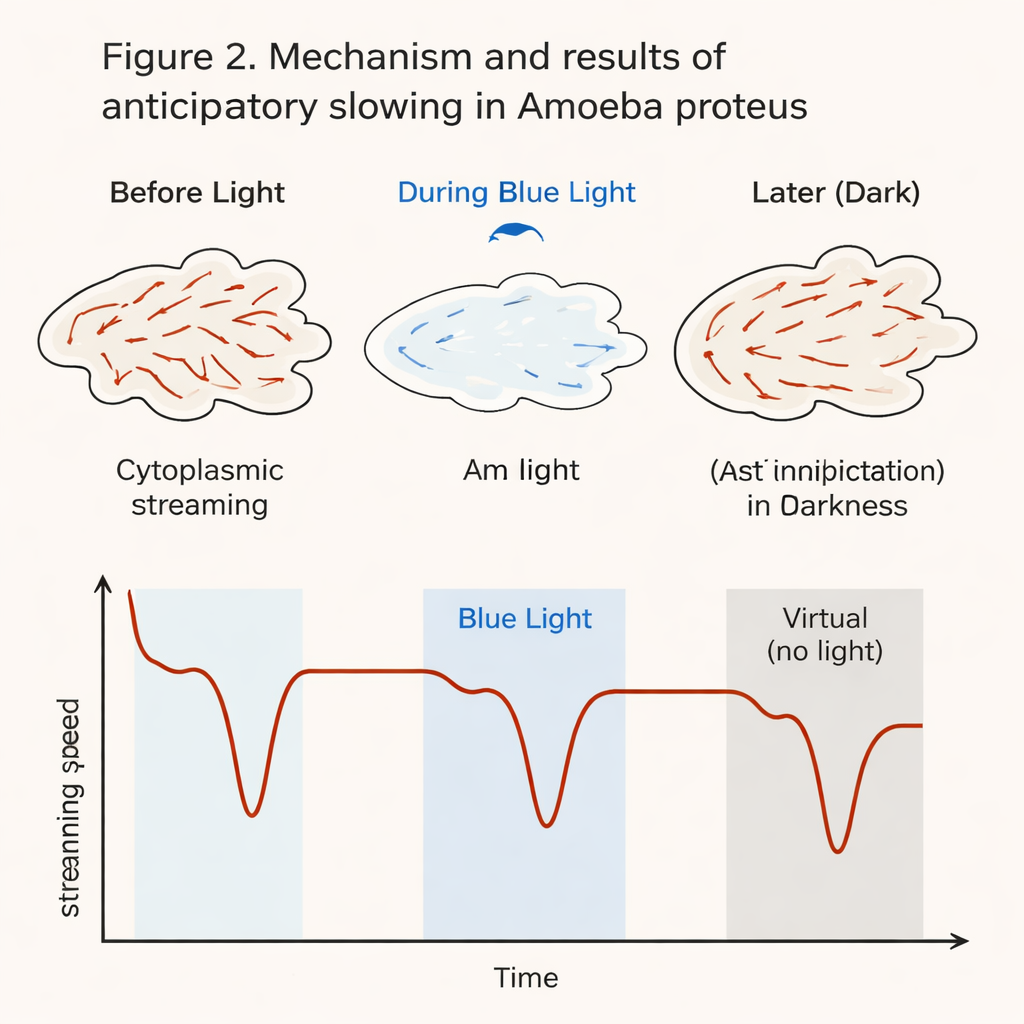
What Might Be Happening Inside the Cell?
How can a brainless cell behave as if it is predicting the future? The authors discuss ideas drawn from physics and cell biology. Some researchers model such behavior with “memory-like” electrical elements called memristors, which can store a history of past signals. In living cells, an equivalent memory could arise from slow, repeating chemical cycles. In Amoeba proteus, movement relies on a dynamic framework of actin fibers and motor proteins that push and pull the cell’s interior. Other cell types show rhythmic changes in this actin system, suggesting that built-in biological “oscillators” might tune themselves to repeated stimuli, such as periodic blue light. The authors outline future experiments that would gently disrupt actin, motor proteins, calcium signals, or cellular energy to see whether such changes weaken or erase the amoeba’s anticipatory behavior.
Why This Matters Beyond Amoebas
This work strengthens the idea that anticipation is not limited to animals with nervous systems. Instead, the ability to detect patterns and prepare for what comes next may be a basic property of life, emerging from the physics and chemistry inside even a single cell. For a layperson, the striking message is that a one-celled organism—without a brain, nerves, or senses as we usually define them—can nevertheless “learn” a repeating threat well enough to slow down before it arrives. Understanding these simple, robust forms of cellular memory could eventually inform how we view cell behavior in development, immunity, cancer, and even future strategies for regenerative medicine.
Citation: Mueller, S.M., Martin, S., Morawski, M. et al. Anticipation of periodic events influences cell motility in amoeba proteus. Sci Rep 16, 4762 (2026). https://doi.org/10.1038/s41598-026-37298-0
Keywords: cell motility, single-celled learning, amoeba behavior, anticipation, blue light stimulation