Clear Sky Science · en
Contrast-enhanced T1-weighted MRI, 11C-DPA-713 PET and 11C-CPPC PET as predictive imaging biomarkers of neuroinflammation in radiotherapy-induced brain injury
Why this research matters
As treatments for childhood and young adult brain tumors improve, more patients are surviving for many years after therapy. Unfortunately, radiation to the brain can leave a lasting mark, damaging blood vessels and brain cells and leading to problems with memory, attention, and thinking. Doctors need safe, noninvasive ways to spot this hidden damage early, long before daily life is clearly affected. This study asks whether a common hospital scan—contrast-enhanced MRI—can track harmful brain inflammation as reliably as more specialized PET scans that involve extra radiation exposure.
Hidden damage after life-saving treatment
Radiotherapy-induced brain injury develops slowly over months to years after radiation. The process is driven in part by neuroinflammation: the brain’s own immune cells become overactive, blood vessels are damaged, and the blood–brain barrier—normally a tight filter—starts to leak. This leak allows more immune cells from the bloodstream to enter the brain, fueling a cycle that can ultimately kill neurons and impair cognition. Today, doctors usually detect this injury only when patients already show learning or memory problems on neuropsychological testing. The authors aimed to find imaging “early warning signs” that could reveal this process much sooner.
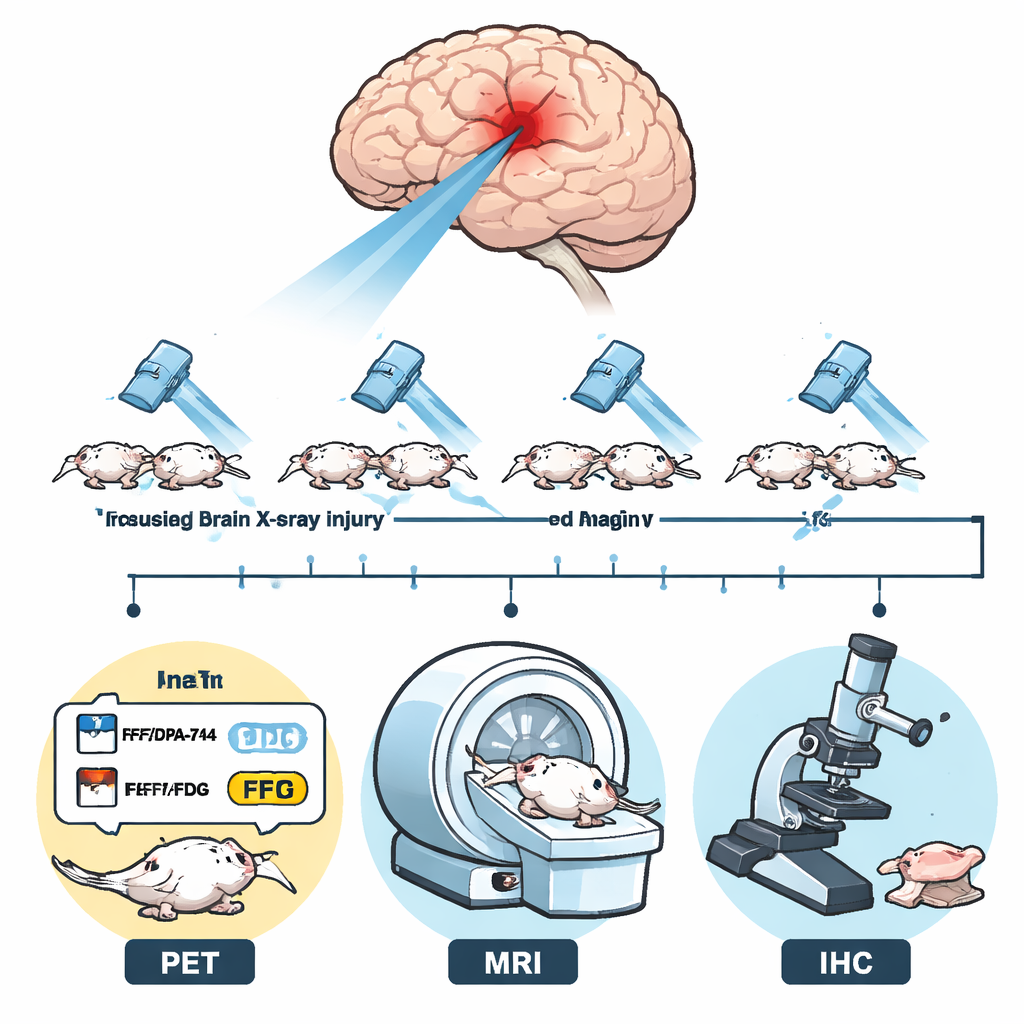
Comparing three ways of seeing brain inflammation
To tackle this question, researchers used a mouse model that mimics focused radiation to a small region of the brain, comparable to what some adolescent patients receive. After irradiation, four groups of mice were followed for nine months. One group was scanned with a PET tracer called 11C-DPA-713, which binds to a protein (TSPO) that increases when many brain immune cells are activated. A second group received a different PET tracer, 11C-CPPC, which homes in on a receptor found specifically on microglia, the brain’s resident immune cells. A third group underwent standard contrast-enhanced T1-weighted MRI, which highlights places where the blood–brain barrier has become leaky. The final group did not receive imaging but instead had their brain tissue examined under the microscope using fluorescent staining for markers of activated microglia and infiltrating immune cells.
