Clear Sky Science · en
Diagnostic performance of multimodal biomarkers in colorectal cancer
Why this matters for everyday health
Colorectal cancer is one of the most common and deadly cancers worldwide, yet it is also highly curable when caught early. Many people avoid colonoscopy because it is invasive and inconvenient, and common non-invasive tests can miss cancers. This study explores whether combining several simple blood and stool tests can offer a more accurate, comfortable way to flag who is likely to have colorectal cancer and who may safely avoid unnecessary procedures.
Looking for cancer clues in blood and stool
The researchers followed 881 people treated at a large hospital in China: 188 had confirmed colorectal cancer and 693 had similar digestive symptoms but no cancer. Instead of relying on a single marker, they measured several different signals that cancer or its surrounding environment can leave behind. These included a special DNA change in the blood (called methylated SEPT9), classic tumor markers used in many hospitals (CEA and CA19-9), a stool test that detects hidden blood (fecal occult blood test, or FOBT), and two measures from a standard blood count that reflect inflammation and changes in red blood cells (RDW-CV and PLR). By studying how each of these behaved alone and in combination, they asked a simple question: can a multi-test panel do a better job of spotting cancer than any single test?
How the study was carried out
All participants gave routine blood and, when available, stool samples. The team tested blood for methylated SEPT9 using a commercial kit that looks for cancer-related DNA changes, measured CEA and CA19-9 with automated hospital analyzers, and calculated RDW-CV and PLR from standard complete blood counts. FOBT was done on stool using a widely used immunochemical kit. Using statistical models, they compared results between cancer and non-cancer groups, identified which markers were independently linked to having colorectal cancer, and then built a prediction model. They evaluated performance using receiver operating characteristic (ROC) curves, which summarize how well a test balances catching true cancers (sensitivity) against avoiding false alarms (specificity).
What each test can – and cannot – do alone
Every individual marker showed meaningful differences between the cancer and non-cancer groups. The cancer group had far higher positive rates of methylated SEPT9 and FOBT, and elevated levels of CEA, CA19-9, RDW-CV, and PLR. When examined one by one, methylated SEPT9 excelled at ruling out non-cancer cases, correctly identifying about 95% of people without cancer, but it missed a substantial share of cancers. FOBT showed the opposite pattern: it caught about 85% of cancers but incorrectly labeled around one in five non-cancer patients as high risk. CEA provided the strongest overall single-test performance, while CA19-9, RDW-CV, and PLR on their own were only modestly informative. These results highlight a core problem of single tests: each captures only one slice of how cancer behaves.
Stronger together: combining multiple signals
Because each marker reflects a different biological process—genetic changes in tumor DNA, proteins shed by tumor cells, bleeding from bowel lesions, and cancer-related inflammation—the researchers tested how well combinations perform. Pairing methylated SEPT9 with FOBT sharply boosted the chance of catching cancers, and adding tumor markers CEA and CA19-9 improved detection further. When the six key indicators (methylated SEPT9, FOBT, CEA, CA19-9, RDW-CV, and PLR) were combined into a single prediction model, performance was striking: the panel correctly flagged about 92% of cancer patients while correctly reassuring roughly 84% of people without cancer. Statistically, this combined model achieved an area under the ROC curve of 0.939, much higher than any single test or simple pair, underscoring the power of integrating multiple, complementary biomarkers.
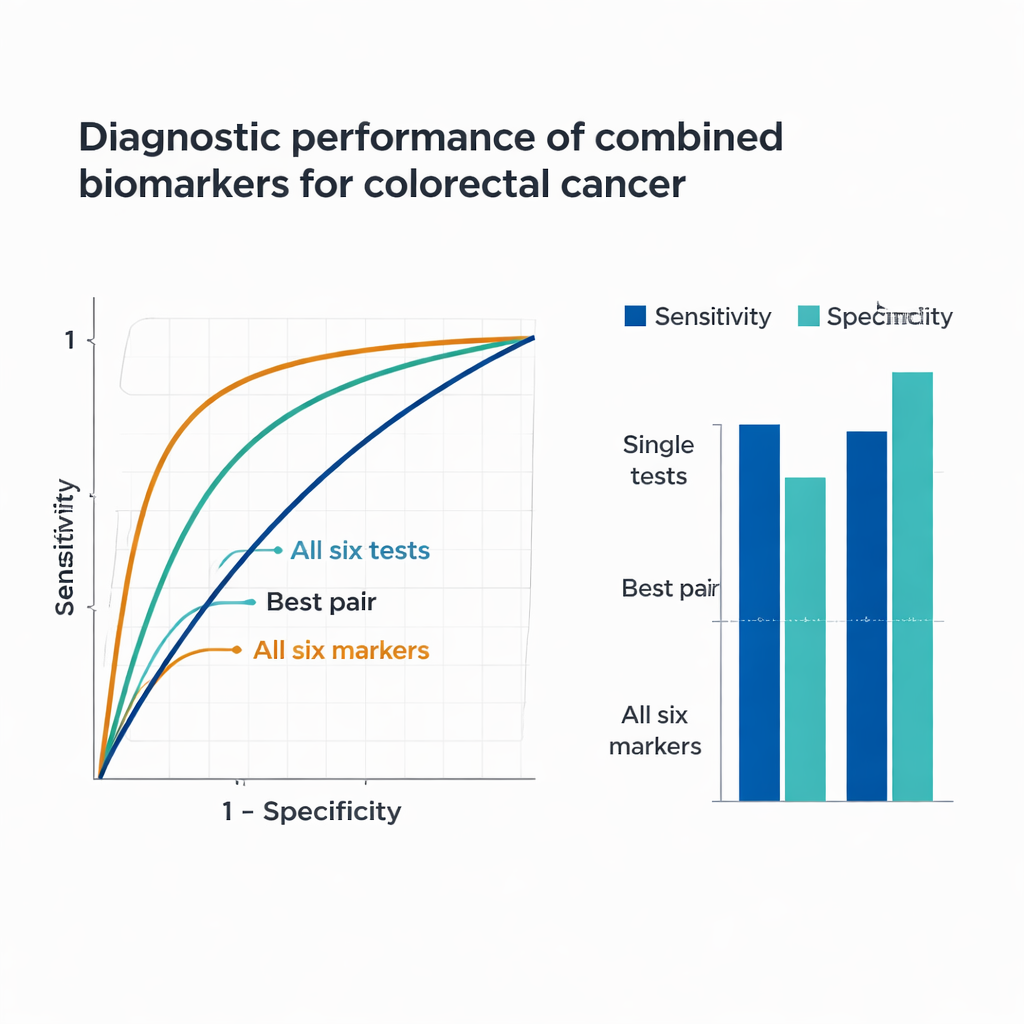
What this could mean for screening and care
For people wary of colonoscopy, this multi-test approach offers a promising, non-invasive way to assess cancer risk using only blood and stool samples. While it cannot fully replace colonoscopy, a panel with such high sensitivity and good specificity could be used to decide who most urgently needs the invasive test and who might be safely monitored. The authors note that their study was conducted in one hospital and did not fully analyze performance in very early cancers or in long-term follow-up, so broader, multi-center research is still needed. Even so, the work suggests that combining several simple, widely available tests could reshape colorectal cancer screening, catching more cancers earlier while reducing unnecessary procedures, costs, and anxiety for patients.
Citation: Yang, S., Wang, Y., Li, J. et al. Diagnostic performance of multimodal biomarkers in colorectal cancer. Sci Rep 16, 6273 (2026). https://doi.org/10.1038/s41598-026-37280-w
Keywords: colorectal cancer, cancer screening, blood test, stool test, biomarkers