Clear Sky Science · en
Downregulation of the deubiquitinating enzyme USP10 correlates with neuronal apoptosis in HTLV-1-associated myelopathy
Why this matters for people living with viral nerve diseases
Some viruses do not just cause short‑lived infections; they can quietly damage the nervous system over many years. Human T‑cell leukemia virus type 1 (HTLV‑1) is one such virus and can lead to a chronic spinal cord disorder called HTLV‑1‑associated myelopathy (HAM). People with HAM gradually lose strength and control in their legs, along with bladder and sensory problems. This study asks a simple but crucial question: why do the nerve cells in the spinal cord die in this condition, and could a single protective molecule help explain who fares better or worse?
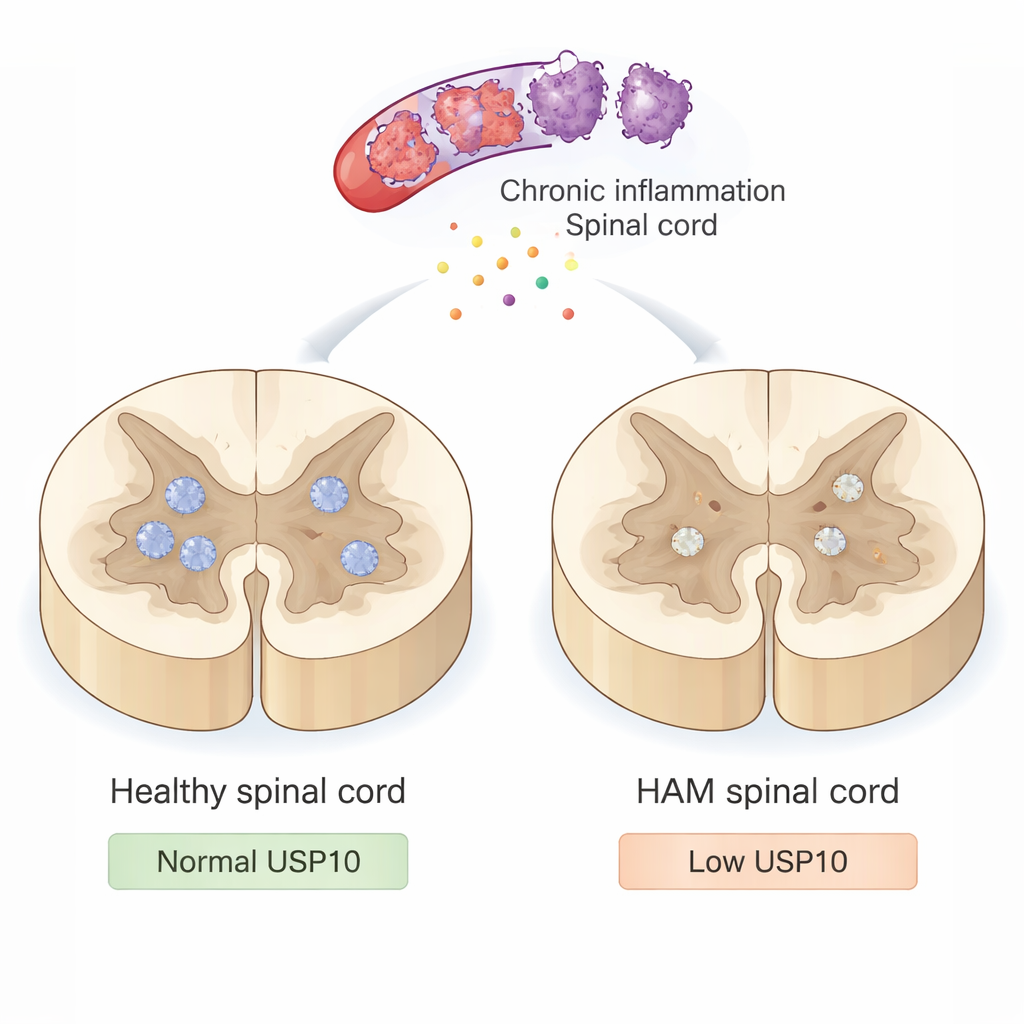
A slow attack on the spinal cord
HAM is not caused by the virus directly infecting nerve cells. Instead, HTLV‑1 infects immune cells that invade the spinal cord and trigger long‑lasting inflammation. These overactive immune cells release toxic chemicals and inflammatory signals that, while meant to control infection, also injure nearby neurons and their insulating myelin. Over time, this smoldering assault leads to stiff, weak legs and difficulty walking. Yet doctors still do not fully understand why some neurons succumb to this inflammatory environment while others survive for decades.
A cellular “cleanup” enzyme under suspicion
The researchers focused on a protein called USP10, an enzyme that helps manage the cell’s protein quality‑control system and is known to limit cellular stress and programmed cell death (apoptosis). USP10 is active in many tissues, including the brain and spinal cord, and previous work in Parkinson’s disease, stroke, and other conditions suggests it helps neurons withstand oxidative and inflammatory damage. Using spinal cord samples from eight people who died with HAM and two neurologically healthy controls, the team used staining techniques to see where USP10 was present and how strongly it was expressed in nerve cells.
Less USP10, more dying neurons
In healthy spinal cords, USP10 was strongly visible in many neurons. In most HAM cases, however, USP10 staining in neurons was clearly reduced, and in one patient it was almost absent. The scientists then looked for hallmarks of apoptosis using two methods: a TUNEL test that detects fragmented DNA and staining for active caspase‑3, a protein involved early in cell death. In the controls, virtually no neurons showed these death signals. In contrast, in HAM patients, a large fraction of neurons were TUNEL‑positive and many showed active caspase‑3. Importantly, samples with lower USP10 expression tended to have more apoptotic neurons and fewer surviving neurons marked by NeuN, a standard neuronal marker. One notable patient with HAM who still walked with only a cane after 22 years had strong USP10 expression and relatively preserved neuron numbers, hinting that higher USP10 may help protect against severe disability.
