Clear Sky Science · en
Choice of lipid supplementation for in vitro erythroid cell culture impacts reticulocyte yield and characteristics
Why growing blood in the lab matters
Modern medicine depends on donated blood, but supplies are often tight and not every patient can safely receive blood from every donor. Scientists are therefore working on ways to grow red blood cells in the lab as a reliable, customizable backup. This study asks a surprisingly basic but crucial question: when we grow young red blood cells (reticulocytes) outside the body, does the type of fat and cholesterol we feed them change how well they work?
The special skin of red blood cells
Red blood cells survive for months while being squeezed through tiny blood vessels thousands of times a day. They can do this because of their unusual outer “skin”: a flexible skeleton of proteins anchored to a membrane rich in cholesterol. Unlike most cells, up to half of the membrane fat in a red blood cell is cholesterol. That balance keeps the cell tough yet bendable. Too little cholesterol and the membrane becomes fragile; too much and it stiffens. In people, disturbed cholesterol balance can shorten red cell life and worsen oxygen delivery. For lab-grown blood to function like the real thing, it needs to copy this finely tuned membrane.
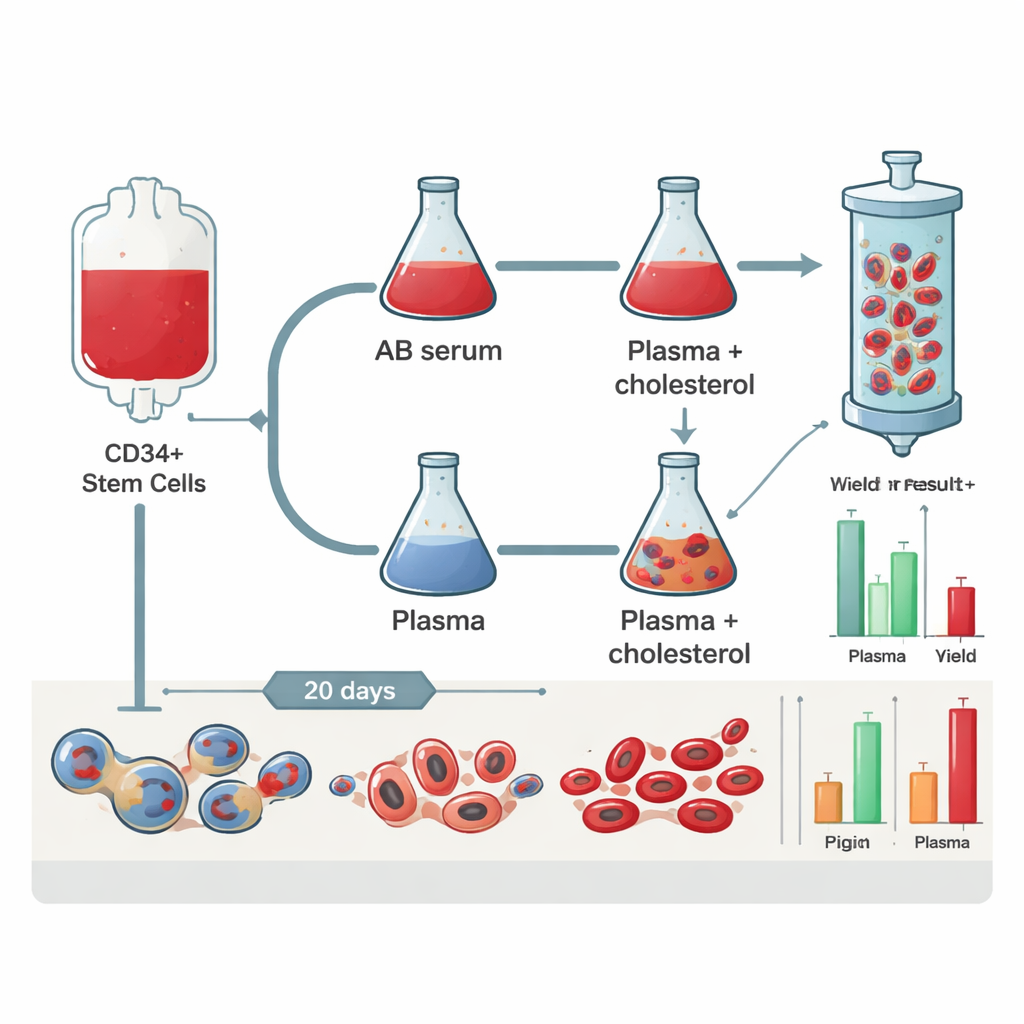
Testing different recipes for lab-grown blood
The researchers used human stem cells marked by CD34, which can mature into red blood cells when given the right growth factors. They kept the basic culture recipe the same but changed the main source of lipids (fats and cholesterol). One group received human AB serum, a blood fraction long used in red cell culture. Another group received a pooled plasma product that has been treated with solvents and detergents to inactivate viruses. A third group received the same plasma plus an extra mix of cholesterol-rich lipids. Over 20 days, the team tracked how much the cells multiplied, how many successfully kicked out their nucleus to become reticulocytes, and how easily the young cells could be filtered and handled, mimicking what would happen when preparing them for transfusion.
Cholesterol-poor plasma makes weaker cells
Cells grown with AB serum expanded well and produced reticulocytes that could pass through standard blood filters with reasonable efficiency, a sign that they were deformable enough to behave like normal red cells. In contrast, cells grown in solvent-treated plasma alone multiplied less and, most importantly, yielded very few reticulocytes after filtration, suggesting stiffer, less flexible membranes. Direct measurements using a fluorescent cholesterol probe, as well as detailed lipid profiling, confirmed that these plasma-grown reticulocytes were strikingly low in cholesterol compared with both normal blood cells and those grown with AB serum. The cells appeared to sense this shortage: genes and proteins involved in making and importing cholesterol were switched on, indicating that the cells were trying to compensate for a lean environment but could not fully correct it.
