Clear Sky Science · en
Antibacterial activity and cytotoxicity of tricalcium silicate-based cements with different antibacterial additives
Why this matters for your next filling
When you have a deep cavity, dentists now try to save as much natural tooth as possible instead of drilling everything away. That often means a thin layer of softened, bacteria‑containing dentin is left behind under the filling. This study asks a simple but crucial question: can we upgrade a widely used tooth‑repair cement so it quietly kills leftover germs without harming the living cells inside the tooth?
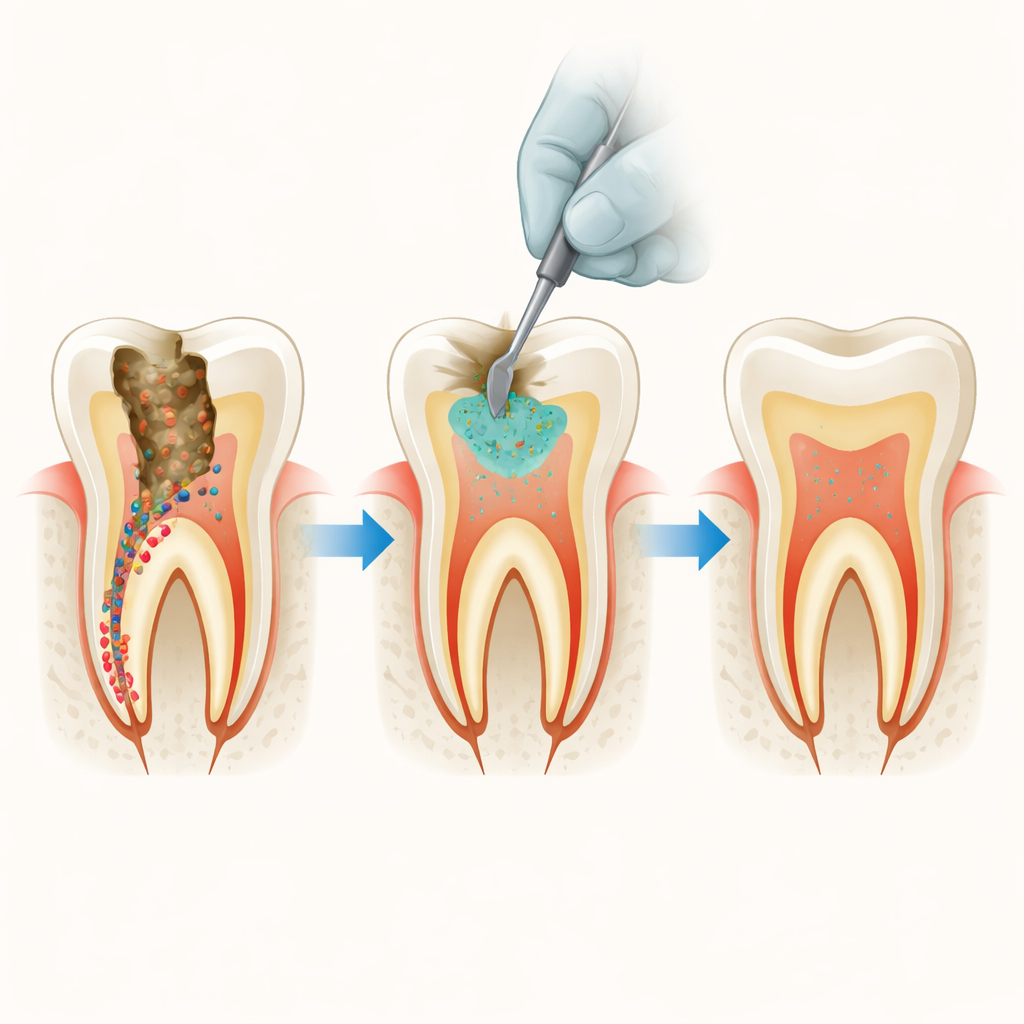
Deep cavities and hidden germs
Modern "minimal drilling" techniques aim to avoid exposing the tooth’s soft core, or pulp, by leaving behind dentin that can still harden again. The downside is that some cavity‑causing bacteria, such as Streptococcus mutans and Lactobacillus species, can linger in that layer. Over time, these bacteria may trigger new decay beneath a seemingly healthy filling. Tricalcium silicate‑based cements are popular for sealing deep cavities because they help minerals re‑form and create a tight barrier, but on their own they are not very good at stopping bacteria.
Turning a common cement into a germ fighter
The researchers took this familiar cement and mixed in five different antibacterial additives at various strengths. Two were organic compounds often found in disinfectants (benzalkonium chloride and cetrimide), and three were inorganic substances used in dental products or materials (titanium oxide, zinc oxide, and stannous fluoride). They pressed these mixtures into tiny discs and placed them on plates coated with five types of bacteria linked to tooth decay and oral infections. By measuring the clear circles around each disc where bacteria could not grow, they could see how strongly each recipe held germs at bay.
Finding the sweet spot between killing germs and sparing cells
Because a material that kills bacteria but also poisons tooth cells would be useless in the mouth, the team also tested how the most promising additives affected living cells. They exposed two human cell types—fibroblasts, which are common support cells, and dental pulp stem cells, which help repair tooth tissue—to the cements indirectly. A color‑change test measured how many cells stayed active, and special fluorescent dyes showed live (green) and dead (red) cells under the microscope. This setup mimicked the real situation in a deep cavity, where the cement is separated from the pulp by a thin dentin layer and releases substances that diffuse toward the cells.