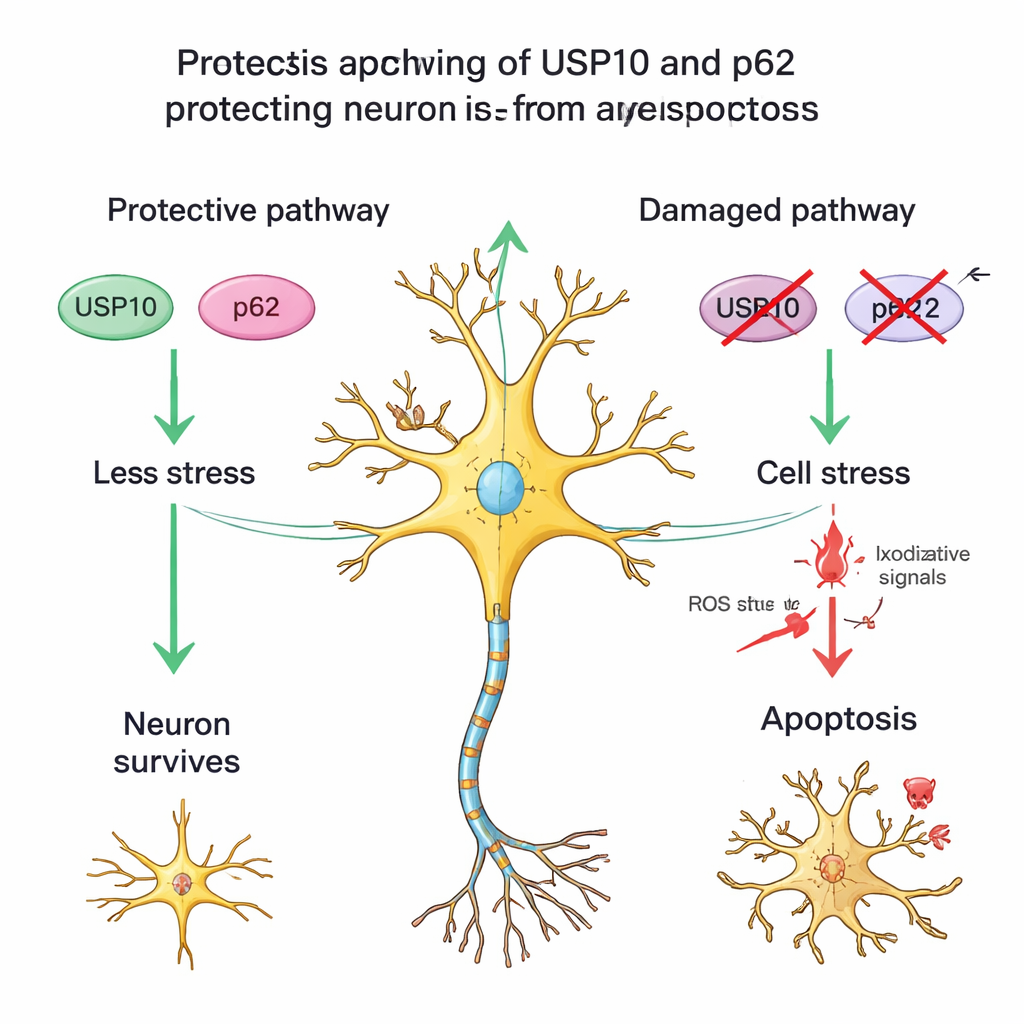
A partner protein and stressed but surviving cells
The team also examined another protein, p62, which works with USP10 in handling damaged proteins and regulating stress and survival pathways. In healthy spinal cords, p62 staining was moderate; among HAM patients, those with low USP10 often also had weaker p62 staining and fewer surviving neurons. This pattern supports the idea of a USP10–p62 partnership that helps neurons withstand chronic inflammation. The researchers further noticed many neurons in HAM tissue that had the typical shape of nerve cells but had lost NeuN staining, a sign seen in other brain injuries when neurons are under serious stress yet not fully dead. This suggests that in HAM, some neurons are not only lost outright but others remain in a damaged, vulnerable state, potentially because USP10‑linked defenses are weakened.
What this could mean for future diagnosis and treatment
This study does not prove that loss of USP10 directly causes neuron death, because it is based on a small number of autopsy samples and cannot follow changes over time. Still, the consistent links between low USP10, reduced p62, more signs of apoptosis, and fewer surviving neurons point to a common pathway that might tie together chronic inflammation and slow neuronal degeneration in HAM. For patients, this raises an encouraging possibility: measuring or boosting USP10‑related pathways in the spinal cord—or potentially in more accessible samples like spinal fluid—could one day help identify those at higher risk of rapid progression, guide monitoring, or inspire new treatments designed to reinforce the cell’s own stress‑protection systems in virus‑induced neuroinflammatory diseases.
Citation: Arishima, S., Takahashi, M., Dozono, M. et al. Downregulation of the deubiquitinating enzyme USP10 correlates with neuronal apoptosis in HTLV-1-associated myelopathy. Sci Rep 16, 6062 (2026). https://doi.org/10.1038/s41598-026-37271-x
Keywords: HTLV-1-associated myelopathy, neuroinflammation, neuronal apoptosis, USP10, spinal cord degeneration