Clear Sky Science · en
Transcriptome analysis of perforated small cocoon from Bombyx mori mutants
Why tiny, holed cocoons matter
Silk doesn’t start as fabric; it begins as a tiny caterpillar spinning a snug, continuous cocoon. When that process goes wrong—producing small cocoons riddled with holes—farmers lose silk and income. This study looks inside such defective silkworms at the level of genes to find out what’s misfiring, revealing how growth, feeding, and spinning are wired together in an insect that underpins a major textile industry.
From healthy silk spinners to troubled mutants
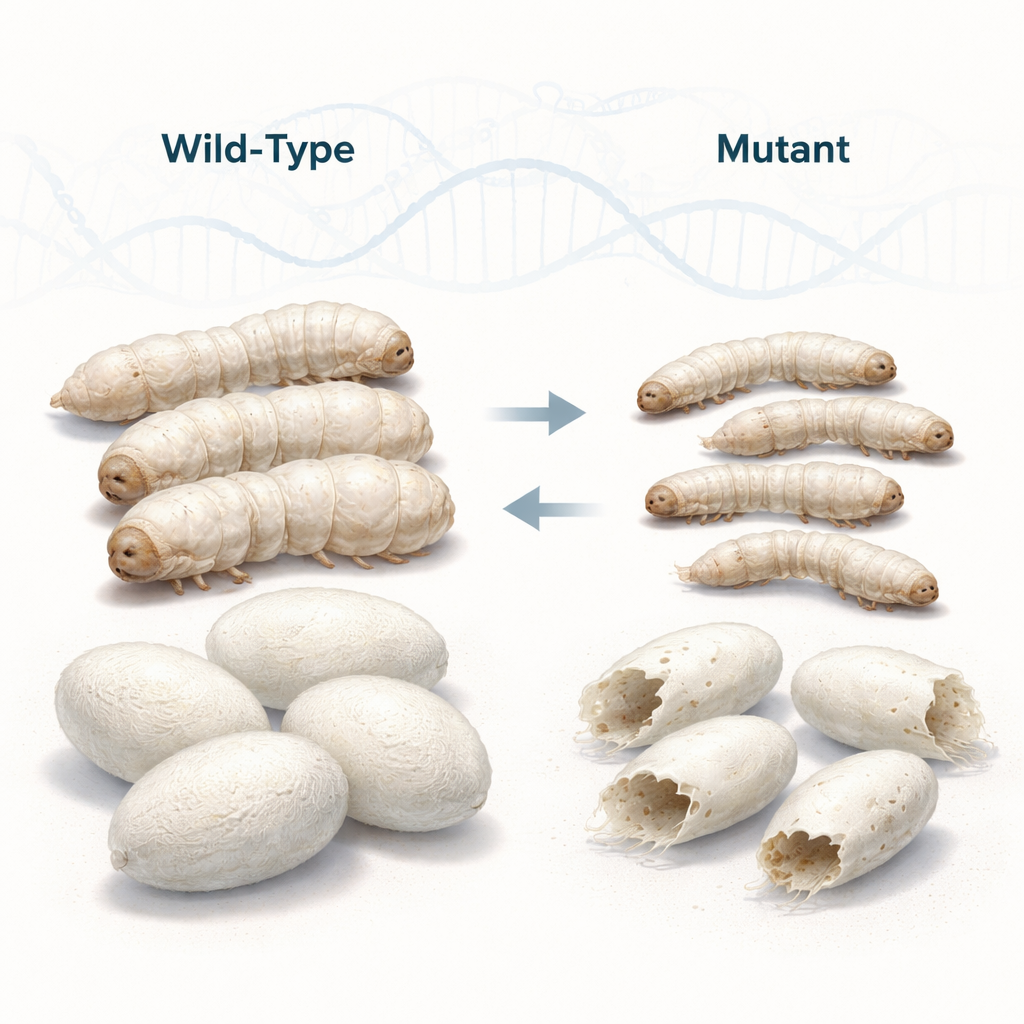
Researchers worked with a common silkworm strain and a newly discovered mutant called perforated small cocoon (psc). Although both were raised under identical conditions, the mutant larvae grew more slowly, stayed smaller and thinner, and, as adults, produced fewer and lighter cocoons. The mutant cocoons were not only undersized but often thin at one or both ends and punctured with small holes—defects that make them nearly useless for commercial silk reeling. By carefully timing feeding and development, the team pinpointed the third larval stage as the moment when the mutant’s growth began to fall behind, suggesting that something early in development derails its later cocoon building.
