Clear Sky Science · en
N-acetyl cysteine amide mitigates oxidative stress and apoptosis in a rat model of renal ischemia-reperfusion injury
Why protecting our kidneys matters
Every day, our kidneys quietly filter our blood, remove waste, and keep our bodies in balance. But during major surgery, severe infection, or transplantation, blood flow to the kidneys can briefly shut down and then restart, causing a burst of damage known as ischemia–reperfusion injury. This study explores whether a more powerful form of a familiar antioxidant supplement can help shield kidneys from this kind of stress—potentially reducing the risk of acute kidney injury in people who face high‑risk medical procedures.
A brief pause that can do lasting harm
When blood flow to the kidneys is cut off and then restored, the sudden return of oxygen can paradoxically harm the tissue. Cells unleash large amounts of reactive oxygen species—highly reactive molecules that damage fats, proteins, and DNA. This “oxidative stress” triggers inflammation and programmed cell death (apoptosis), which together can severely impair kidney function. Doctors currently have limited tools to prevent this type of injury, so researchers turn to animal models to test new protective strategies before they can be tried in humans.
A stronger twist on a common antioxidant
N-acetylcysteine (NAC) is a well‑known antioxidant used clinically, for example, to treat acetaminophen overdose. However, in kidney protection studies its benefits have been inconsistent, partly because it does not enter cells and mitochondria efficiently. The team in this study focused instead on N-acetyl cysteine amide (NACA), a modified form of NAC that is more fat‑soluble and can more easily cross cell membranes. They asked two main questions in rats: does NACA lessen kidney damage after ischemia–reperfusion, and does the timing of NACA—given before versus after the blood flow blockage—change how well it works?
Putting NACA to the test in rats
Researchers used four groups of rats. One group served as a control, while a second group underwent kidney ischemia–reperfusion without treatment. Two additional groups received NACA at the same dose: one group got the drug half an hour before blood flow was cut off, and the other received it right after the ischemic period, just before reperfusion. The scientists then measured blood markers of kidney function, chemical signs of oxidative stress in kidney tissue, microscopic damage to kidney structures, and levels of a key cell‑death protein called caspase‑3. This combination of tests allowed them to see not just whether the kidneys looked healthier, but also whether core damage pathways were being quieted.
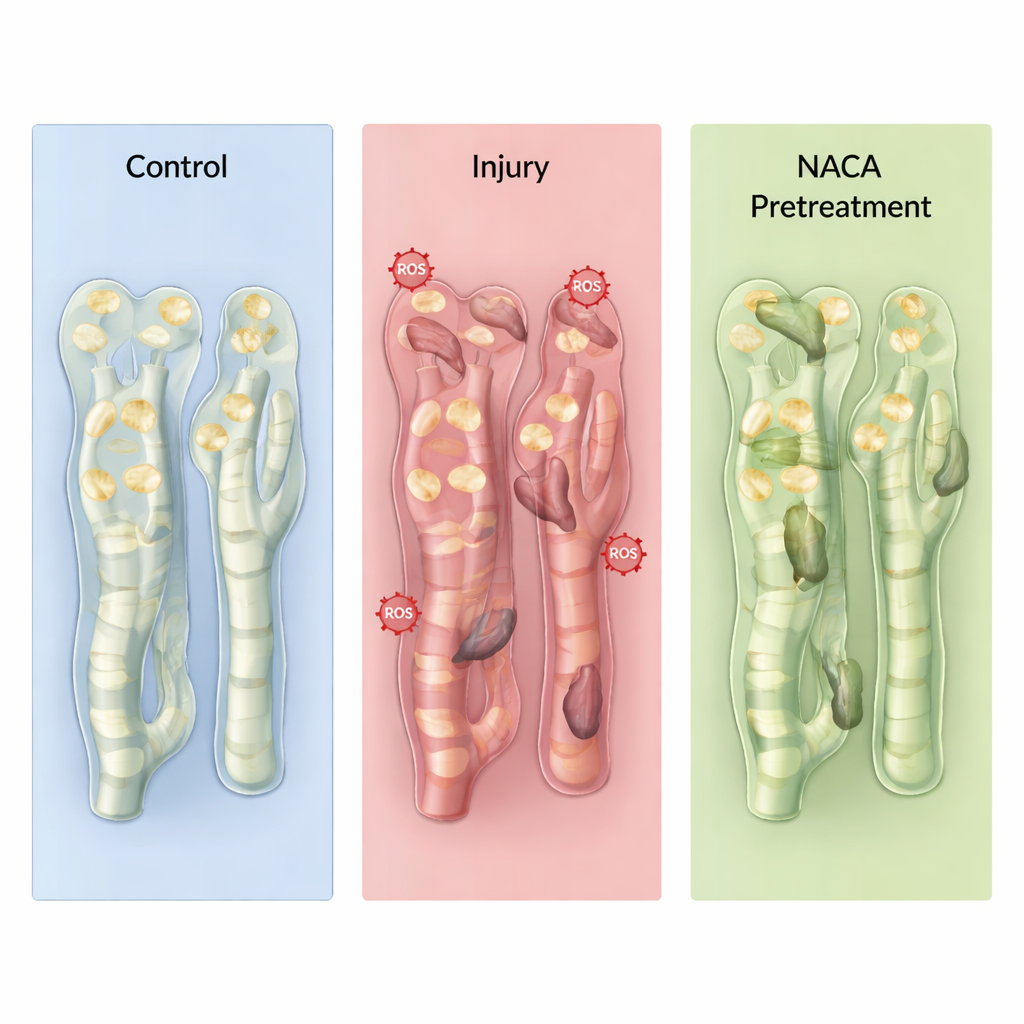
Partial protection—and why timing counts
As expected, rats that underwent ischemia–reperfusion without NACA showed clear signs of acute kidney injury: waste products such as urea and creatinine rose, chemical markers of fat damage increased, and microscopic examination revealed dilated tubules, debris “casts,” and inflamed tissue. When NACA was given before ischemia, several of these changes were blunted. Markers of lipid damage fell, key antioxidant enzymes were closer to normal, inflammatory cell infiltration was reduced, and caspase‑3 activity—an indicator of cells entering programmed death—dropped markedly. Giving NACA only after the ischemic period helped less; by the time it arrived, many damaging reactions had already been triggered, so apoptosis and inflammation were not reduced to the same degree.
What this could mean for patients
The study shows that NACA cannot completely prevent kidney injury, but it can provide meaningful “partial renoprotection” by reducing oxidative stress and slowing early cell death, particularly when present in the tissue before blood flow is restored. For a layperson, this suggests that a better‑designed antioxidant, delivered at the right moment, might one day help protect vulnerable kidneys during events like transplantation or complex surgery. The work is still at the animal stage and only one dose and time window were tested, so human benefit is not yet proven. However, the findings strengthen the idea that anticipating kidney stress and pre‑arming the organ with targeted antioxidants like NACA could become an important part of future strategies to lower the risk of acute kidney injury.
Citation: Ozhan, O., Ekici, C., Ates, B. et al. N-acetyl cysteine amide mitigates oxidative stress and apoptosis in a rat model of renal ischemia-reperfusion injury. Sci Rep 16, 6323 (2026). https://doi.org/10.1038/s41598-026-37274-8
Keywords: acute kidney injury, oxidative stress, ischemia reperfusion, antioxidant therapy, N-acetyl cysteine amide