Clear Sky Science · en
Rapid clearance of bacteria from maternal bloodstream after delivery in pregnancies complicated by preterm pre-labor rupture of the membranes
Why this research matters for mothers and babies
Pregnancy complications that lead to very early birth are among the most frightening situations for families and doctors. One such condition, called preterm pre-labor rupture of membranes (PPROM), happens when a woman’s “water” breaks weeks before the due date. This raises the risk of dangerous infections for both mother and baby. The study summarized here asks a simple but important question: when germs get close to the baby’s sac in PPROM, do they spill over into the mother’s bloodstream around the time of birth, and if so, how quickly can her body clear them?
A closer look at early water breaking
In a normal pregnancy, the fluid around the baby and the thin fetal membranes that hold it are usually free of microbes. But when the membranes rupture too early, vaginal bacteria can climb upward and infect the sac and placenta. Doctors worry about sepsis, a life-threatening bodywide reaction to infection, yet they often must balance the dangers of infection against the harms of delivering an extremely premature infant. The researchers enrolled 66 women with PPROM, most around 32 weeks of pregnancy, to track what was happening with bacteria and inflammation in the mothers’ blood and in the tissues supporting the fetus at delivery.
Following bacteria from womb to bloodstream
The team collected maternal blood just before birth and again within an hour after delivery. They also sampled fetal membranes and placental tissue in a subset of cases. Instead of relying on standard cultures, which miss many microbes, they used sensitive DNA-based methods that can detect even tiny traces of bacterial genetic material. At the same time, they measured signaling proteins in the mothers’ blood—cytokines such as interleukin-6 (IL-6) and interleukin-10 (IL-10)—and used cord blood tests and placental examination to decide whether each baby had been exposed in the womb to a strong inflammatory state, a condition they refer to as “Triple I.”
Hidden germs and a swift cleanup
Bacterial DNA was surprisingly common in the mothers’ blood: it was found in over 80% of samples both before and after birth. However, when bacterial levels were carefully compared to a stable human reference gene, the overall amount of bacterial DNA in blood actually fell significantly after delivery in about two-thirds of women. At the same time, levels of IL-6 and IL-10 in maternal blood rose sharply, especially in pregnancies where the fetus showed clear evidence of inflammatory exposure. In these higher-risk pregnancies, the bigger the jump in IL-6 after birth, the larger the drop in bacterial DNA, suggesting that the mother’s immune response was actively helping to clear bacterial fragments from her circulation.
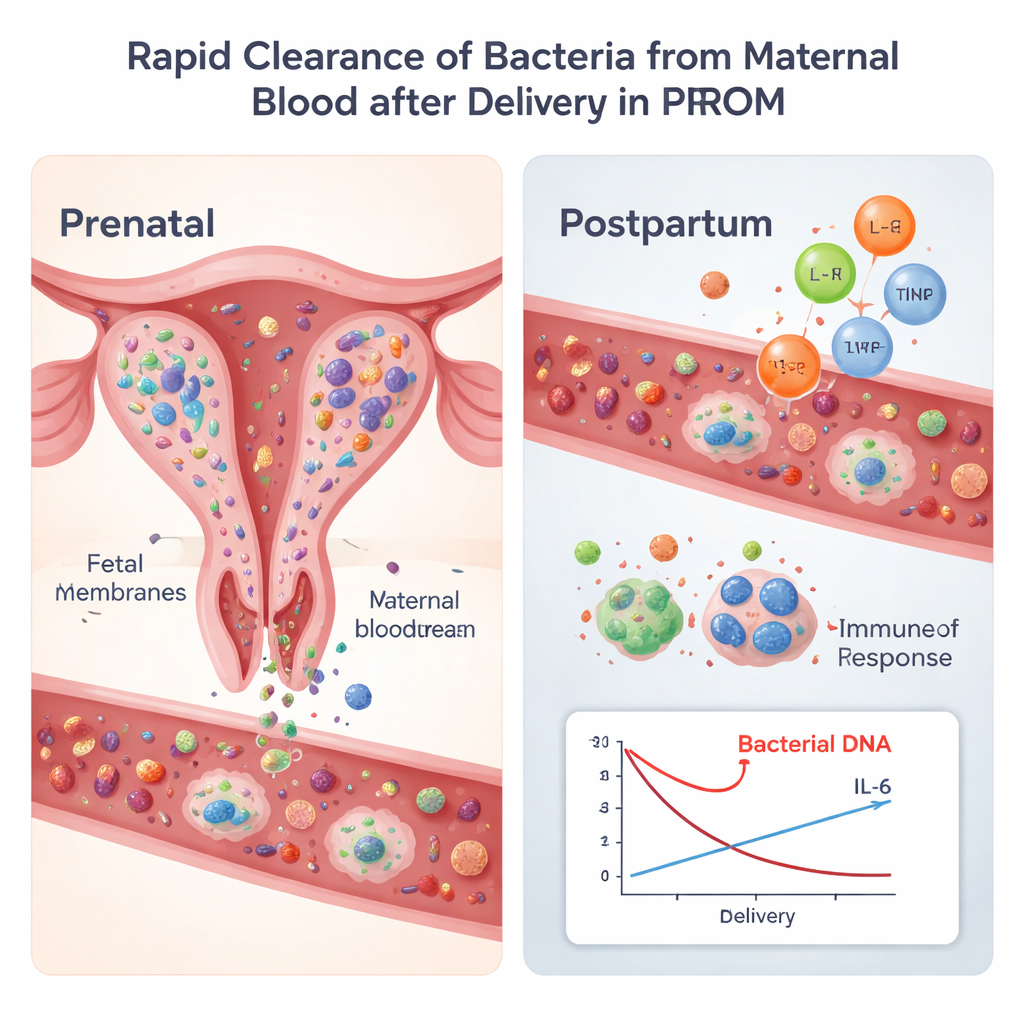
Where the bacteria really live
When the researchers compared bacterial DNA in blood to that in fetal membranes and placental tissue, they found that the membranes carried far more bacteria and a richer mix of species than the placenta or blood. The membranes were dominated by Mycoplasma and other organisms commonly linked to genital tract infections. Yet these same bacteria were largely absent from maternal blood, where only a limited set of bacterial families—often related to gut or mouth microbes such as Escherichia-Shigella and Pseudomonas—were detected. There was very little overlap between the specific bacteria found in blood and those found in the reproductive tissues, arguing against the idea that delivery routinely “flushes” membrane bacteria into the mother’s circulation.
What this means for patients and care
For families and clinicians facing PPROM, this study offers a measure of reassurance. It shows that while traces of bacterial DNA are commonly present in the mother’s blood before birth, her body is usually able to reduce this load quickly after delivery, in concert with a surge of protective immune signals. The fetal membranes can harbor a substantial community of microbes, but these organisms rarely seem to flood the maternal bloodstream in large amounts. Instead, the mother’s immune system appears to mount a dynamic, effective cleanup. Understanding exactly how this protection works could help doctors better identify the rare patients whose defenses fail—and, ultimately, improve outcomes for both mothers and their premature infants.
Citation: Buhimschi, C.S., Zhao, G., Rood, K.M. et al. Rapid clearance of bacteria from maternal bloodstream after delivery in pregnancies complicated by preterm pre-labor rupture of the membranes. Sci Rep 16, 6725 (2026). https://doi.org/10.1038/s41598-026-37231-5
Keywords: preterm birth, PPROM, maternal infection, blood microbiome, fetal membranes