Reading the silkworm’s molecular script
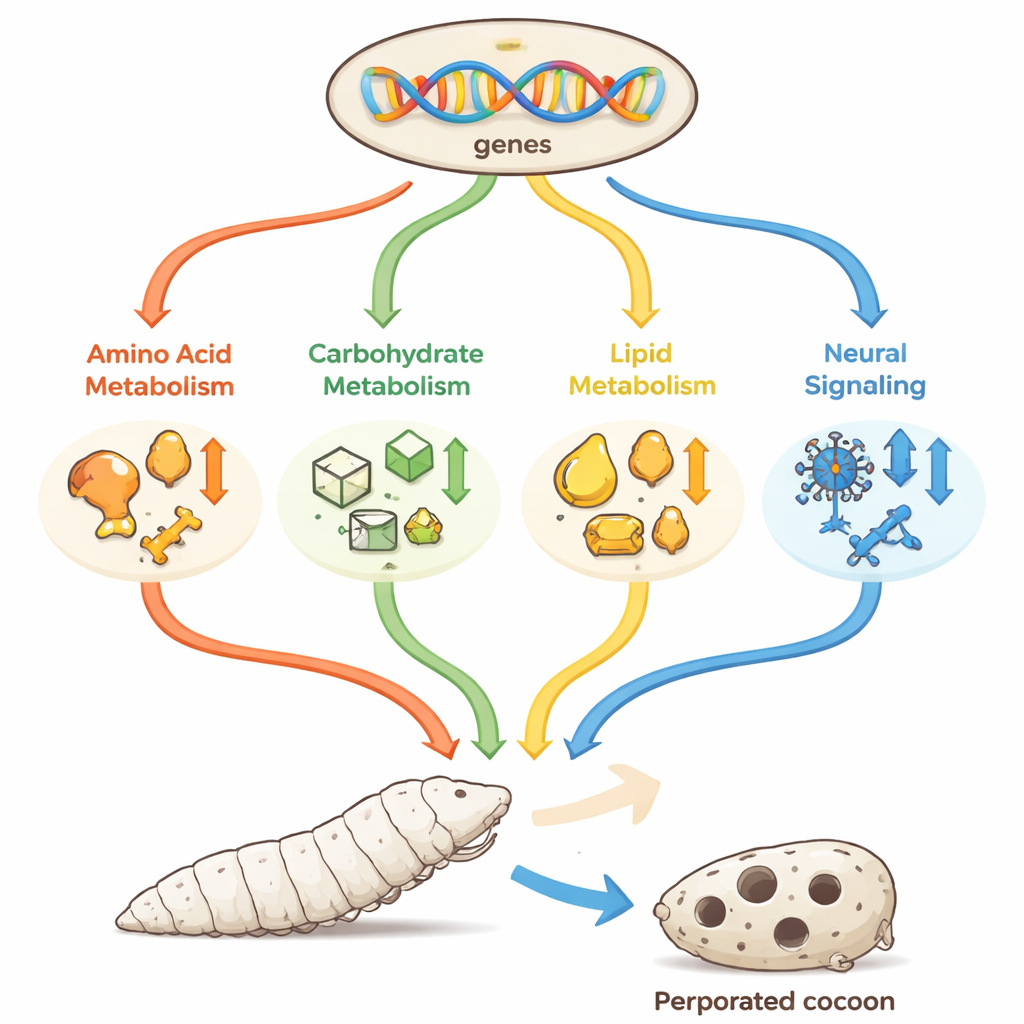
To uncover what goes wrong, the scientists examined the transcriptome—the full set of active genes—of whole larvae at the start of the third stage, comparing the mutant with the normal strain. They found 716 genes whose activity levels differed, roughly half turned up and half turned down. Many of these genes clustered in pathways that manage the body’s basic fuel and building blocks: amino acids (the components of proteins), carbohydrates (sugars and starches), and lipids (fats), as well as in pathways that help nerve signals travel through the body. The team cross-checked a subset of genes using a separate technique, confirming that the RNA sequencing data accurately reflected real changes in gene activity.
Starved of fuel and building materials
Digging deeper, the researchers saw that key steps in amino acid metabolism, especially those leading from the amino acid tyrosine to melanin pigments, were dialed down. In silkworms, melanin is not just for color; it helps harden body parts like mouthparts, which are essential for steady feeding, and it also contributes to the structure of the shell. Several "yellow" family genes and a related gene involved in pigment chemistry were less active, which may soften mouthparts and cuticle, matching the observed slow, hesitant feeding. At the same time, genes for carbohydrate processing enzymes, such as alpha-amylase that digests starch and enzymes that detoxify plant chemicals, were reduced, potentially limiting how much energy the larvae can extract from mulberry leaves. Lipid-handling genes that help make and reshape fatty acids also tended to be suppressed, threatening energy storage and hormone production important for growth and reproduction.
Miswired signals from the nervous system
Beyond metabolism, some of the most striking changes appeared in genes encoding neuropeptide receptors, which sit on nerve and other cells and respond to small signaling molecules. Several of these receptors belong to families that, in other animals, regulate appetite, movement, and daily activity rhythms. In the mutant silkworms, multiple receptors were turned down, including ones related to hunger and to coordinated movements. Because silkworms must rhythmically swing their heads and control silk release to wind an even, closed cocoon, weakened signaling in these pathways could easily produce the jerky behavior and poor control that yield thin, perforated shells. The study also detected shifts in genes for hormone-processing enzymes and waste-handling pathways, hinting that disrupted signaling ripples out into many bodily systems.
Linking faulty genes to broken cocoons
Taken together, the findings paint a picture of a silkworm whose growth is undermined on two fronts: its brain receives weaker “eat and spin” signals, and its metabolism is less able to turn food into energy, structural proteins, and shell material. With fewer resources and poorer coordination, the larvae stay small, eat slowly, and ultimately spin light, holed cocoons. By mapping the specific genes and pathways involved, this work offers breeders concrete molecular targets for developing robust silkworm lines that grow well and produce strong, high-quality cocoons—helping secure the living thread between a tiny insect and a global silk industry.
Citation: Zhou, K., Wei, X., Shen, D. et al. Transcriptome analysis of perforated small cocoon from Bombyx mori mutants. Sci Rep 16, 6654 (2026). https://doi.org/10.1038/s41598-026-37263-x
Keywords: silkworm, cocoon defects, insect genetics, metabolism, RNA sequencing