Rescuing function by restoring cholesterol
When extra cholesterol-rich lipids were added to the plasma-based medium, many of the problems were reversed. Filter yields returned close to those seen with AB serum, and total cholesterol levels in the reticulocytes rose to match native young red cells. Osmotic fragility tests, which expose cells to increasingly dilute salt solutions, showed that cholesterol-poor reticulocytes burst early, while serum-grown and cholesterol-supplemented cells behaved more like normal, slightly tougher reticulocytes. The team also examined PIEZO1, a membrane channel that senses mechanical stress and helps red cells adjust their volume. In low-cholesterol cells, PIEZO1 responded poorly to a chemical activator, but its activity was restored when cholesterol was replenished. Even the ability of the malaria parasite Plasmodium falciparum to invade the cells tracked with cholesterol content: invasion was reduced in cholesterol-deficient reticulocytes and normalized when cholesterol was brought back.
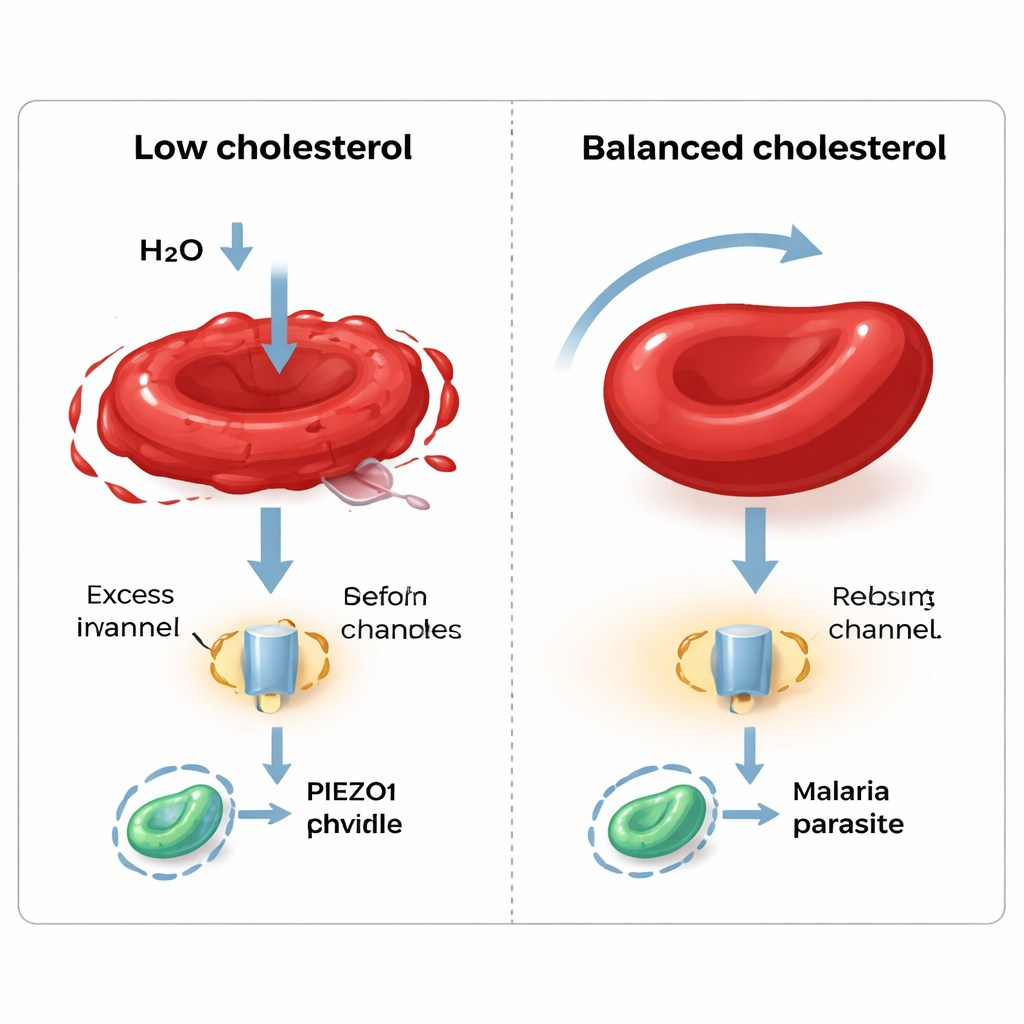
Finding the right balance for future transfusions
Together, these results show that the kind of lipid supplement used during lab growth of red blood cells can strongly shape the cholesterol content, strength, and behavior of the resulting reticulocytes. Human AB serum naturally provides enough cholesterol to support healthy development, whereas some treated plasma products do not, unless specifically supplemented. Adding too much cholesterol can, however, begin to stiffen cells, so careful tuning is needed. For people hoping to receive lab-grown blood in the future, the message is simple: getting the “membrane recipe” right is just as important as growing enough cells. By mapping how fats, metabolites, and proteins shift under different conditions, this work offers a roadmap for standardizing culture media so that lab-grown reticulocytes truly mimic their natural counterparts and are safe, durable, and effective in the bloodstream.
Citation: Freire, C.M., King, N.R., Dzieciatkowska, M. et al. Choice of lipid supplementation for in vitro erythroid cell culture impacts reticulocyte yield and characteristics. Sci Rep 16, 6632 (2026). https://doi.org/10.1038/s41598-026-37229-z
Keywords: lab-grown red blood cells, cholesterol and membranes, reticulocyte culture, blood substitutes, erythropoiesis in vitro