What the PET and MRI scans revealed over time
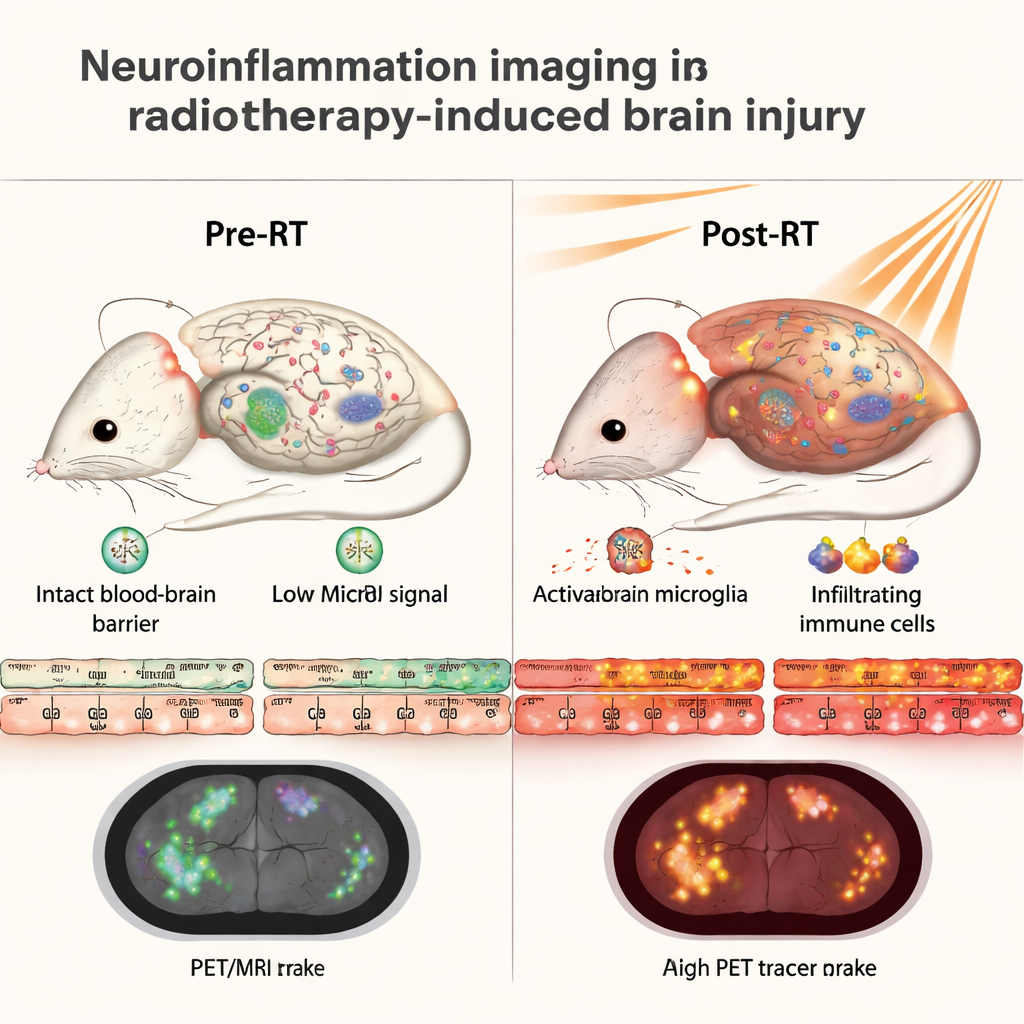
The team found that both PET tracers lit up the irradiated side of the brain most strongly about one month after radiation, then gradually declined over the next several months. This pattern matched what one would expect for a surge of neuroinflammation that then slowly subsides, though it does not fully disappear. Importantly, the contrast-enhanced MRI signal—reflecting how leaky the blood–brain barrier had become—showed a very similar time course. The irradiated hemisphere had dramatically higher contrast uptake than the untouched side, peaking at one month and then falling but remaining elevated even at nine months. When the researchers quantified these changes, the MRI measurements closely tracked the PET signals, with strong statistical correlations.
Microscope evidence backs up the scans
To confirm that these imaging changes really reflected inflammation, the scientists examined brain slices stained for several immune markers. At one month after radiation, the irradiated hemisphere showed a surge in activated microglia and infiltrating macrophages, along with very high levels of the TSPO protein. These microscopic signals decreased over time in parallel with the PET and MRI findings. When the authors directly compared the numbers, they found that both PET tracers and the MRI contrast signal lined up closely with the amount of TSPO and microglial activation seen under the microscope. This suggests that a leaky blood–brain barrier and neuroinflammation are tightly linked in this form of radiation injury.
What this could mean for patients
The study shows that a widely available clinical technique—contrast-enhanced T1-weighted MRI—can serve as a strong indirect indicator of neuroinflammation in radiation-injured brain, performing nearly as well as specialized PET scans in this mouse model. While MRI does not label specific cell types the way PET tracers do, it avoids additional radiation exposure, making it especially attractive for children and young adults who have already received high doses to the brain. Used thoughtfully, contrast MRI could help clinicians monitor the brain health of survivors over time and evaluate new treatments designed to protect or repair the irradiated brain, offering a safer window into a process that has long been difficult to see.
Citation: Maiti, S., Yadav, S.K., Teitz, M. et al. Contrast-enhanced T1-weighted MRI, 11C-DPA-713 PET and 11C-CPPC PET as predictive imaging biomarkers of neuroinflammation in radiotherapy-induced brain injury. Sci Rep 16, 6384 (2026). https://doi.org/10.1038/s41598-026-37264-w
Keywords: radiotherapy-induced brain injury, neuroinflammation, contrast-enhanced MRI, PET imaging, blood-brain barrier