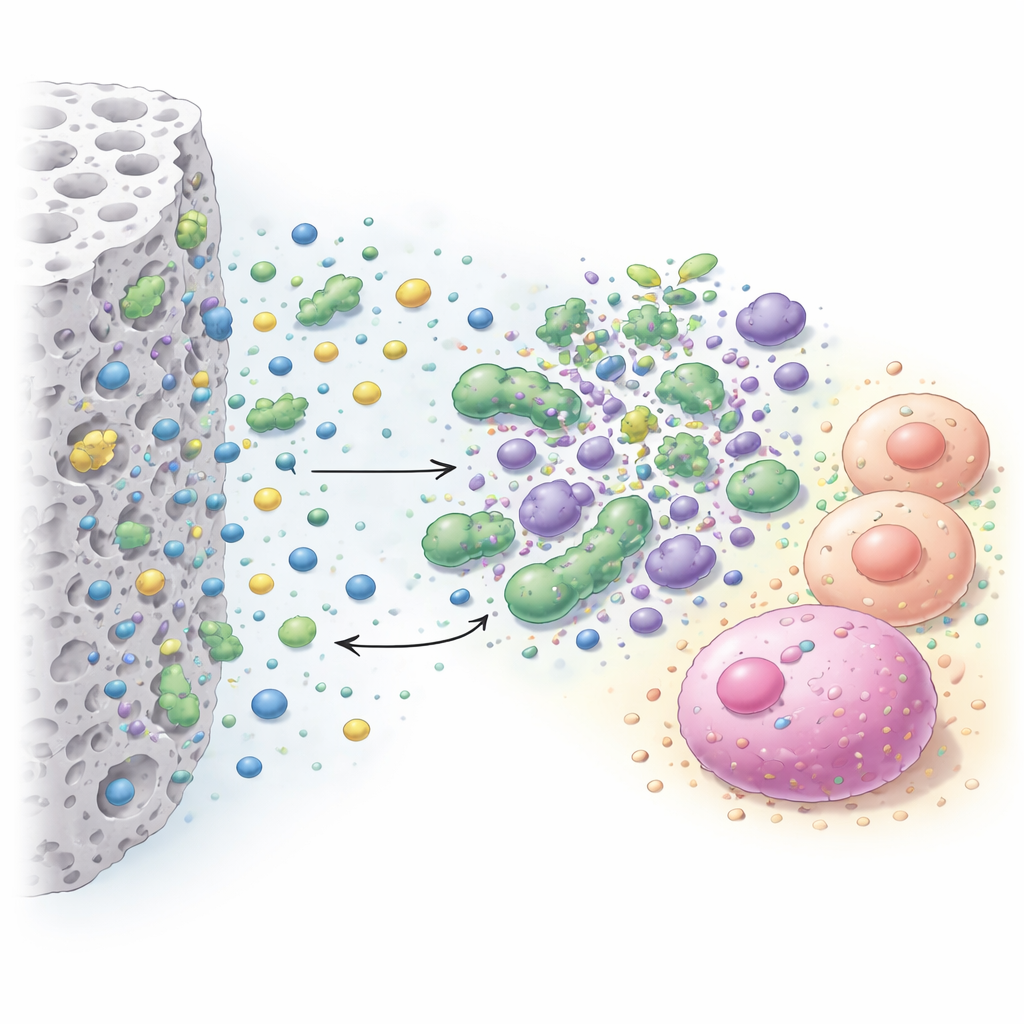
What worked best and what was too harsh
The results showed that the type and amount of additive mattered a great deal. Benzalkonium chloride gave the strongest and broadest antibacterial effect, producing large germ‑free zones against all tested species, especially the Lactobacillus strains often found in deep decay. Cetrimide also helped, but generally needed higher concentrations to match benzalkonium’s performance. The three inorganic agents only improved the cement’s antibacterial effect at the higher doses tested, and even then mainly against certain bacteria such as S. mutans and Actinomyces. On the safety side, the base cement alone was kind to both cell types and even seemed to support fibroblast health. Adding benzalkonium chloride remained harmless to fibroblasts up to about 1%, became slightly damaging at 2–4%, and clearly more harmful at 7%. Dental pulp stem cells were more sensitive overall: both benzalkonium chloride and cetrimide reduced their survival, with cetrimide becoming moderately toxic from 2% upward.
What this could mean for future dental care
Putting these findings together, the study points to one especially promising recipe: tricalcium silicate‑based cement containing 1% benzalkonium chloride. At this level, the material strongly suppressed harmful bacteria while keeping fibroblasts fully alive and dental pulp stem cells mostly viable. In everyday terms, it hints that future deep fillings could be made from a cement that quietly fights leftover germs under the surface while still being gentle enough to the living tissue inside the tooth. More work is needed—especially in complex, real‑world mouth conditions—but this balanced formula could help dentists better protect teeth treated with minimally invasive techniques.
Citation: Banon, R., Martens, L., De Coster, P. et al. Antibacterial activity and cytotoxicity of tricalcium silicate-based cements with different antibacterial additives. Sci Rep 16, 8349 (2026). https://doi.org/10.1038/s41598-026-37269-5
Keywords: deep dental caries, antibacterial dental cement, tricalcium silicate, benzalkonium chloride, pulp cell biocompatibility